K63 vs K27 Ubiquitin Chains: Decoding Their Divergent Roles in Immune Signaling and Therapeutic Potential
This article provides a comprehensive comparison of K63- and K27-linked ubiquitin chains, two pivotal non-proteolytic regulators of innate and adaptive immunity.
K63 vs K27 Ubiquitin Chains: Decoding Their Divergent Roles in Immune Signaling and Therapeutic Potential
Abstract
This article provides a comprehensive comparison of K63- and K27-linked ubiquitin chains, two pivotal non-proteolytic regulators of innate and adaptive immunity. We explore their foundational biology, distinct structural characteristics, and specific mechanisms in immune pathways such as NF-κB, IRF3 activation, and T-cell regulation. For researchers and drug development professionals, we detail methodological approaches for studying these modifications, address common experimental challenges, and present a direct functional comparison. The synthesis of this information highlights emerging therapeutic opportunities for immune-related diseases, including viral infections, chronic inflammation, and cancer, by targeting these specific ubiquitin linkages.
Ubiquitin Code Fundamentals: K63 and K27 Linkages in Immune System Regulation
The Expanding Landscape of the Ubiquitin Code
Ubiquitination represents a complex post-translational regulatory system where a small 8.6 kDa protein, ubiquitin, tags proteins to dictate their traffic, fate, and function [1]. The versatility of ubiquitin signaling stems from its ability to form diverse chain architectures through different linkage types—creating what is known as the "ubiquitin code" [2] [3]. While K48-linked ubiquitin chains have been extensively characterized as signals for proteasomal degradation, recent research has revealed sophisticated functions for non-degradative ubiquitin chains, particularly K63- and K27-linked chains, in regulating immune signaling pathways [4] [5].
This complex ubiquitin code encompasses homotypic chains (uniform linkages), heterotypic mixed chains (alternating linkages), and branched chains (multiple linkages from a single ubiquitin molecule) [6]. The biological information transmitted by these different chain topologies expands the functional repertoire of ubiquitin far beyond its initial degradation paradigm, especially within immune system regulation.
Comparative Analysis of Ubiquitin Linkages in Immune Signaling
Table 1: Characteristics of Key Ubiquitin Linkages in Immune Regulation
| Linkage Type | Primary Functions | Key E3 Ligases | Associated Deubiquitinases | Immune Pathway Roles |
|---|---|---|---|---|
| K48-Linked | Proteasomal degradation [7] | BRCA1-BARD1, HUWE1 [7] | USP30, USP8 [7] | Limits inflammation by degrading immune signaling components [5] |
| K63-Linked | Signal transduction, protein trafficking, DNA repair [4] [7] | TRAF6, TRAF2, cIAP1/2 [4] | A20, CYLD, OTUD7B, USP5 [4] | NF-κB activation, NLRP3 inflammasome regulation, T/B cell receptor signaling [4] [8] |
| K27-Linked | Balancing immune activation/inhibition [5] | TRIM23 [5] | A20 (indirect) [5] | RLR signaling, NEMO activation, IRF3 induction [5] |
| K11-Linked | Proteasomal degradation, cell cycle regulation [7] | APC/C, UBE2S [7] | USP19 [5] | STING regulation, Beclin-1 degradation, innate immune factor turnover [5] |
| Branched (K48/K63) | Enhanced proteasomal targeting [4] | TRAF6/HUWE1, ITCH/UBR5 [6] | Not specified | NF-κB signaling enhancement [2] [6] |
Table 2: Experimental Approaches for Studying Ubiquitin Linkages
| Methodology | Key Features | Applications | Considerations |
|---|---|---|---|
| Ubiquitin Pulldown + MS [2] [3] | Uses immobilized ubiquitin chains to enrich interactors from cell lysate | Identification of linkage-specific ubiquitin-binding proteins | Requires DUB inhibitors (CAA/NEM); potential off-target effects [2] |
| TUBE-Based Assays [8] | Tandem ubiquitin-binding entities with nanomolar affinity for specific chains | High-throughput screening of linkage-specific ubiquitination | Enables study of endogenous proteins; applicable to PROTAC development [8] |
| UbiCRest [2] | Selective disassembly with linkage-specific DUBs | Verification of chain linkage composition | Uses DUBs like OTUB1 (K48-specific) and AMSH (K63-specific) [2] |
| Mass Spectrometry Ub Clipping [1] | Comprehensive ubiquitin chain linkage analysis | Defining polyubiquitin chain architecture | Used for characterizing branched chains on MHC II [1] |
K63 vs. K27-Linked Chains: Distinct Roles in Immune Signaling Pathways
K63-Linked Ubiquitin Chains: Master Regulators of Immune Activation
K63-linked ubiquitination serves as a major control point for immune signaling, regulating chronic inflammation, pathogen responses, lymphocyte activation, and regulatory signaling [4]. These chains function as scaffolding devices that facilitate the assembly of signaling complexes rather than targeting proteins for degradation.
The Ubc13-Uev1a E2 complex specifically catalyzes K63-linked chain formation [4], which then regulates multiple immune pathways:
NF-κB Signaling: K63 ubiquitination of RIPK1 by the TRAF2-cIAP1-Ubc13-UbcH5 complex enables the formation of TNFR1 Complex I, which activates NF-κB and MAPK-driven transcriptional activation of prosurvival genes [4]. Additionally, K63 chains on NEMO promote IKK complex assembly [8].
NLR and RLR Signaling: Multiple nucleotide-binding oligomerization domain (NOD)-like receptors and RIG-I-like receptors depend on K63 ubiquitination for signal transduction. For instance, NOD2 receptor oligomerization upon muramyldipeptide (MDP) binding recruits RIPK2 and E3 ligases that induce K63 ubiquitination of RIPK2, creating a signaling scaffold for TAK1/TAB1/TAB2/IKK kinase complexes [8].
STING Activation: Stimulator of interferon genes (STING) activation involves K63 ubiquitination, which serves as a docking platform for downstream signaling components in the antiviral response [4].
K27-Linked Ubiquitin Chains: Balancing Immune Activation and Inhibition
While less characterized than K63 linkages, K27-linked chains play crucial balancing roles in immune regulation:
RLR Signaling Activation: TRIM23 conjugates K27-linked chains to NEMO, which is required for the induction of NF-κB and IRF3 upon activation of RLR signaling [5]. These K27-linked chains on NEMO subsequently serve as interaction platforms for other regulatory factors.
Prevention of Excessive Inflammation: K27-linked chains on NEMO recruit Rhbdd3, which in turn recruits the deubiquitinase A20. A20 then removes K63-linked chains from NEMO, preventing excessive NF-κB activation and maintaining immune homeostasis [5].
Direct Comparison of Signaling Mechanisms
The fundamental distinction between these chain types lies in their functional outcomes: K63 chains primarily activate signaling pathways through scaffold formation, while K27 chains appear to provide more nuanced regulation that balances activation with restraint mechanisms to prevent excessive inflammation.
Experimental Approaches for Studying Ubiquitin Linkages
Ubiquitin Interactor Screen Methodology
Recent advances in ubiquitin research have enabled sophisticated analysis of chain-specific interactions:
Chain Synthesis and Immobilization: Enzymatic synthesis of homotypic K48 and K63 Ub2 and Ub3 chains, along with K48/K63 branched Ub3, using specific E2 enzymes (CDC34 for K48, Ubc13/Uev1a for K63) [2] [3]. Chains are immobilized on streptavidin resin via a serine/glycine linker with biotin conjugation.
DUB Inhibition Considerations: Use of deubiquitinase inhibitors (CAA or NEM) to preserve chain integrity during pulldown experiments, with recognition that inhibitor choice affects results—NEM provides nearly complete chain stabilization while CAA allows partial disassembly [2].
Interactor Identification: Enriched proteins are identified via liquid chromatography-mass spectrometry (LC-MS) with statistical comparison of chain-type enrichment patterns [2] [3].
TUBE-Based High-Throughput Applications
Tandem ubiquitin-binding entities (TUBEs) with nanomolar affinities for specific polyubiquitin chains enable high-throughput screening of linkage-specific ubiquitination [8]. This approach allows:
- Specific capture of K63 ubiquitination induced by inflammatory stimuli (e.g., L18-MDP stimulation of RIPK2)
- Detection of K48 ubiquitination induced by PROTAC compounds
- Quantitative assessment of endogenous protein ubiquitination without requiring genetic modification
Ubiquitin Linkages in Immune Signaling Pathways
Essential Research Reagent Solutions
Table 3: Key Research Reagents for Ubiquitin Studies
| Reagent/Category | Specific Examples | Function/Application | Research Context |
|---|---|---|---|
| DUB Inhibitors | Chloroacetamide (CAA), N-ethylmaleimide (NEM) [2] | Preserve ubiquitin chain integrity during experiments | CAA is more cysteine-specific; NEM more potent but with potential off-target effects [2] |
| Linkage-Specific TUBEs | K48-TUBE, K63-TUBE, Pan-TUBE [8] | High-affinity capture of specific ubiquitin linkages | Enable study of endogenous protein ubiquitination in high-throughput formats [8] |
| E2 Enzyme Systems | Ubc13/Uev1a (K63), CDC34 (K48), UBE2S (K11) [2] [7] | Synthesis of linkage-specific ubiquitin chains in vitro | Ubc13 with different cofactors: Mms2 for DNA repair, Uev1a for immune signaling [4] |
| Linkage-Specific DUBs | OTUB1 (K48-specific), AMSH (K63-specific) [2] | Verification of chain linkage composition via UbiCRest | Used analytically to confirm chain linkage types [2] |
| Branched Chain Reagents | K48/K63-branched Ub3 [2] [3] | Study of branched ubiquitin chain functions | Identification of branch-specific interactors like PARP10, UBR4, HIP1 [2] |
The complexity of the ubiquitin code represents both a challenge and opportunity for therapeutic development. The distinct functions of K63-linked chains in immune activation versus K27-linked chains in balanced immune regulation illustrate how understanding linkage-specific ubiquitination can inform drug discovery. Current approaches leveraging this knowledge include PROTACs that exploit K48-linked ubiquitination for targeted protein degradation, while emerging strategies aim to modulate K63-dependent inflammatory pathways [8]. As research methodologies continue to advance—particularly in high-throughput screening and branched chain analysis—the potential for developing more precise therapeutics that target specific ubiquitin linkages continues to grow, offering promising avenues for treating inflammatory diseases, cancer, and neurodegenerative disorders.
Ubiquitination is a crucial post-translational modification that regulates diverse cellular functions, with different ubiquitin chain linkages encoding distinct biological signals. Among these, K63-linked ubiquitination has emerged as a specialized non-proteolytic signal particularly important for immune cell activation, standing in stark contrast to the canonical K48-linked chains that target proteins for proteasomal degradation. This specialized form of ubiquitination serves as a key regulatory mechanism in pattern recognition receptor signaling, lymphocyte activation, and inflammatory responses, functioning primarily as a scaffolding modification that facilitates protein-protein interactions and complex assembly rather than promoting degradation. The distinct roles of K63 and K48 linkages illustrate how the ubiquitin system has evolved to generate specific signals for different cellular processes, with K63 ubiquitination controlling immune signaling pathways through mechanisms that are precisely regulated by specific E3 ligases and deubiquitinating enzymes. Understanding these specialized ubiquitin codes provides critical insights for developing targeted therapeutic strategies for inflammatory diseases, autoimmune disorders, and cancer.
Comparative Functions of K63 vs. K48 Ubiquitination
Table 1: Functional Comparison of K63 and K48 Ubiquitin Linkages
| Feature | K63-Linked Ubiquitination | K48-Linked Ubiquitination |
|---|---|---|
| Primary Function | Non-proteolytic signaling [8] [4] | Proteasomal degradation [8] [7] |
| Key Immune Pathways | NF-κB, MAPK, NLR, TLR, IL-1R signaling [8] [4] | Protein turnover, RIPK2 degradation via PROTACs [8] |
| Cellular Processes | Signal transduction, protein trafficking, DNA repair, autophagy [4] [6] | Protein quality control, cell cycle progression [9] [7] |
| Chain Recognition | TUBEs, UBDs in signaling proteins [8] [10] | Proteasomal receptors, UBDs in degradation machinery [8] |
| Regulatory Enzymes | TRAF6, Ubc13-Uev1a, cIAPs [8] [4] | Various E3 ligases, PROTAC-recruited ligases [8] |
| Deubiquitinases | USP5, CYLD, A20 [9] [4] | Various proteasomal-associated DUBs [7] |
K63 Ubiquitination in Immune Signaling Pathways
Pattern Recognition Receptor Signaling
K63-linked ubiquitination plays an essential role in pattern recognition receptor signaling, serving as a critical scaffold for the assembly of signaling complexes. In NOD2 signaling, bacterial muramyldipeptide (MDP) stimulation induces K63 ubiquitination of RIPK2, which serves as a platform for recruiting and activating the TAK1/TAB1/TAB2/IKK kinase complexes, leading to NF-κB activation and production of proinflammatory cytokines [8]. This process is mediated by E3 ligases including XIAP, which binds RIPK2 via its BIR2 domain and builds K63 ubiquitination on multiple lysine residues of RIPK2 [8]. Similarly, K63 ubiquitination regulates Toll-like receptor (TLR) and RIG-I-like receptor (RLR) pathways through modification of adaptor proteins, creating docking sites for downstream signaling components [4].
T and B Cell Receptor Signaling
In adaptive immunity, K63 ubiquitination is indispensable for lymphocyte activation. T cell receptor (TCR) engagement triggers K63 ubiquitination of multiple signaling components, facilitating the assembly of complexes that lead to NF-κB and MAPK pathway activation [4]. In B cells, K63 ubiquitination regulates B cell receptor (BCR) signaling through TRAF3, which functions as a bridge to transfer ubiquitin chains in a process that ultimately activates the alternative NF-κB pathway [4]. The deubiquitinase CYLD negatively regulates these processes, with CYLD-deficient T cells showing hypersensitivity to TCR/CD3 and TCR/CD28 activation [4].
NF-κB and Inflammatory Signaling
K63 ubiquitination serves as a master regulator of NF-κB signaling through multiple mechanisms. NF-κB essential modulator (NEMO) undergoes K63 ubiquitination, promoting IKK complex assembly and activation of genes related to inflammation [8] [4]. Additionally, K63 ubiquitination contributes to NLRP3 inflammasome activation, which is involved in pro-inflammatory cytokine production [8]. The regulatory significance of this modification is highlighted by the fact that many pathogens, including SARS-CoV-2, specifically target K63 ubiquitination to evade host immune responses [4].
Experimental Analysis of K63 Ubiquitination
Chain-Specific TUBE Methodology
Tandem Ubiquitin Binding Entities (TUBEs) have emerged as powerful tools for studying linkage-specific ubiquitination. These specialized affinity matrices with nanomolar affinities for polyubiquitin chains enable precise capture of chain-specific polyubiquitination events on native target proteins with high sensitivity [8] [10]. The methodology involves:
- Cell Lysis: Cells are lysed in optimized buffer to preserve polyubiquitination states [8]
- Affinity Capture: Lysates are incubated with chain-specific TUBE-coated microplates (96-well format for high-throughput applications) [8] [10]
- Target Detection: Captured ubiquitinated proteins are detected using target-specific antibodies [8]
This approach has been successfully applied to investigate the ubiquitination dynamics of RIPK2, demonstrating that inflammatory agent L18-MDP stimulates K63 ubiquitination that can be faithfully captured using K63-TUBEs or pan-selective TUBEs but not K48-TUBEs [8]. Conversely, RIPK2 PROTAC-mediated ubiquitination is captured using K48-TUBEs and pan-selective TUBEs but not K63-TUBEs [8].
Ub-DiGGer Proteomic Approach
For comprehensive mapping of K63 ubiquitination sites, the Ub-DiGGer method provides a sequential enrichment strategy that preserves linkage type information while extracting modification sites with high specificity and proteome coverage [11]. This methodology involves:
- SILAC Labeling: Cells are cultured in heavy and light isotope-containing media [11]
- Stress Induction: Oxidative stress induced by H₂O₂ treatment [11]
- TUBE Enrichment: Initial enrichment using FLAG K63-TUBE peptide [11]
- Diglycine Remnant Capture: Trypsin digestion followed by anti-GG antibody enrichment [11]
- LC-MS/MS Analysis: Identification and quantification of K63 ubiquitination sites [11]
This approach identified over 1,100 K63 ubiquitination sites in yeast responding to oxidative stress, revealing modification of proteins involved in translation, ion transport, and protein trafficking [11].
K63 vs. K27 Linkages in Immune Signaling
Table 2: Comparative Analysis of K63 and K27 Ubiquitin Linkages in Immune Signaling
| Characteristic | K63-Linked Chains | K27-Linked Chains |
|---|---|---|
| Established Immune Functions | Well-established in PRR, TCR, BCR, cytokine signaling [4] | Less characterized, emerging roles in immune regulation |
| Signaling Mechanisms | Scaffold for signalosome assembly [8] [4] | Proposed role in kinase complex assembly [7] |
| Pathway Associations | NF-κB, MAPK, NLRP3 inflammasome [8] [4] | IFN signaling, DNA damage response [7] |
| Regulatory Enzymes | TRAF6, cIAPs, Ubc13-Uev1a [8] [4] | Limited characterization of specific E3s [7] |
| Known Deubiquitinases | USP5, CYLD, A20, OTUD7B [9] [4] | Poorly defined DUB specificity [7] |
| Therapeutic Targeting | Multiple inhibitors in development [8] [4] | Limited targeted approaches |
Key Signaling Pathways and Experimental Workflows
Research Reagent Solutions
Table 3: Essential Research Tools for K63 Ubiquitination Studies
| Reagent/Tool | Specific Function | Application Examples |
|---|---|---|
| Chain-Specific TUBEs | High-affinity capture of linkage-specific ubiquitin chains [8] [10] | RIPK2 ubiquitination dynamics, HTS assays [8] |
| K63-Ub Antibodies | Detection of K63-linked chains in Western blot [11] | Verification of K63 ubiquitination in oxidative stress [11] |
| Ubc13-Uev1a Complex | K63-specific E2 enzyme complex [4] | In vitro ubiquitination assays, mechanism studies [4] |
| USP5 Enzymes | K63-specific deubiquitinase [9] [4] | Deubiquitination assays, necroptosis regulation studies [9] |
| Ub-DiGGer Platform | Sequential enrichment for K63 site mapping [11] | Proteomic identification of K63 sites in stress response [11] |
| SMURF1/USP5 System | Opposing regulation of RIPK3 K63 ubiquitination [9] | Necroptosis mechanism studies, cancer models [9] |
K63-linked ubiquitination represents a sophisticated non-proteolytic signaling mechanism that is indispensable for proper immune cell activation and function. Through its role as a scaffold for signalosome assembly, this specific ubiquitin linkage controls critical pathways including NF-κB signaling, inflammasome activation, and lymphocyte responses. The development of specialized research tools such as chain-specific TUBEs and advanced proteomic methods has significantly enhanced our ability to study these processes with unprecedented specificity. As our understanding of the complexity of ubiquitin codes deepens, particularly with the emerging recognition of branched chains incorporating K63 linkages, new therapeutic opportunities continue to emerge for modulating immune responses in disease contexts. The continued refinement of experimental approaches to differentiate K63 signaling from other linkages such as K27 will undoubtedly yield new insights into the sophisticated language of ubiquitin in immune regulation.
Ubiquitination is a crucial post-translational modification that regulates virtually every cellular process, from protein degradation to immune signaling. The functional diversity of ubiquitination is largely governed by the type of polyubiquitin chain linkage formed between ubiquitin molecules. While K63-linked chains have been extensively characterized as key regulators of immune signaling pathways, K27-linked ubiquitination has emerged as a structurally and functionally unique player with distinctive properties, including remarkable resistance to deubiquitinating enzymes (DUBs). Understanding the contrast between these chain types is fundamental for researchers and drug development professionals seeking to target ubiquitin signaling in immune-related diseases and cancer.
This review provides a comprehensive comparison between K27 and K63 ubiquitin linkages, focusing on their structural characteristics, functional roles in immune signaling, and the experimental tools available for their study. We present consolidated experimental data and methodologies to facilitate research in this rapidly evolving field.
Structural and Functional Comparison of K27 and K63 Linkages
Table 1: Fundamental Properties of K27 vs. K63 Ubiquitin Linkages
| Property | K27-Linked Ubiquitination | K63-Linked Ubiquitination |
|---|---|---|
| Structural Conformation | Adopts open conformations capable of bidentate binding to receptors [12] | Extended, open conformation optimal for signaling complex assembly |
| DUB Susceptibility | Resistant to cleavage by most deubiquitinases (DUBs) [12] | Susceptible to various DUBs (e.g., A20, CYLD, OTUDs) [13] [14] |
| Primary Cellular Functions | DNA damage response, immune signaling [15] | NF-κB and MAPK activation, signal transduction, protein trafficking [16] [17] |
| Representative E2 Enzymes | Ube2D2 (versatile, requires specific context) [15] | Ubc13 (in complex with Uev1A/Mms2) [17] |
| Representative E3 Ligases | RNF168 (H2A), TRIM23 (NEMO) [15] | TRAF6, TRAF2, cIAP1/2 [16] [17] |
| Representative Binding Domains | Binds UBA2 domain of hHR23A similarly to K48 chains [12] | Recognized by NZF (Npl4 zinc finger), UIM (Ubiquitin-interacting motif) domains [14] |
| Role in NF-κB Pathway | Modifies NEMO via TRIM23; antiviral immunity [15] | Modifies RIP1, RIP2, NEMO; pro-inflammatory signaling [13] [14] |
Table 2: Experimental Evidence for Distinct Functional Roles
| Experimental Context | K27-Linked Chain Findings | K63-Linked Chain Findings |
|---|---|---|
| In Vitro Biochemical Assays | Not cleaved by most DUBs; adopts unique conformations by NMR and SANS [12] | Efficiently cleaved by specific DUBs (e.g., A20, CYLD) [13] |
| Immune Signaling Studies | RNF168 catalyzes K27 on H2A for DNA damage response [15] | TRAF6/Ubc13 catalyze K63 on RIPK2 for NOD2 inflammatory signaling [16] [8] |
| Pathogen Response | TRIM23 mediates K27 linkage on NEMO for antiviral innate immunity [15] | Ubc13 catalyzes K63 on STING for antiviral immunity, including SARS-CoV-2 [17] |
| Tools for Identification | Challenging due to lack of specific antibodies; requires engineered OUT pathways [15] | Can be studied with chain-specific TUBEs, antibodies, and mass spectrometry [16] [8] |
Unique Structural Characteristics and DUB Resistance of K27 Linkages
Among all lysine linkages, K27-linked ubiquitin chains display exceptional biochemical properties. Nuclear magnetic resonance (NMR) and small-angle neutron scattering (SANS) analyses reveal that K27-Ub2 adopts open conformations in solution that are capable of bidentate interactions with ubiquitin receptors, similar to K48-linked chains [12]. This structural feature suggests a mechanism for specific protein-protein interactions in signaling cascades.
Most notably, K27-linked ubiquitin chains demonstrate pronounced resistance to cleavage by most deubiquitinating enzymes. This property was systematically established through comprehensive DUB assays comparing all possible lysine linkages [12]. The structural basis for this DUB resistance likely stems from the unique conformation of K27 chains, which may occlude the catalytic sites of many DUBs. This characteristic has significant implications for the temporal regulation of K27-mediated signaling, potentially resulting in more sustained signals compared to other linkage types.
Distinct Roles in Immune Signaling Pathways
K63-Linked Ubiquitination: The Master Regulator of Pro-Inflammatory Signaling
K63-linked ubiquitination serves as a central mechanism for activating NF-κB and MAPK signaling pathways, which govern the production of pro-inflammatory cytokines and immune cell activation [17] [14]. This linkage type functions primarily through non-proteolytic mechanisms by serving as a scaffolding platform that facilitates the assembly of signaling complexes.
Key molecular events in K63-mediated immunity include:
- NOD2/RIPK2 Signaling: Upon sensing bacterial muramyl dipeptide (MDP), the NOD2 receptor recruits RIPK2 and E3 ligases (XIAP, cIAP1/2, TRAF2) that catalyze K63-linked ubiquitination of RIPK2. This creates a scaffold that recruits TAK1/TAB1/TAB2 and IKK complexes, leading to NF-κB activation [16] [8].
- TLR/IL-1R Signaling: Activation of Toll-like receptors (TLRs) and interleukin-1 receptor (IL-1R) leads to the recruitment of MyD88 and IRAK1/4, which activate TRAF6. TRAF6, in conjunction with Ubc13, catalyzes K63 ubiquitination that activates TAK1 and IKK complexes [13] [14].
- T Cell Regulation: K63 ubiquitination regulates T cell receptor (TCR) signaling and the balance between regulatory T cells (Tregs) and inflammatory effector T cell subsets [17].
The diagram below illustrates the central role of K63 ubiquitination in NOD2 inflammatory signaling:
K27-Linked Ubiquitination: A Specialized Player in Immune Regulation
While less comprehensively characterized, K27-linked ubiquitination participates in specialized immune signaling contexts with distinct regulatory properties:
- Antiviral Innate Immunity: TRIM23 (tripartite motif-containing protein 23) catalyzes K27-linked ubiquitination of NEMO (NF-κB essential modulator), which is crucial for antiviral innate immunity and inflammatory responses [15].
- DNA Damage Response: RNF168 mediates K27-linked ubiquitination of histone H2A, which is essential for activation of the DNA damage response [15].
- Inflammasome Regulation: The HECT domain-containing E3 ligase HUWE1 modifies NLRP3 through K27 chains to regulate inflammasome activity [13].
The DUB-resistant nature of K27 linkages suggests they may provide more stable signaling platforms in these pathways, potentially serving as sustained modification signals that persist after K63-linked chains have been disassembled by DUBs.
Experimental Approaches for Studying K27 and K63 Linkages
Methodologies for K27-Linked Ubiquitination Research
Studying K27 linkages presents unique challenges due to the lack of specific antibodies and their low abundance. The Orthogonal Ubiquitin Transfer (OUT) pathway has been engineered to specifically identify K27 chain linkage substrates [15]:
Table 3: Orthogonal Ubiquitin Transfer (OUT) Protocol for K27 Substrate Identification
| Step | Description | Key Reagents/Plasmids |
|---|---|---|
| 1. System Design | Engineer mutant ubiquitin (xUb-K27) with only K27 lysine resident | pET28a-HA-xUb-K27 plasmid [15] |
| 2. E1-E2 Engineering | Create xUba1-xUbe2D2 pairs that transfer xUb-K27 but not wild-type Ub | xUba1-f+b4/xUba1-f+b6 and xUbe2D2-9 mutants [15] |
| 3. In Vitro Ubiquitination | Incubate engineered system with wild-type E3s to form K27 linkages | ATP, E1/E2 enzymes, TRIM23-Myc or CHIP E3s [15] |
| 4. Substrate Identification | Purify and identify proteins modified with K27 chains | His-purification, mass spectrometry [15] |
The experimental workflow for the OUT pathway is summarized below:
Methodologies for K63-Linked Ubiquitination Research
K63-linked ubiquitination can be studied using more established methods, including chain-specific affinity tools:
Table 4: TUBE-Based Assay for K63 Ubiquitination Detection
| Step | Description | Key Reagents |
|---|---|---|
| 1. Cell Stimulation | Treat THP-1 cells with L18-MDP to induce K63 ubiquitination of RIPK2 | L18-MDP (200-500 ng/mL, 30-60 min) [16] [8] |
| 2. Cell Lysis | Lyse cells with buffer that preserves polyubiquitination | Lysis buffer with protease and DUB inhibitors [16] |
| 3. Affinity Capture | Incubate lysates with chain-specific TUBEs in 96-well plate | K63-TUBEs (LifeSensors UM401M) [16] [8] |
| 4. Target Detection | Detect ubiquitinated targets by immunoblotting | Anti-RIPK2 antibody [16] |
The Scientist's Toolkit: Essential Research Reagents
Table 5: Key Reagents for Studying K27 and K63 Ubiquitination
| Reagent/Tool | Specificity | Research Application | Key Features |
|---|---|---|---|
| K63-TUBEs | K63-linked chains | Capture and detection of endogenous K63-ubiquitinated proteins [16] [8] | Nanomolar affinity; works in HTS formats; preserves labile ubiquitination |
| Orthogonal OUT System | K27-linked chains | Identification of K27-specific substrates in cells [15] | Engineered xE1-xE2 pairs transfer xUb-K27 to wild-type E3s |
| Ubiquitin Mutants (xUb-K27) | K27 linkage formation | Study chain formation and substrate identification [15] | All lysines mutated to arginine except K27 |
| Ube2D2 Mutants | K27 chain transfer | Engineered E2 for orthogonal ubiquitin transfer [15] | Versatile E2 that forms various chains; works with OUT system |
| L18-MDP | NOD2/RIPK2 pathway activator | Induces K63 ubiquitination of RIPK2 in cellular assays [16] [8] | Potent muramyldipeptide derivative; activates inflammatory signaling |
| PR619 | Pan-DUB inhibitor | Broad inhibition of cysteine DUBs to study ubiquitination dynamics [18] | Increases global ubiquitination; reveals DUB-regulated substrates |
K27-linked ubiquitination represents a structurally and functionally unique regulatory modification characterized by its extended conformation and notable resistance to deubiquitinating enzymes. While K63 linkages serve as master regulators of pro-inflammatory signaling through well-characterized scaffolding functions, K27 linkages play more specialized roles in processes including antiviral immunity and DNA damage response. The contrasting properties of these linkage types highlight the complexity of the ubiquitin code in immune regulation.
Future research will benefit from continued development of linkage-specific tools, particularly for the challenging study of K27 ubiquitination. The engineered OUT system provides a powerful approach for identifying K27 substrates, while TUBE-based technologies enable sensitive detection of K63 linkages in high-throughput formats. Understanding the nuanced functions of these distinct ubiquitin linkages will advance targeted therapeutic development for inflammatory diseases, cancer, and immune disorders.
Ubiquitination is a crucial post-translational modification that regulates numerous cellular processes, with the functional outcome largely determined by the topology of the polyubiquitin chain. Among the different linkage types, K63-linked and K27-linked ubiquitin chains have emerged as critical non-degradative signals in immune signaling pathways [4] [7]. While K63-linked chains are well-established regulators of inflammatory responses, DNA damage repair, and adaptive immunity, K27-linked chains represent a more enigmatic class with unique structural properties and emerging roles in immune regulation [19] [7]. This comparison guide examines the key enzymatic machinery—specifically the E2 enzyme Ubc13 and E3 ligases including TRAFs and Cullin-3—responsible for assembling these chains, providing experimental data and methodologies for researchers studying ubiquitination in immune signaling.
Comparative Analysis of K63 and K27 Enzymatic Machinery
The following table summarizes the core enzymatic components, mechanisms, and functional properties of the K63 and K63 linkage machinery.
| Feature | K63-Linked Ubiquitination Machinery | K27-Linked Ubiquitination Machinery |
|---|---|---|
| Key E2 Enzyme | Ubc13 (in heterodimer with Uev1A/Mms2) [20] | Not well-defined; multiple E2s may be involved [19] |
| Key E3 Ligases | TRAF6, TRAF2, RNF4, RNF8 [4] [21] | RNF185, AMFR, Parkin, Cullin-3 complexes [21] [7] |
| Catalytic Mechanism | RING E3-dependent; Ubc13/Uev1A positions donor and acceptor ubiquitin [22] [20] | Not fully elucidated; may involve RING or HECT-type E3s [19] |
| Complex Formation | Requires non-catalytic E2 variant (Uev1A/Mms2) for specificity [20] [23] | No known requirement for E2 variants |
| Structural Features | "Folded-back" E2~Ub conformation stabilized by RING E3 [22] | Restricted conformation with limited interdomain contacts [19] |
| Linkage Specificity | Highly specific for K63 linkage [20] | Specificity determined by E3 ligase [19] |
| DUB Sensitivity | Sensitive to specific DUBs (e.g., CYLD, A20) [4] | Resistant to most deubiquitinases (e.g., USP2, USP5) [19] |
Detailed Enzyme Profiles and Experimental Analysis
K63-Linked Chain Synthesis: Ubc13 and Partner E3 Ligases
Ubc13-Uev1A Heterodimer Mechanism: Ubc13, in complex with a non-catalytic E2 variant (Uev1A in cytoplasm or Mms2 in nucleus), forms the dedicated enzymatic core for K63-linked chain synthesis [20]. The mechanism involves Ubc13 forming a thioester bond with the donor ubiquitin (E2~Ub), while Uev1A/Mms2 non-covalently binds an acceptor ubiquitin, positioning its K63 residue for nucleophilic attack on the thioester bond [20]. The reaction is facilitated by Ubc13 Asn79, which stabilizes the oxyanion thioester intermediate, and Asn123, which engages the acceptor ubiquitin Lys63 [20].
RING E3 Ligase Activation: RING E3 ligases such as RNF4 and RNF8 activate Ubc13~Ub by binding both the E2 and donor ubiquitin, locking the ubiquitin in an active "folded-back" conformation that optimizes the geometry for nucleophilic attack [22] [20]. Structural studies of RNF4 with Ubc13~Ub and Ube2V2 have captured this activated state, showing Ube2V2 bound to the acceptor ubiquitin with K63 positioned to attack the thioester linkage [22].
TRAF Family E3 Ligases in Immune Signaling: TRAF6, a key RING E3 in immune signaling, catalyzes K63-linked ubiquitination of various substrates including itself during NF-κB activation [4] [14]. TRAF6 collaborates with the Ubc13-Uev1A heterodimer to build K63 chains that serve as docking platforms for TAK1 complex recruitment via ubiquitin-binding domains, leading to IKK and NF-κB activation [4] [14].
K27-Linked Chain Synthesis: Emerging Enzymatic Machinery
E3 Ligases with K27 Specificity: Unlike K63 linkage formation, the E2 enzymes for K27 linkage are not well characterized, with linkage specificity appearing to be determined primarily at the E3 level [19]. RNF185 and AMFR have been identified as E3 ligases that catalyze K27-linked ubiquitination of mitochondrial proteins and the STING adaptor in innate immune signaling, respectively [21] [7]. Parkin, an RBR E3 ligase associated with Parkinson's disease, has been reported to assemble K27-linked chains alongside other linkage types during mitophagy [7].
Cullin-3 Complex Potential: While not explicitly documented for K27 linkages in the literature reviewed, Cullin-3-based RING E3 ligases represent potential candidates for K27 chain formation given their diverse substrate specificity and involvement in immune regulation, though further research is needed to establish this connection.
Unique Structural Properties: K27-linked di-ubiquitin exhibits distinctive structural characteristics, including the smallest chemical shift perturbations in the distal ubiquitin among all linkage types, suggesting limited non-covalent interdomain contacts [19]. This restricted conformation may explain its unique resistance to most deubiquitinases, setting it apart from other ubiquitin linkages [19].
Experimental Protocols for Key Studies
In Vitro K63 Ubiquitination Assay (RNF4/Ubc13/Ube2V2)
Objective: To reconstitute K63-linked ubiquitin chain synthesis in vitro and assess the requirement for specific complex components [22].
Methodology:
- Reaction Setup: Combine 50-100 nM human E1 enzyme, 5 µM Ubc13, 5 µM Ube2V2, 2.5 µM RNF4 RING dimer, and 0.5-1 µM ubiquitin-primed tetraSUMO substrate in reaction buffer (50 mM Tris-HCl pH 7.5, 50 mM KCl, 5 mM MgCl₂, 2 mM ATP) [22].
- Ubiquitin Activation: Initiate reaction by adding 200 µM ubiquitin and 2 mM ATP to the master mix.
- Time Course Incubation: Incubate at 30°C for 0-60 minutes, removing aliquots at specified timepoints.
- Reaction Termination: Stop by adding SDS-PAGE loading buffer with DTT to reduce thioester bonds.
- Analysis: Resolve proteins by SDS-PAGE, transfer to membrane, and probe with K63-linkage specific antibody to detect chain formation [22].
Key Controls:
- Omit Ube2V2 to demonstrate requirement for E2 variant
- Omit RNF4 to show E3 dependence
- Include catalytically inactive Ubc13 (C87A) as negative control
- Test unprimed tetraSUMO substrate to show priming requirement
Deubiquitination Resistance Assay for K27 Linkages
Objective: To characterize the unique resistance of K27-linked chains to deubiquitinase cleavage [19].
Methodology:
- Substrate Preparation: Generate fully natural K27-Ub2 with native isopeptide linkages using non-enzymatic chemical assembly methods with mutually orthogonal removable amine-protecting groups (Alloc and Boc) [19].
- DUB Screening: Incubate 5 µg of K27-Ub2 with 0.5-1 µM of various DUBs (Cezanne, OTUB1, AMSH, USP2, USP5, Ubp6) in appropriate reaction buffers at 37°C for 30-60 minutes.
- Reaction Analysis: Terminate reactions with SDS loading buffer, analyze by SDS-PAGE and Coomassie staining, or use NMR spectroscopy to monitor chain disassembly.
- Comparative Assessment: Parallel testing with other linkage types (K6-, K11-, K29-, K33-, K48-Ub2) to demonstrate unique K27 resistance.
Expected Results: K27-Ub2 shows complete resistance to cleavage by linkage-nonspecific DUBs (USP2, USP5, Ubp6), while other linkages are efficiently processed under identical conditions [19].
Signaling Pathways and Molecular Interactions
K63 Ubiquitination in Immune Signal Transduction
The diagram below illustrates the central role of Ubc13-dependent K63 ubiquitination in pattern-recognition receptor (PRR) signaling pathways.
Mechanistic Insights: Upon PRR activation, adaptor proteins (MyD88 for TLRs, MAVS for RLRs) recruit TRAF family E3 ligases, which in turn engage the Ubc13-Uev1A E2 complex [4] [14]. K63-linked ubiquitin chains assembled on signaling components (e.g., RIP1, IRAK1) or on the E3 itself serve as docking platforms for proteins containing ubiquitin-binding domains (UBDs), particularly the TAK1 complex, leading to downstream activation of NF-κB and MAPK pathways and induction of proinflammatory cytokines [4] [14].
K27 Ubiquitination in Immune Regulation
The diagram below outlines the emerging roles of K27 ubiquitination in immune regulatory pathways.
Mechanistic Insights: K27-linked ubiquitination is catalyzed by specific E3 ligases in response to immune stimuli. RNF185 and AMFR target the STING adaptor in the DNA-sensing pathway for K27-linked ubiquitination, promoting antiviral type I interferon responses [21] [7]. Parkin assembles K27 chains on mitochondrial proteins like Miro1 during mitochondrial damage, regulating mitophagy [7]. The unique resistance of K27 linkages to most deubiquitinases enables persistent signaling, which may be particularly important for sustained immune responses and mitochondrial quality control [19].
Research Reagent Solutions
The following table compiles key reagents and their applications for studying K63 and K27 ubiquitination.
| Reagent Category | Specific Examples | Research Application | Key Features |
|---|---|---|---|
| E2 Enzymes | Recombinant Ubc13/Uev1A heterodimer | K63 chain formation assays | Forms stable complex; specific for K63 linkage [20] |
| E3 Ligases | TRAF6, RNF4, RNF8 RING domains | E3-dependent ubiquitination | Requires dimerization for activity [22] [4] |
| E3 Ligases | RNF185, AMFR, Parkin | K27 linkage studies | Potential K27-specific E3s [21] [7] |
| Substrates | Ubiquitin-primed tetraSUMO | In vitro ubiquitination | Mimics physiological primed substrate [22] |
| Chemical Tools | K27-Ub2 with native isopeptide bond | DUB resistance studies | Generated via non-enzymatic assembly [19] |
| Stable Mimetics | Ubc13~Ub isopeptide conjugate | Structural studies | Mimics E2~Ub thioester for crystallography [22] |
| Detection Reagents | K63-linkage specific antibodies | Western blot detection | Specific recognition of K63 linkage [22] |
| DUBs | USP2, USP5, OTUB1, AMSH | Linkage specificity profiling | Assess cleavage resistance of different chains [19] |
This comparison guide highlights the distinct enzymatic machinery and properties of K63 and K27 ubiquitination systems. The Ubc13-Uev1A heterodimer represents a highly specialized E2 complex dedicated to K63 chain formation, working with RING E3s like TRAF6 and RNF4 to assemble chains critical for immune signal transduction [22] [4] [20]. In contrast, K27 linkage formation relies primarily on E3 ligase specificity, with emerging candidates including RNF185 and AMFR, and produces chains with unique structural properties and exceptional resistance to deubiquitination [19] [21] [7]. For researchers investigating ubiquitin signaling in immune pathways, these distinct enzymatic systems offer complementary mechanistic insights—with K63 linkages providing a paradigm for reversible, dynamic signaling and K27 linkages representing a more persistent regulatory modification. Future research should focus on identifying the specific E2 enzymes for K27 linkage formation and developing more sensitive tools to detect and manipulate these modifications in physiological contexts.
The innate immune response must be potent enough to combat pathogens yet tightly controlled to prevent immunopathology. Ubiquitination, a key post-translational modification, regulates the stability, activity, and localization of countless proteins in these signaling pathways [24]. Deubiquitinases (DUBs) perform the critical function of reversing ubiquitination, allowing for dynamic and precise control of immune activation. Among the numerous DUBs, A20 (also known as TNFAIP3), CYLD (cylindromatosis), and USP19 (ubiquitin-specific protease 19) have emerged as essential negative regulators. Their activity is particularly significant in the context of different polyubiquitin chain linkages, especially the non-degradative K63-linked chains that promote signal transduction and the less-understood K27-linked chains. This guide provides a comparative analysis of these three DUBs, focusing on their mechanisms, substrates, and specific roles in immune regulation, providing researchers with a structured overview of their functions and the experimental methods used to characterize them.
Molecular Mechanisms and Specificity
A20, CYLD, and USP19 employ distinct molecular strategies to deubiquitinate their substrates, with varying linkage specificities that determine their biological roles.
Table 1: Molecular Characteristics and Specificity of A20, CYLD, and USP19
| Feature | A20 (TNFAIP3) | CYLD | USP19 |
|---|---|---|---|
| DUB Family | OTU (Ovarian Tumor protease) | USP (Ubiquitin-Specific Protease) | USP (Ubiquitin-Specific Protease) |
| Primary Linkage Specificity | Preferentially K63-linked and M1-linked (linear) chains [25] | Preferentially K63-linked and M1-linked chains [25] [26] | Primarily K11-linked chains; also K63-linked [5] |
| Catalytic-Independent Functions | Yes, acts as a scaffold to inhibit E3 ligases [25] | Predominantly catalytic activity-dependent | Information Not Specified in Search Results |
| Key Protein Domains | OTU domain (catalytic), A20-type Zinc Finger (ZnF) domains [25] | USP domain, CAP-Gly domains (for microtubule/signaling protein binding) [25] [26] | USP domain |
A20 is a unique DUB that employs both enzymatic and non-enzymatic mechanisms. Its catalytic OTU domain hydrolyzes K63-linked and M1-linked ubiquitin chains from substrates like RIPK1 and TRAF6 [25]. Independently of its deubiquitinase activity, A20 can also function as a scaffold protein, using its A20-type zinc finger domains to inhibit E3 ubiquitin ligases, thereby preventing the addition of activating ubiquitin chains [25]. In contrast, CYLD's activity is largely dependent on its catalytic USP domain, which is structurally specialized for recognizing and cleaving K63-linked and M1-linked polyubiquitin chains [25] [26]. Its N-terminal CAP-Gly domains facilitate substrate targeting by interacting with proteins such as NEMO [25]. USP19, another USP family member, shows a different specificity profile, primarily cleaving K11-linked ubiquitin chains and being implicated in the regulation of K63-linked chains on specific substrates like Beclin-1 to modulate autophagy and immune signaling [5].
Roles in Immune Signaling Pathways and Disease
These DUBs are pivotal negative regulators in multiple innate immune signaling pathways, and their dysfunction is linked to human diseases.
3.1 Regulation of Antiviral and NF-κB Signaling A20 and CYLD are central negative regulators of the NF-κB signaling pathway activated by cytokine receptors (e.g., TNFR), antigen receptors, and pattern recognition receptors (PRRs) [25]. A20 deubiquitinates key signaling molecules including RIPK1, RIPK2, TRAF6, and NEMO, thereby shutting down NF-κB activation and preventing excessive inflammation [25]. Similarly, CYLD removes K63-linked and M1-linked chains from a wide array of NF-κB signaling components, including TRAF2, TRAF6, RIPK1, TAK1, and NEMO [25]. In the context of antiviral immunity, a systematic CRISPR/Cas9 screen highlighted the broad and dynamic involvement of DUB family proteins, including A20 and CYLD, in feedback loops that modulate the type I interferon response [24]. USP19, meanwhile, regulates the RIG-I/MAVS antiviral pathway indirectly by controlling the stability of Beclin-1. USP19 removes K11-linked ubiquitin chains from Beclin-1, preventing its proteasomal degradation. Stabilized Beclin-1 then promotes autophagy and inhibits the interaction between RIG-I and MAVS, ultimately limiting the production of type I interferons [5].
3.2 Implications in Cancer and Inflammatory Disease The critical regulatory functions of these DUBs are underscored by the pathological consequences of their dysregulation. CYLD acts as a tumor suppressor, and its loss leads to constitutive NF-κB activation, promoting the expression of pro-survival genes and the development of skin tumors (cylindromatosis) [25]. A20 is a crucial gatekeeper against autoimmune and inflammatory diseases; mutations in its gene, TNFAIP3, are associated with a range of inflammatory pathologies [25]. While the direct disease links for USP19 are less clear in the provided search results, its role in stabilizing oncogenic proteins like c-Myc through deubiquitination suggests a potential role in tumorigenesis [27].
K63 vs. K27 Linked Chains in Immune Regulation
The functional outcome of ubiquitination is largely dictated by the type of polyubiquitin chain formed. K63-linked and K27-linked chains are both "non-degradative" but play distinct roles in immune signaling.
K63-Linked Ubiquitin Chains: These chains are well-established as key activators of immune signaling pathways. They serve as platforms for the assembly of large signaling complexes, such as those formed around RIPK1 in the TNFR pathway or around TRAF6 in the TLR/IL-1R pathways [25] [27]. The primary role of A20 and CYLD as negative regulators is to dismantle these K63-linked chain-based platforms to terminate signaling [25]. Their specificity for K63-linked chains makes them master regulators of inflammation and immunity.
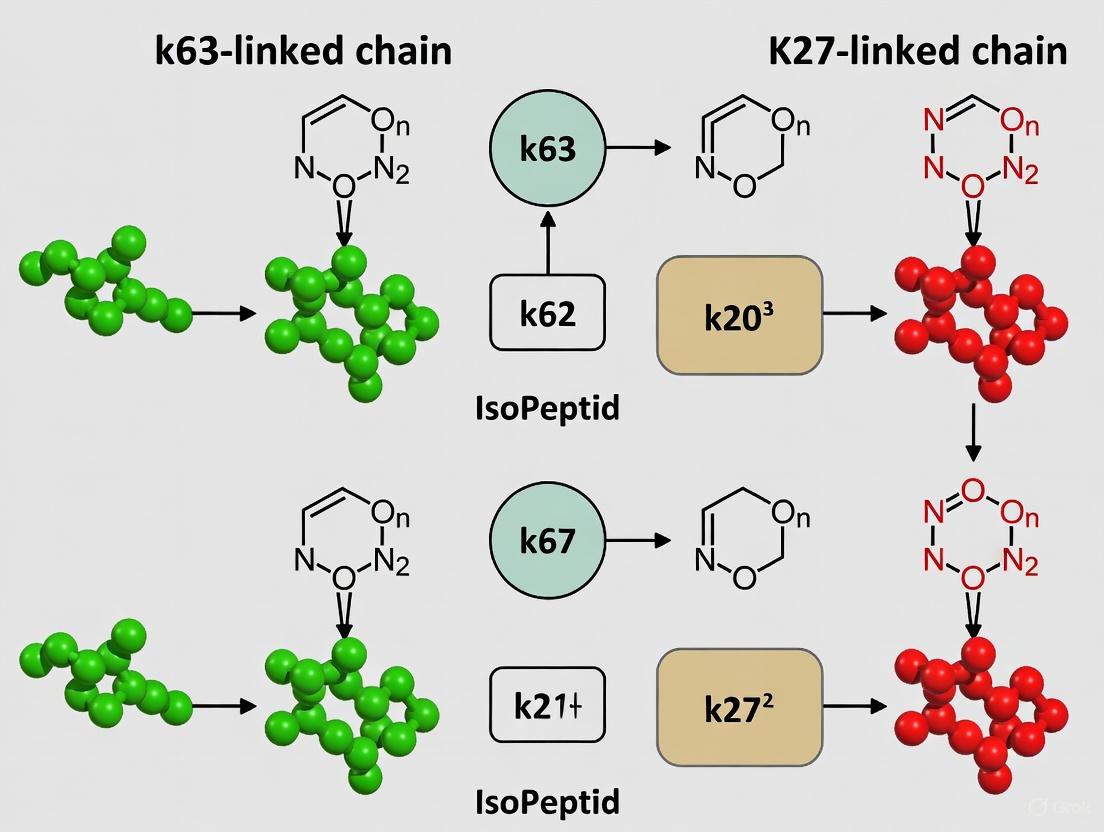
K27-Linked Ubiquitin Chains: The role of K27-linked chains is less defined but emerging as crucial. Structurally, K27 is the least solvent-exposed lysine in ubiquitin, making K27-linked chains highly resistant to cleavage by most DUBs [19] [28]. Functionally, K27-linked ubiquitination has been implicated in both activation and inhibition of immune signaling. For instance, the E3 ligase TRIM23 conjugates K27-linked chains to NEMO, which is required for the induction of NF-κB and IRF3 upon RLR activation [5]. Conversely, K27-linked chains on NEMO can also recruit proteins like Rhbdd3 and A20 to deubiquitinate K63-linked chains and prevent excessive NF-κB activation [5]. This dual role highlights the complexity of the ubiquitin code. The extreme resistance of K27-linked chains to DUBs [19] suggests they may provide a more stable signal, potentially for sustained responses or for recruiting specific effectors that are not easily displaced.
Experimental Data and Methodologies
The characterization of DUB specificity and function relies on a suite of well-established biochemical and cellular assays.
Table 2: Key Experimental Assays for DUB Characterization
| Assay Type | Description | Key Findings from Literature |
|---|---|---|
| In Vitro Deubiquitination Assay | Purified DUB is incubated with defined ubiquitin chains (e.g., K63-Ub~4~, K48-Ub~4~). Cleavage is monitored by gel shift or mass spectrometry. | Used to establish CYLD's preference for K63-linked and M1-linked chains over K48-linked chains [25]. Revealed USP53/USP54 (not focus DUBs) are highly specific for K63-linked chains [29]. |
| CRISPR/Cas9 Functional Screen | Cells with individual DUBs knocked out are stimulated (e.g., with VSV) and tested for phenotypes like IFN-β production or virus replication [24]. | Identified novel roles for USP5, USP14, USP22, and others in inhibiting antiviral immunity [24]. |
| Linkage-Specific Ubiquitin Probes | Activity-based probes with ubiquitin C-terminal warhead (e.g., Ub-PA) form covalent bonds with active DUBs for profiling and identification [29]. | Used to confirm the catalytic activity and specificity of DUBs like USP54, revising previous annotations of inactivity [29]. |
| Co-immunoprecipitation & In Vivo Ubiquitination | DUB is co-expressed with substrates; ubiquitination status of substrate is examined with or without DUB presence, often under proteasome inhibition. | Validated RIPK1, TRAF6, and NEMO as substrates for CYLD and A20 [25]. Confirmed BRAF is modified by non-proteolytic K27-linked chains [30]. |
Detailed Protocol: In Vitro DUB Specificity Assay
- Protein Purification: Express and purify the catalytic domain of the DUB of interest (e.g., CYLD USP domain) from E. coli or insect cells.
- Substrate Preparation: Source a panel of purified polyubiquitin chains of defined linkages (K11, K27, K29, K33, K48, K63, M1). These can be produced enzymatically with specific E2 enzymes or via chemical synthesis.
- Reaction Setup: Incubate the DUB (e.g., 100 nM) with the different ubiquitin chain substrates (e.g., 1 µM) in a suitable reaction buffer (e.g., 50 mM Tris-HCl pH 7.5, 150 mM NaCl, 1 mM DTT) at 37°C.
- Time-Course Sampling: Remove aliquots from the reaction at various time points (e.g., 0, 5, 15, 30, 60 minutes) and quench the reaction with SDS-PAGE loading buffer.
- Analysis: Analyze the quenched samples by SDS-PAGE and Coomassie blue staining or immunoblotting with ubiquitin antibody. Monitor the disappearance of the polyubiquitin chain and the appearance of lower molecular weight fragments (e.g., di-ubiquitin) or free ubiquitin to determine cleavage activity and specificity [19] [29].
Signaling Pathways and Experimental Workflows
The following diagrams illustrate the regulatory roles of A20, CYLD, and USP19 in key immune pathways and a standard experimental workflow for DUB characterization.
Diagram Title: DUB Regulation of Innate Immune Signaling Pathways
Diagram Title: Functional Screening Workflow for DUBs
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for Studying DUBs and Ubiquitin Linkages
| Reagent Category | Specific Example | Function in Research |
|---|---|---|
| Linkage-Specific Ubiquitin Mutants | "K63-only" Ubiquitin (all lysines except K63 mutated to Arg) | To determine if a specific ubiquitin linkage is necessary and sufficient for a DUB's activity or a signaling event in cells [30]. |
| Activity-Based Probes (ABPs) | Ubiquitin-Propargylamide (Ub-PA) | To covalently label and identify active DUBs in cell lysates or to perform structural studies on DUB-probe complexes [29]. |
| Defined Polyubiquitin Chains | Purified K63-linked tetraubiquitin, K48-linked diubiquitin, etc. | The essential substrate for in vitro deubiquitination assays to directly characterize a DUB's linkage specificity and catalytic efficiency [19] [29]. |
| Linkage-Specific Antibodies | Anti-K63-linkage, Anti-K27-linkage antibodies | To immunoprecipitate or visualize specific chain types from cell lysates under endogenous conditions, allowing assessment of their dynamics [30]. |
| CRISPR/Cas9 Tools | sgRNA libraries targeting DUB families; stable Cas9 cell lines. | To perform systematic, loss-of-function screens to identify DUBs involved in specific pathways, such as antiviral immunity [24]. |
A20, CYLD, and USP19 are critical, non-redundant negative regulators of immune signaling, with their specificities for different ubiquitin chain linkages defining their unique biological roles. A20 and CYLD, both targeting K63-linked and M1-linked chains, are master regulators of NF-κB activation and cell death, with their loss leading to severe inflammatory pathology and cancer. USP19, with its activity towards K11-linked chains, plays a distinct role in cross-regulating autophagy and immune signaling. The emerging role of atypical chains like K27-linked ubiquitin adds a layer of complexity to the ubiquitin code in immunity, representing a fertile area for future research. Understanding the precise mechanisms and substrates of these DUBs continues to provide valuable insights into immune homeostasis and offers potential therapeutic avenues for treating inflammatory diseases and cancer.
Research Tools and Techniques for Probing K63 and K27 Ubiquitination in Immune Pathways
Protein ubiquitination is a crucial post-translational modification that regulates virtually all cellular processes, with particular importance in immune signaling pathways [31]. The versatility of ubiquitin signaling originates from the ability of this 76-amino acid protein to form polyubiquitin chains through eight distinct linkage types, connecting via one of seven internal lysine residues (K6, K11, K27, K29, K33, K48, K63) or the N-terminal methionine (M1) [32]. This combinatorial complexity creates a "ubiquitin code" that determines diverse functional outcomes for modified substrates, ranging from proteasomal degradation to activation of signaling cascades [33].
Within immune signaling, the K63 and K27 linkage types have emerged as particularly important regulators. K63-linked ubiquitin chains are well-established as key mediators of inflammatory and antiviral signaling pathways, serving as scaffolds for protein complex assembly in NF-κB activation, Toll-like receptor signaling, and T-cell receptor pathways [4]. More recently, K27-linked chains have been recognized as critical players in immune regulation, with specific E3 ligases like TRIM23 conjugating K27 chains to NEMO to activate both NF-κB and IRF3 pathways [33]. The ability to distinguish between these linkage types is therefore essential for understanding the molecular mechanisms controlling immune responses.
This guide compares two principal methodologies for linkage-specific detection of ubiquitin chains: the biochemical approach Ubiquitin Chain Restriction (UbiCRest) and various mass spectrometry-based techniques. We evaluate their respective capabilities, limitations, and applications in the context of immune signaling research, with particular focus on differentiating K63 versus K27-linked ubiquitin chains.
Methodology Comparison: UbiCRest versus Mass Spectrometry
Ubiquitin Chain Restriction (UbiCRest)
Experimental Principle and Workflow UbiCRest is a qualitative biochemical method that exploits the intrinsic linkage specificity of deubiquitinating enzymes (DUBs) to characterize ubiquitin chain architecture [31] [34]. The technique involves treating ubiquitinated substrates or purified polyubiquitin chains with a panel of linkage-specific DUBs in parallel reactions, followed by gel-based analysis (typically SDS-PAGE and immunoblotting) to observe cleavage patterns [31].
Table 1: Key DUBs for UbiCRest Analysis of K27 and K63 Linkages
| Linkage Type | Recommended DUB | Working Concentration | Specificity Notes |
|---|---|---|---|
| K63-linked | OTUD1 | 0.1-2 µM | Highly specific for K63 at low concentrations; may target other linkages at higher concentrations [31] |
| K27-linked | OTUD2 | 1-20 µM | Also cleaves K11, K29, K33; prefers longer K11 chains [31] |
| All linkages (positive control) | USP21 or USP2 | 1-5 µM (USP21) | Cleaves all eight linkage types including proximal ubiquitin [31] |
| K48-linked (negative control) | OTUB1 | 1-20 µM | Highly K48-specific; useful for demonstrating specificity [31] |
Protocol Implementation The standard UbiCRest protocol involves the following key steps [31]:
- Substrate Preparation: Immunoprecipitate the ubiquitinated protein of interest or purify polyubiquitin chains. As little as western blotting quantities of endogenously ubiquitinated proteins can be sufficient.
- DUB Panel Setup: Prepare parallel reactions with individual DUBs at their optimized working concentrations in appropriate reaction buffers.
- Incubation and Termination: Incubate reactions at 37°C for 1-3 hours, then terminate with SDS-PAGE loading buffer.
- Analysis: Separate proteins by SDS-PAGE and transfer to membranes for immunoblotting with ubiquitin-specific or substrate-specific antibodies.
The interpretation relies on comparing cleavage patterns across the DUB panel. For example, complete digestion of a ubiquitin smear by OTUD1 but not OTUD2 suggests the presence of K63- but not K27-linked chains.
Mass Spectrometry-Based Approaches
Experimental Principles and Workflows Mass spectrometry (MS) methods for ubiquitin chain analysis employ various enrichment and fragmentation techniques to identify linkage types and ubiquitination sites [32]. These can be broadly categorized into:
- Bottom-up Approaches: Proteins are trypsin-digested into peptides before LC-MS/MS analysis. Ubiquitination sites are identified through the characteristic 114.04 Da mass shift from the Gly-Gly remnant on modified lysine residues [32].
- Middle-down Approaches: Partial trypsin digestion under optimized native conditions results in cleavage at Arg74 of ubiquitin, allowing analysis of monoubiquitin remnants with preserved linkage information [31].
- Top-down Approaches: Recently developed methods analyze intact ubiquitinated proteins without proteolysis, enabling simultaneous determination of ubiquitination sites and chain architecture [35].
Table 2: Mass Spectrometry Method Variations for Ubiquitin Analysis
| Method Type | Enrichment Strategy | Key Readout | Linkage Information |
|---|---|---|---|
| Ubiquitin Tagging | His- or Strep-tagged ubiquitin expressed in cells [32] | Ubiquitination sites via GG-remnant (114.04 Da) | Limited to linkage types when combined with linkage-specific antibodies |
| Antibody-Based Enrichment | Anti-ubiquitin antibodies (P4D1, FK1/FK2) or linkage-specific antibodies [32] | Global ubiquitination profiling | Direct linkage typing with specific antibodies |
| UBD-Based Enrichment | Tandem ubiquitin-binding domains [32] | Interaction profiling | Can be linkage-specific depending on UBD preference |
| Top-down MS | Asp-N proteolysis preserves Ub chains [35] | Intact protein mass and fragmentation | Simultaneous site and chain architecture mapping |
Advanced MS Protocol for Linkage Determination The recently developed top-down methodology with computational integration (UbqTop platform) represents the cutting edge for comprehensive ubiquitin characterization [35]:
- Sample Preparation: Selective Asp-N proteolysis digests substrate proteins while preserving intact ubiquitin chains.
- LC-MS/MS Analysis: Intact proteins and ubiquitin chains are separated and fragmented.
- Computational Analysis: The UbqTop platform utilizes Bayesian-like scoring algorithms to predict ubiquitin chain topology from MS2 fragmentation data.
- Validation: Comparison with known standards and orthogonal methods confirms linkage assignments.
This approach has demonstrated utility in resolving isomeric chains and branched architectures that challenge conventional methods [35].
Performance Comparison in Immune Signaling Context
Technical Capabilities and Limitations
Table 3: Direct Comparison of UbiCRest and Mass Spectrometry for Ubiquitin Analysis
| Parameter | UbiCRest | Mass Spectrometry |
|---|---|---|
| Linkage Specificity | High for specific DUB-substrate pairs | Variable depending on method; excellent with advanced approaches |
| Sensitivity | Western blotting quantities (low microgram range) [31] | High sensitivity (potentially femtomole range) but requires significant enrichment [32] |
| Throughput | Medium (parallel DUB reactions) | Low to medium (sample preparation can be lengthy) |
| Architectural Insight | Can distinguish homotypic vs. heterotypic/branched chains [31] | Top-down methods can directly determine branching [35] |
| Quantitation Capability | Qualitative or semi-quantitative | Excellent with stable isotope labeling or label-free approaches |
| Implementation Time | Hours from sample to result [31] | Days including sample preparation and analysis |
| Cost | Low to moderate (commercial DUBs available) | High (instrumentation and specialized expertise) |
| Key Advantages | Rapid, accessible, no specialized equipment | Comprehensive, can identify novel linkages, high specificity |
| Major Limitations | Qualitative only, dependent on well-characterized DUBs | Complex data analysis, may miss low-abundance modifications |
Application to K63 vs K27 Chain Differentiation in Immune Signaling
The differentiation between K63 and K27-linked ubiquitin chains presents specific challenges and considerations for method selection. In immune signaling contexts:
K63-Linked Chains are well-established in RIG-I-like receptor signaling, STING activation, and T-cell receptor pathways [4]. They typically function as scaffolding elements that recruit signaling complexes through ubiquitin-binding domains. UbiCRest reliably identifies these chains using OTUD1 or AMSH [31], while MS approaches can provide additional information about chain length and interacting proteins.
K27-Linked Chains have emerged as important regulators in innate immunity, with TRIM family E3 ligases modifying substrates like NEMO to activate NF-κB and IRF3 pathways [33]. The characterization of K27 linkages is more challenging due to the relative lack of highly specific reagents. OTUD2 shows preference for K27 chains but also cleaves K11, K29, and K33 linkages [31], requiring careful interpretation of UbiCRest results. MS methods face challenges due to the lower abundance of K27 chains and technical difficulties in their enrichment and identification.
Experimental Design for Immune Signaling Studies
Integrated Approach for Comprehensive Analysis
For robust characterization of K63 and K27 chains in immune pathways, we recommend an integrated methodological approach:
- Initial Screening: Use UbiCRest as a first-line tool for rapid assessment of linkage types present in immunoprecipitated immune signaling proteins (e.g., NEMO, MAVS, STING).
- Targeted Validation: Apply linkage-specific antibodies where available to confirm UbiCRest findings.
- Comprehensive Mapping: Employ middle-down or top-down MS approaches for detailed architectural analysis, particularly when investigating potential branched chains.
- Functional Correlation: Combine linkage data with functional assays to establish physiological relevance.
This integrated strategy was successfully implemented in a recent study of MHC class II ubiquitination, which combined UbiCRest validation with MS-based linkage analysis to identify a previously unknown branched K11/K63 ubiquitin chain controlling antigen presentation [1].
The Scientist's Toolkit: Essential Research Reagents
Table 4: Key Reagents for Ubiquitin Linkage Analysis in Immune Signaling Research
| Reagent Category | Specific Examples | Application in K63/K27 Research |
|---|---|---|
| Linkage-Specific DUBs | OTUD1 (K63), OTUD2 (K27), Cezanne (K11), OTUB1 (K48) [31] | UbiCRest analysis of immune signaling complexes |
| Ubiquitin Variants | K63-only ubiquitin (all lysines except K63 mutated to arginine), K27-only ubiquitin | Control substrates for method validation |
| Linkage-Specific Antibodies | Anti-K63 linkage, anti-K27 linkage, anti-linear/M1 linkage [32] | Immunoblotting after UbiCRest; enrichment for MS |
| E3 Ligase Tools | TRIM23 (K27-specific), Ubc13-Uev1a (K63-specific E2 complex) [33] [4] | In vitro ubiquitination to generate specific chain types |
| Mass Spectrometry Standards | Synthetic ubiquitin chains, Heavy isotope-labeled ubiquitin | Quantitation standardization and method calibration |
| Computational Tools | UbqTop platform for top-down MS analysis [35] | Interpretation of complex ubiquitin chain architectures |
Both UbiCRest and mass spectrometry offer distinct advantages for the analysis of K63 and K27-linked ubiquitin chains in immune signaling pathways. UbiCRest provides a rapid, accessible biochemical approach that can be implemented in most molecular biology laboratories, making it ideal for initial screening and validation studies. Mass spectrometry approaches, particularly emerging top-down methodologies, offer unparalleled detail on chain architecture and can identify novel linkage types without prior assumptions.
The choice between these methods depends on specific research goals, available resources, and the particular biological questions being addressed. For most investigators studying K63 and K27 chains in immune signaling, a sequential approach that leverages the strengths of both methodologies will yield the most comprehensive and reliable insights into the complex ubiquitin code controlling immune responses.
Reporter Gene Assays (RGAs) are indispensable tools for investigating gene expression regulation and cellular signal transduction pathways. These assays utilize easily detectable reporter genes to provide a quantifiable readout of specific pathway activity, offering high accuracy, precision, and mechanism-relevance for studying immune signaling pathways [36]. In the context of innate immunity and stress responses, understanding the intricate regulation of pathways like NF-κB, IRF3, and MAPK is crucial, particularly as they are modulated by post-translational modifications including ubiquitination [37]. The assessment of K63-linked versus K27-linked ubiquitin chains has emerged as a significant regulatory mechanism in immune signaling, with K63-linked chains typically facilitating signal transduction and complex assembly, while K27-linked chains can serve distinct regulatory functions that vary by cellular context and target protein [37].
The design of RGAs enables researchers to create sensitive cellular systems that respond to pathway activation through measurable signals such as luminescence or fluorescence. When properly engineered, these assays can distinguish subtle differences in pathway activation under various experimental conditions, including those involving ubiquitination events. This technical guide provides a comprehensive comparison of reporter systems for three critical immune signaling pathways—NF-κB, IRF3, and MAPK—with particular attention to their application in studying ubiquitination-dependent regulation and their performance characteristics under diverse experimental conditions.
Reporter System Fundamentals and Design Considerations
Molecular Principles of Reporter Gene Assays
Reporter gene systems fundamentally consist of two key components: regulatory response elements that control gene expression and the reporter gene itself that produces a detectable signal [36]. The regulatory elements are typically DNA sequences corresponding to transcription factor binding sites or other cis-regulatory elements that respond to pathway activation. When a signaling pathway is activated, transcription factors translocate to the nucleus and bind these regulatory sequences, initiating transcription of the reporter gene. The resulting reporter protein accumulation can then be quantified using appropriate detection methods.
Common reporter genes include various forms of luciferase (Firefly, Nano luciferase, Gaussia) and fluorescent proteins (RFP, GFP variants) [36] [38]. Luciferase reporters generally offer higher sensitivity and broader dynamic range due to the enzymatic amplification of signal, while fluorescent proteins enable live-cell imaging and temporal tracking of pathway activity. More recent advances have focused on developing spectrally distinct reporters to enable multiplexed monitoring of multiple pathways simultaneously [39].
Critical Design Parameters for Optimal Performance
Selecting the appropriate reporter system requires careful consideration of several performance parameters. The table below summarizes key characteristics of common reporter types used in immune signaling research:
Table 1: Comparison of Reporter Gene Characteristics for Immune Signaling Studies
| Reporter Gene | Detection Method | Dynamic Range | Limit of Detection | Compatibility with Complex Fluids | Half-life | Spectral Properties |
|---|---|---|---|---|---|---|
| Firefly Luciferase | Luminescence | ~10²-10⁵ RLU | ~10⁻⁹-10⁻¹² M | Good | Stable (hours) | 560 nm peak |
| Nano Luciferase (stable) | Luminescence | ~10²-10⁶ RLU | ~10⁻¹² M | Good | Stable (hours) | 460 nm peak |
| Nano Luciferase (unstable, PEST) | Luminescence | ~10²-10⁶ RLU | ~10⁻¹² M | Good | Short (~1-2 h) | 460 nm peak |
| Gaussia Luciferase | Secreted luminescence | ~10²-10⁵ RLU | ~10⁻¹² M | Poor (high variability) | Stable | 480 nm peak |
| Red Fluorescent Protein (tdTomato) | Fluorescence | Moderate | ~10⁻⁸ M | Good | Stable (or destabilized) | 581 nm excitation/581 nm emission |
The dynamic range and limit of detection values demonstrate why luciferase-based reporters generally outperform fluorescent proteins for quantitative pathway activity assessment [38]. However, the optimal choice depends on specific experimental needs. For studies involving complex biological fluids like serum or synovial fluid, intracellular luciferases (Firefly and Nano luciferase) show superior performance due to reduced interference from fluid components, whereas secreted reporters like Gaussia luciferase exhibit significant inter-donor signal variability in these environments [38].
For kinetic studies or when measuring rapid changes in pathway activity, destabilized reporter variants with shortened half-lives (e.g., Nanoluc-PEST) provide superior temporal resolution by reducing signal accumulation from previous activation states [38]. The PEST degradation tag promotes rapid turnover of the reporter protein, enabling closer coupling between transcriptional activity and detectable signal.
Reporter Systems for NF-κB Pathway Analysis
NF-κB Signaling and Regulatory Mechanisms
The NF-κB pathway represents a critical signaling node in immune and inflammatory responses, responding to diverse stimuli including pathogen-associated molecular patterns, cytokines, and cellular stress. Pathway activation leads to translocation of NF-κB transcription factors to the nucleus, where they bind specific DNA sequences and initiate target gene expression. Ubiquitination plays a fundamental regulatory role in NF-κB activation, with K63-linked chains typically promoting activation through their function in IKK complex recruitment, while K48-linked chains primarily target proteins for proteasomal degradation.
NF-κB Reporter Assay Design and Performance
NF-κB reporter systems typically utilize synthetic promoters containing multiple NF-κB response elements (NF-κB-RE) driving expression of a reporter gene. A well-characterized design incorporates five NF-κB binding sites (GGGAATTTCC) in tandem repeats upstream of a minimal promoter and reporter gene [38]. This configuration provides high sensitivity and specific responsiveness to NF-κB pathway activation.
Experimental data demonstrate that NF-κB reporter assays exhibit excellent performance characteristics, with intra-batch coefficients of variation (CV) typically below 10% and inter-batch CV below 15% [36]. The dynamic range spans approximately 10²-10⁵ relative light units (RLU), enabling detection of subtle modulations in pathway activity. When using destabilized luciferase variants, the kinetic profile of NF-κB activation can be precisely tracked, with signal induction detectable within 1-2 hours of stimulation and peak responses occurring between 4-8 hours depending on the stimulus and cell type.
NF-κB Reporter Protocol
Key Reagents:
- NF-κB reporter plasmid (e.g., pNL1.1[NF-κB-RE] or pGL4.20[NF-κB-RE])
- Stimuli: TNF-α (10-100 ng/mL), IL-1β (10-50 ng/mL), or LPS (100 ng/mL-1 μg/mL)
- Transfection reagent (e.g., FuGENE6)
- Luciferase detection reagent compatible with reporter type
- Reference plasmid (e.g., pcDNA4/TO/LacZ for normalization) [38]
Experimental Workflow:
- Cell Seeding and Transfection: Seed appropriate cell type (e.g., SW1353, HeLa, or HEK293) at 60,000 cells/cm² in 96-well plates. After adherence, transfect with NF-κB reporter plasmid using Fugene6 transfection reagent according to manufacturer's instructions. Co-transfect with reference plasmid (10% of total DNA) for normalization [38].
- Starvation and Stimulation: 24 hours post-transfection, starve cells in serum-free medium for 16 hours to reduce basal activity. Stimulate with appropriate agonists or test compounds in fresh medium.
- Signal Detection: At experimental endpoint (typically 6-8 hours post-stimulation), lyse cells and measure luciferase activity using compatible detection reagent. For secreted luciferase, collect conditioned medium for measurement.
- Data Analysis: Normalize reporter activity to reference signal. Calculate fold induction relative to unstimulated controls. Perform dose-response analysis using 4-parameter logistic curve fitting for IC₅₀/EC₅₀ determination.
Diagram 1: NF-κB Reporter System Workflow. This diagram illustrates the signaling pathway from receptor activation to measurable reporter output, highlighting key regulatory steps including ubiquitination-dependent IκB degradation.
Reporter Systems for IRF3 Pathway Analysis
IRF3 Signaling in Antiviral Immunity
Interferon regulatory factor 3 (IRF3) serves as the master transcription factor initiating type I interferon expression in response to viral infection [40]. IRF3 is present constitutively in the cytoplasm of most cell types and undergoes rapid activation through phosphorylation upon detection of viral components by pattern recognition receptors including RIG-I-like receptors (RLRs) and cGAS [41]. Activated IRF3 forms dimers that translocate to the nucleus and bind to interferon-sensitive response elements (ISRE) in promoter regions of target genes, particularly IFN-β.
The cGAS-STING pathway represents a major IRF3-activating pathway that is extensively regulated by ubiquitination [37]. K63-linked ubiquitination of STING by TRIM56 and other E3 ligases promotes its dimerization, Golgi accumulation, and recruitment of TBK1, which phosphorylates IRF3 [37]. Conversely, K48-linked ubiquitination by RNF5 and TRIM29 targets STING for proteasomal degradation, limiting signaling duration. K27-linked ubiquitination by TRIM10 and the AMFR/INSIG1 complex also facilitates STING trafficking and activation, demonstrating the complex interplay between ubiquitin linkage types in regulating IRF3 activity [37].
IRF3 Reporter Implementation and Applications
IRF3 reporter systems typically utilize either the native IFN-β promoter or synthetic promoters containing multiple IRF3 binding sites. The core IFN-β promoter region spanning nucleotides -126 to +4 relative to the transcription start site contains essential IRF3 binding elements and provides specific responsiveness to IRF3 activation [41]. For enhanced sensitivity, synthetic promoters with multimerized IRF response elements (IRF-RE) are also employed.
Research has identified regulatory proteins that modulate IRF3 activity, including TOB1, which recruits HDAC8 to the IFN-β promoter region to suppress IRF3-directed transcription [41]. This regulatory mechanism represents a feedback inhibitor of host antiviral response and can be effectively studied using IRF3 reporter systems. The kinetic profile of IRF3 activation is typically rapid, with detectable reporter induction within 4-6 hours of viral infection or STING agonist stimulation.
IRF3 Reporter Protocol
Key Reagents:
- IRF3 reporter plasmid (IFN-β promoter or multimerized IRF-RE)
- Stimuli: Sendai virus (SeV, 50-100 HAU/mL), vesicular stomatitis virus (VSV, MOI 1-5), HSV-1 (MOI 1-5), or STING agonists (cGAMP 1-10 μg/mL)
- Transfection reagents
- Luciferase detection system
- Reference plasmid for normalization [41]
Experimental Workflow:
- Cell Preparation and Transfection: Seed appropriate cell type (primary macrophages, HEK293, or specialized reporter cells) in 96-well plates. Transfect with IRF3 reporter construct using suitable transfection method. Include reference plasmid for normalization.
- Stimulation and Incubation: 24 hours post-transfection, stimulate cells with viral particles or pathway-specific agonists. Incubate for appropriate duration (typically 8-16 hours) to allow pathway activation and reporter accumulation.
- Pathway Modulation: To study ubiquitination effects, co-transfect with plasmids expressing wild-type or mutant ubiquitin, E3 ligases (TRIM56, RNF5), or deubiquitinating enzymes (USP21). Alternatively, pretreat with proteasome inhibitors (MG132) or lysosome inhibitors (chloroquine) to examine degradation pathways.
- Signal Detection and Analysis: Measure reporter activity using compatible detection system. Normalize to reference signal. For studies involving viral infection, include appropriate biosafety containment.
Diagram 2: IRF3 Reporter System Workflow. The visualization shows parallel pathway activation through cGAS-STING and RIG-I-like receptors, converging on TBK1-mediated IRF3 phosphorylation and subsequent reporter gene activation, with highlighted ubiquitination regulatory nodes.
Reporter Systems for MAPK Pathway Analysis
MAPK/ERK Signaling Dynamics
The Mitogen-Activated Protein Kinase (MAPK) pathway, particularly the ERK1/2 cascade, regulates fundamental cellular processes including proliferation, differentiation, and survival. ERK serves as a central node in signaling networks, with its temporal activity patterns determining specific cell fate decisions [39]. In immune contexts, MAPK signaling contributes to lymphocyte activation, cytokine production, and inflammatory responses.
MAPK pathway activity demonstrates complex regulation, with recent studies revealing non-monotonic (biphasic) responses to increasing pathway activation. In fibroblast-to-motor neuron conversion models, optimal "Goldilocks" levels of MAPK signaling efficiently drive cell-fate programming, while both insufficient and excessive activation impair the process [42]. This nuanced relationship underscores the importance of quantitative reporter systems that can accurately capture pathway dynamics.
Advanced MAPK Biosensors and Applications
Traditional MAPK reporters have relied on transcriptional reporters with serum response elements (SRE) or other MAPK-responsive promoters. However, recent advances have focused on Förster Resonance Energy Transfer (FRET)-based biosensors that report direct kinase activity rather than transcriptional output. The EKAR (ERK Activity Reporter) series represents optimized FRET biosensors that detect ERK-mediated phosphorylation through conformational changes that alter FRET efficiency between cyan and yellow fluorescent protein variants [39].
Newer developments have addressed spectral limitations of traditional biosensors. The recently developed REKAR67 and REKAR76 biosensors utilize red-shifted FRET pairs (miRFP670nano3 and miRFP720) operating in the 670-720 nm range, enabling multiplexing with other CFP/YFP-based biosensors [39]. These red-FRET ERK biosensors show high consistency with existing CFP/YFP biosensors while offering spectral compatibility for simultaneous monitoring of multiple signaling pathways.
MAPK Reporter Protocol
Key Reagents:
- MAPK transcriptional reporter (SRE-driven) or FRET biosensor (EKAR variants)
- Stimuli: EGF (10-100 ng/mL), PMA (100 nM), or serum (10-20%)
- Inhibitors: MEK inhibitors (PD0325901, trametinib), EGFR inhibitors (gefitinib)
- Transfection or transduction reagents
- Appropriate detection system (luminescence plate reader or fluorescence microscope with FRET capabilities) [39]
Experimental Workflow:
- Cell Engineering: Stably express MAPK reporter in desired cell line using lentiviral transduction or stable transfection. For FRET biosensors, select clonal populations with consistent expression.
- Starvation and Baseline Measurement: Serum-starve cells for 4-16 hours to reduce basal MAPK activity. Measure baseline reporter activity.
- Stimulation and Kinetic Monitoring: Stimulate with growth factors or other agonists. For transcriptional reporters, measure endpoint signal after 4-6 hours. For FRET biosensors, perform continuous or time-lapse monitoring to capture dynamic activity patterns.
- Inhibition Studies: Pre-treat with pathway inhibitors (MEKi, BRAFi) for 1-2 hours before stimulation to confirm pathway specificity.
- Data Analysis: For FRET biosensors, calculate emission ratio (acceptor/donor) and normalize to baseline. For transcriptional reporters, normalize to reference signal and calculate fold induction.
Table 2: MAPK Pathway Activity Assessment Methods
| Method Type | Principle | Temporal Resolution | Information Content | Applications |
|---|---|---|---|---|
| Transcriptional Reporters | MAPK-responsive promoter driving luciferase | Hours (transcription-dependent) | Bulk population, endpoint | Drug screening, pathway modulation studies |
| FRET Biosensors | Conformational change after ERK phosphorylation | Minutes to hours (real-time) | Single-cell, spatiotemporal dynamics | Live-cell imaging, signaling heterogeneity |
| MAPKi Sensitivity Scores | Gene expression signature from RNA sequencing | N/A (static snapshot) | Predictive of drug response | Patient stratification, precision oncology |
| Phospho-antibody Detection | Immunodetection of phosphorylated ERK | Hours (fixed time points) | Bulk population, snapshot | Clinical pathology, endpoint assessment |
Diagram 3: MAPK Reporter System Workflow. The diagram illustrates parallel detection methods for MAPK pathway activity, including direct kinase activity monitoring via FRET biosensors and transcriptional output measurement through SRE-driven reporters.
Comparative Performance Analysis and Technical Considerations
Direct Performance Comparison Across Pathway Reporters
The quantitative performance of reporter systems varies significantly based on reporter design, detection method, and pathway characteristics. The following table summarizes key performance metrics for the three pathway reporter systems:
Table 3: Performance Comparison of NF-κB, IRF3, and MAPK Reporter Systems
| Performance Parameter | NF-κB Reporter | IRF3 Reporter | MAPK Transcriptional Reporter | MAPK FRET Biosensor |
|---|---|---|---|---|
| Time to Peak Signal | 4-8 hours | 8-16 hours | 4-6 hours | 5-30 minutes |
| Fold Induction Range | 10-100x | 5-50x | 5-30x | 1.5-3x (ratio change) |
| Limit of Detection | ~10⁻¹² M | ~10⁻¹² M | ~10⁻¹² M | N/A (single-cell) |
| Intra-batch CV | <10% | <10% | <10% | 5-15% (cell-to-cell) |
| Inter-batch CV | <15% | <15% | <15% | 10-20% |
| Compatibility with Complex Fluids | Good | Good | Good | Excellent |
| Temporal Resolution | Moderate | Slow | Moderate | Excellent |
| Spatial Resolution | None (bulk) | None (bulk) | None (bulk) | Subcellular |
The data reveal that while luciferase-based transcriptional reporters for all three pathways show similar sensitivity (limit of detection ~10⁻¹² M), they differ significantly in their temporal profiles. NF-κB reporters typically show the most rapid induction, making them suitable for acute response studies, while IRF3 reporters require longer incubation periods due to the indirect nature of interferon pathway activation. MAPK FRET biosensors provide vastly superior temporal resolution but lower overall signal dynamic range compared to transcriptional amplification-based systems.
Technical Considerations for Experimental Design
Several critical factors influence reporter system performance and data interpretation. First, promoter selection significantly impacts specificity and background. Minimal synthetic promoters with multimerized response elements generally provide higher induction ratios and lower baseline activity compared to natural promoters, which may contain additional regulatory elements that confound specific pathway assessment [38].
Second, reporter stability affects kinetic resolution. Stable reporters (e.g., standard luciferase) accumulate over time, providing robust endpoint measurements but blurring temporal resolution. Conversely, destabilized reporters (e.g., Nanoluc-PEST) with shortened half-lives more accurately reflect real-time pathway activity but may yield lower overall signals [38].
Third, the cellular context profoundly influences reporter performance. Endogenous pathway components, feedback regulators, and cross-talk with other signaling networks all vary by cell type. Primary cells typically show more physiological responses but lower transfection efficiency, while engineered cell lines offer consistency and ease of use but may lack relevant regulatory mechanisms.
The Scientist's Toolkit: Essential Research Reagents
Successful implementation of pathway reporter assays requires access to specialized reagents and tools. The following table catalogues essential research solutions for studying immune signaling pathways:
Table 4: Essential Research Reagents for Pathway Reporter Studies
| Reagent Category | Specific Examples | Function/Application | Key Characteristics |
|---|---|---|---|
| Reporter Plasmids | pNL1.1[NF-κB-RE], pGL4.20[NF-κB-RE], IFN-β promoter constructs, SRE-luciferase, EKAR biosensors | Pathway-specific activity reporting | Varying dynamic range, specificity, and temporal resolution |
| Stimuli/Agonists | TNF-α, IL-1β, LPS (NF-κB); Sendai virus, cGAMP, HSV-1 (IRF3); EGF, PMA, serum (MAPK) | Pathway-specific activation | Different potency, mechanism, and kinetics |
| Inhibitors | IKK inhibitors (IKK-16), TBK1 inhibitors (BX795), MEK inhibitors (PD0325901, trametinib) | Pathway inhibition for specificity confirmation | Varying selectivity, potency, and mechanism |
| Detection Reagents | Luciferase assay substrates, FuGENE6 transfection reagent, normalization controls | Signal detection and experimental normalization | Different sensitivity, compatibility, and dynamic range |
| Ubiquitination Tools | Wild-type and mutant ubiquitin plasmids (K63-only, K48-only, K27-only), E3 ligase expression constructs (TRIM56, RNF5), deubiquitinase expression plasmids | Manipulation and study of ubiquitination | Linkage-specific effects on pathway components |
Reporter systems for NF-κB, IRF3, and MAPK pathway activation provide powerful, sensitive tools for dissecting immune signaling mechanisms and their regulation by ubiquitination. The continuing development of increasingly sophisticated reporters—including multicolor FRET biosensors, destabilized luciferase variants, and pathway-specific signatures—is expanding our capacity to interrogate these critical signaling networks with greater precision, temporal resolution, and physiological relevance.
The integration of pathway reporter data with ubiquitination studies continues to reveal the nuanced regulation of immune signaling by different ubiquitin linkage types. As our understanding of K63 versus K27-linked chain functions evolves, and as additional ubiquitin-like modifications are characterized, reporter systems will remain essential tools for elucidating the complex biochemical networks that control immune responses. Future directions will likely include the development of more specific ubiquitination reporters, enhanced multiplexing capabilities for parallel pathway monitoring, and specialized systems for high-content screening in drug discovery applications.
The post-translational modification of proteins by ubiquitin is a fundamental mechanism for controlling immune signaling pathways. Among the diverse types of polyubiquitin chains, K63-linked ubiquitination has emerged as a critical non-proteolytic signal that regulates innate immune activation, while K27-linked chains are increasingly recognized for their specialized roles in inflammatory responses [4] [43]. Within this complex regulatory network, deubiquitinating enzymes (DUBs) perform the essential function of reversing ubiquitination events, thereby fine-tuning signal transduction. The ubiquitin-specific protease USP19 represents a multifaceted regulator that operates at the intersection of autophagy and antiviral immunity, with genetically modified models providing crucial insights into its physiological functions [44] [45]. This guide objectively compares experimental approaches and findings from key studies utilizing CRISPR/Cas9 knockouts and in vivo models of USP19 deficiency, placing these findings within the broader context of K63 versus K27 ubiquitin signaling research.
Table 1: Key Characteristics of Ubiquitin Linkages in Immune Signaling
| Ubiquitin Linkage | Primary Immune Functions | Representative E3 Ligases | Representative DUBs |
|---|---|---|---|
| K63-linked | NF-κB activation, TAK1 regulation, antiviral signaling [4] | TRAF6, cIAPs [4] | A20, CYLD [4] |
| K27-linked | T cell signaling, cytokine responses [43] | HOIP, HOIL-1 [43] | Unknown |
| K11-linked | Autophagy regulation, ER-associated degradation [44] [7] | APC/C, UBE2S [7] | USP19 [44] |
Methodological Comparison: From Cellular Models to In Vivo Validation
CRISPR/Cas9 Knockout Models and Cell-Based Assays
Studies investigating USP19 function have employed CRISPR/Cas9 technology to generate knockout cell lines, enabling precise characterization of USP19 mechanisms in controlled experimental systems.
Table 2: Key Methodological Approaches in USP19 Research
| Method Category | Specific Techniques | Application in USP19 Studies |
|---|---|---|
| Gene Knockdown | siRNA, shRNA | Initial identification of USP19 in autophagy screens [44] |
| CRISPR/Cas9 | Knockout cell lines | Validation of Beclin-1 and TBK1 mechanisms [44] [45] |
| Protein Interaction | Co-immunoprecipitation, in vitro binding | Identification of USP19 interactors [44] [45] |
| Ubiquitination Status | Ubiquitin assays with mutants (K63R, K48R) | Determination of chain linkage specificity [44] |
A seminal study published in 2016 utilized a systematic siRNA screen targeting 84 human DUBs, identifying USP19 as a positive regulator of autophagy [44]. Researchers confirmed these findings using siRNA-mediated knockdown in 293T and HeLa cells, observing reduced GFP-LC3B puncta formation under both basal conditions and nutrient deprivation induced by Earle's balanced salt solution (EBSS) treatment. To establish USP19's molecular mechanism, the study employed co-immunoprecipitation experiments demonstrating that USP19 interacts with core components of the Beclin-1 complex, including VPS15, VPS34, ATG14L, UVRAG, and AMBRA1. Critically, the catalytic activity of USP19 was shown to be essential for its function, as catalytically inactive mutants (C506S and C506S/H1157A) failed to promote autophagy.
A subsequent 2021 investigation focused on USP19's role in antiviral immunity through regulation of TBK1 stability [45]. This research employed a more targeted genetic approach, testing 43 DUBs for their ability to regulate TBK1 and identifying USP19 as a key regulator. The experimental protocol involved co-transfecting HEK293T cells with TBK1 and DUB expression plasmids, followed by immunoblot analysis to assess TBK1 protein levels. To determine the mechanism of degradation, researchers used specific inhibitors: MG132 (proteasome inhibitor), chloroquine (lysosomal inhibitor), 3-methyladenine (autophagy inhibitor), and bafilomycin A1 (autophagosome-lysosome fusion inhibitor). The finding that only chloroquine prevented TBK1 degradation indicated a lysosome-dependent pathway. Domain mapping experiments further revealed that the USP domain of USP19 interacts with the kinase domain of TBK1, facilitating its degradation through chaperone-mediated autophagy (CMA).
Figure 1: USP19 Promotes TBK1 Degradation via Chaperone-Mediated Autophagy
In Vivo Studies in USP19-Deficient Mice
The transition from cellular models to whole-organism physiology represents a critical step in validating biological mechanisms. Research on USP19 has advanced to include macrophage-specific usp19 knockout mice, providing compelling evidence for its role in antiviral immunity in vivo [45]. The experimental approach involved challenging these genetically modified mice with vesicular stomatitis virus (VSV), a well-established model for studying type I interferon responses. In these studies, USP19 deficiency in macrophages resulted in significantly attenuated VSV replication and enhanced resistance to viral infection compared to wild-type controls. This protective effect correlated with elevated TBK1 protein levels and enhanced activation of the type I interferon signaling pathway in USP19-deficient macrophages following viral challenge. These in vivo findings strongly support the physiological relevance of the cellular mechanisms identified through CRISPR/Cas9 and other cell-based approaches, demonstrating that USP19-mediated regulation of TBK1 stability has meaningful consequences for host defense against viral pathogens.
Comparative Analysis of USP19 Functions in Immune Regulation
USP19 in Autophagy Regulation
The 2016 study by Liang et al. established that USP19 deubiquitinates Beclin-1, specifically removing K11-linked ubiquitin chains from lysine 437 [44]. This action stabilizes Beclin-1, a core component of the autophagy initiation machinery, thereby promoting autophagic flux. The experimental evidence demonstrated that USP19 depletion inhibits the accumulation of LC3-II (a standard autophagy marker) and increases levels of the autophagy substrate p62/SQSTM1 under both basal conditions and upon induction by rapamycin or EBSS treatment. Interestingly, this function of USP19 in autophagy regulation intersects with antiviral immunity, as the study further showed that the USP19-Beclin-1 axis negatively regulates type I interferon signaling by blocking the interaction between RIG-I and MAVS, thereby dampening cellular antiviral responses.
USP19 in TBK1 Regulation and Antiviral Immunity
The 2021 study by He et al. revealed a distinct mechanism by which USP19 promotes TBK1 degradation through chaperone-mediated autophagy (CMA) [45]. This process requires recognition of a canonical CMA motif (KFDKQ) in TBK1 by the chaperone protein HSPA8/HSC70, followed by translocation into lysosomes via LAMP2A. Unlike the deubiquitination activity required for Beclin-1 regulation, USP19's effect on TBK1 degradation was independent of its catalytic activity, instead relying on protein-protein interactions. Functionally, USP19 deficiency enhanced TBK1 stability, IRF3 activation, and IFNB production following viral infection, establishing USP19 as a negative regulator of the antiviral immune response.
Table 3: Comparative Functions of USP19 in Immune Regulation
| Experimental Parameter | Autophagy Regulation [44] | TBK1 Regulation [45] |
|---|---|---|
| Molecular Target | Beclin-1 | TBK1 |
| Ubiquitin Linkage | K11-linked | Not ubiquitin-dependent |
| Catalytic Requirement | Deubiquitinase activity essential | Deubiquitinase activity independent |
| Cellular Process | Autophagy induction | Chaperone-mediated autophagy |
| Immune Pathway | Negative regulation of RIG-I-MAVS | Negative regulation of TBK1-IRF3 |
| Genetic Evidence | siRNA knockdown, BECN1 KO cells | CRISPR/Cas9, CMA component knockdown |
| In Vivo Validation | Not provided | Macrophage-specific KO mice |
The Scientist's Toolkit: Essential Research Reagents
Table 4: Key Research Reagents for USP19 Studies
| Reagent/Cell Line | Specific Application | Experimental Function |
|---|---|---|
| USP19 siRNA/shRNA | Gene knockdown | Initial functional screening and validation [44] |
| CRISPR/Cas9 KO cells | Genetic knockout | Mechanism validation in controlled systems [44] [45] |
| Catalytic mutants (C506S, C607S) | Structure-function studies | Determination of deubiquitinase activity requirement [44] [45] |
| Chloroquine | Lysosomal inhibition | Differentiation of lysosomal vs. proteasomal degradation [45] |
| Anti-Beclin-1 antibody | Immunoprecipitation | Identification of USP19 interaction partners [44] |
| Anti-TBK1 antibody | Immunoblotting, IP | Assessment of TBK1 stability and complex formation [45] |
| USP19-deficient mice | In vivo validation | Physiological relevance of cellular findings [45] |
Figure 2: USP19 in the Context of K63 and K27 Ubiquitin Signaling
The genetic models of USP19 deficiency, spanning from CRISPR/Cas9 knockout cell lines to tissue-specific mouse models, reveal a complex picture of this DUB as a multifaceted regulator of immune homeostasis. USP19 emerges as a node interconnecting autophagy and antiviral signaling through distinct mechanisms: one involving deubiquitination of Beclin-1 (K11-linkage specific) and another facilitating TBK1 degradation via chaperone-mediated autophagy (ubiquitin-independent). When positioned within the broader framework of K63 versus K27 ubiquitin signaling, USP19 appears to function predominantly through alternative mechanisms rather than directly editing these specific chain types. The in vivo evidence demonstrating that USP19 deficiency enhances antiviral resistance provides a compelling rationale for considering USP19 as a potential therapeutic target for modulating immune responses, particularly in the context of viral infections and potentially autoimmune conditions where fine-tuning of type I interferon responses may be beneficial.
Ubiquitination is a crucial post-translational modification that regulates immune signaling pathways by modulating protein function, localization, and stability. Among the diverse ubiquitin chain linkages, K63-linked and K27-linked polyubiquitin chains serve distinct non-degradative roles in orchestrating immune responses [46] [47]. K63-linked ubiquitination is well-established as a key regulator of innate immune signaling, functioning in protein-protein interactions and complex assembly [4]. In contrast, K27-linked chains are less characterized but have been implicated in specific immune processes including mitochondrial autophagy and inflammatory signaling [48]. This guide provides a comparative analysis of physiological substrates—TRIF, RIPK1, STING, and T cell signaling proteins—focusing on their regulation by K63 versus K27 ubiquitin linkages, with supporting experimental data and methodological details for researchers and drug development professionals.
Comparative Functions of K63 and K27 Ubiquitin Linkages
Table 1: Functional Comparison of K63 and K27 Ubiquitin Linkages
| Feature | K63-Linked Ubiquitination | K27-Linked Ubiquitination |
|---|---|---|
| Primary Functions | Non-degradative signaling, protein complex assembly, inflammatory pathways, DNA damage repair [4] | Mitophagy, inflammatory regulation, role in innate immunity [48] |
| Key Signaling Pathways | NF-κB activation, RLR signaling, TLR signaling, TNFR1 signaling [46] [4] | Less defined; implicated in NLRP3 inflammasome regulation, mitophagy [49] [48] |
| Chain Recognition | Recognized by specific ubiquitin-binding domains (e.g., NEMO/IKKγ, ABIN proteins) [46] | Recognition mechanisms less characterized; potential role in recruiting autophagy adapters [48] |
| Experimental Detection | Linkage-specific antibodies, Ubc13 knockout/mutation, ubiquitin binding domain probes [4] | Methodological limitations; linkage-specific tools under development |
Physiological Substrates and Their Ubiquitin Linkage Profiles
TRIF (TIR-Domain-Containing Adapter-Inducing Interferon-β)
TRIF is a crucial adaptor protein in Toll-like receptor signaling that activates both NF-κB and IRF3 pathways to induce type I interferon responses [47]. TRIF-mediated signaling is predominantly regulated through K63-linked ubiquitination, which facilitates downstream protein interactions and pathway activation.
Table 2: TRIF Ubiquitination Profile and Functional Consequences
| Aspect | Experimental Findings |
|---|---|
| Ubiquitin Linkage | Primarily K63-linked polyubiquitination [47] |
| Functional Role | Enables recruitment of downstream signaling complexes including TRAF6 and TBK1 [47] |
| Regulatory E3 Ligases | TRAF6 identified as primary E3 ligase [47] |
| Deubiquitinating Enzymes | Multiple DUBs implicated in negative regulation (e.g., A20, CYLD) [47] |
| Experimental Evidence | Co-immunoprecipitation with linkage-specific ubiquitin antibodies; TRAF6 knockout studies; mass spectrometry analysis |
Experimental Protocol for TRIF Ubiquitination Studies:
- Immunoprecipitation: Transfect HEK293T cells with FLAG-tagged TRIF and HA-tagged ubiquitin (wild-type or K63-only mutant)
- Stimulation: Treat cells with TLR3 agonist (poly(I:C)) for 0, 15, 30, 60 minutes
- Cell Lysis: Use RIPA buffer supplemented with protease inhibitors and deubiquitinase inhibitors (N-ethylmaleimide)
- Detection: Immunoprecipitate TRIF with FLAG antibody, then immunoblot with HA antibody to detect ubiquitination
- Linkage Specificity: Confirm K63-linkage using K63-linkage specific ubiquitin antibodies and ubiquitin mutants (K63R)
RIPK1 (Receptor-Interacting Protein Kinase 1)
RIPK1 serves as a critical signaling node in cell death and inflammation pathways, exhibiting a dual role in promoting either cell survival or death depending on its post-translational modifications [50] [51]. RIPK1 undergoes complex ubiquitination with multiple linkage types including K63, K11, K48, and linear/M1 chains, with K63-linked ubiquitination playing a particularly important role in pro-survival signaling.
Table 3: RIPK1 Ubiquitination Profile and Functional Consequences
| Aspect | Experimental Findings |
|---|---|
| Ubiquitin Linkages | K63, K11, K48, M1-linear chains; context-dependent [50] |
| K63-Linked Function | Promotes pro-survival NF-κB signaling by recruiting NEMO/IKK complex [50] |
| K27 Linkage Evidence | Limited direct evidence; potential role in cell death regulation |
| Regulatory E3 Ligases | cIAP1/2, LUBAC, TRAF2 [50] [52] |
| Deubiquitinating Enzymes | CYLD, A20 (negative regulators) [50] [51] |
| Experimental Evidence | Ubiquitin linkage mapping; cIAP1 UBA domain studies; kinase activity assays |
Experimental Protocol for RIPK1 Ubiquitination Studies:
- Complex Isolation: Stimulate cells with TNFα (20 ng/mL) for 0, 5, 15, 30 minutes to activate TNFR1 complex I formation
- Immunoprecipitation: Isclude endogenous RIPK1 using specific antibodies
- Denaturing Conditions: Use 1% SDS in lysis buffer to isolate covalently attached ubiquitin chains
- Mass Spectrometry: For comprehensive ubiquitin mapping, digest RIPK1 with trypsin and analyze with LC-MS/MS to identify ubiquitination sites and linkage types
- Functional Validation: Combine with cIAP1/2 inhibition (SMAC mimetics) or DUB inhibition to manipulate ubiquitination status
STING (Stimulator of Interferon Genes)
STING is a central adaptor protein in the cytosolic DNA sensing pathway that activates type I interferon responses against viral and bacterial infections. STING activation is primarily regulated through K63-linked ubiquitination, which is essential for its trafficking and ability to activate downstream TBK1 and IRF3.
Table 4: STING Ubiquitination Profile and Functional Consequences
| Aspect | Experimental Findings |
|---|---|
| Ubiquitin Linkage | Predominantly K63-linked chains [4] [53] |
| Functional Role | Facilitates STING trafficking from ER to Golgi and recruitment of TBK1 [53] |
| K27 Linkage Evidence | Not well established for STING |
| Regulatory E3 Ligases | TRIM56, TRIM32, AMFR [53] |
| Deubiquitinating Enzymes | Multiple DUBs regulate STING (e.g., USP13, USP20) [53] |
| Experimental Evidence | Immunofluorescence co-localization studies; TBK1 recruitment assays; IFN-β reporter assays |
Experimental Protocol for STING Ubiquitination Studies:
- Cell Stimulation: Treat macrophages or HEK293T cells with STING agonists (2'3'-cGAMP, 5-10 μg/mL) or DNA transfection
- Time Course Analysis: Collect samples at 0, 2, 4, 6, 8 hours post-stimulation
- Subcellular Fractionation: Separate membrane fractions to monitor STING trafficking
- Immunoprecipitation: Pull down STING under denaturing conditions to preserve ubiquitination
- Downstream Signaling: Assess TBK1 phosphorylation and IRF3 dimerization to correlate ubiquitination with functional output
T Cell Signaling Proteins
T cell receptor (TCR) signaling involves multiple proteins regulated by ubiquitination, with K63-linked chains playing well-established roles in activating NF-κB and MAPK pathways. The role of K27-linked ubiquitination in T cell signaling remains largely unexplored.
Table 5: T Cell Signaling Protein Ubiquitination Profiles
| Protein | Ubiquitin Linkage | Functional Consequences |
|---|---|---|
| TCR Complex | K63-linked chains [4] | Regulates TCR signaling strength and duration |
| CARMA1 | K63-linked chains [4] | Facilitates formation of CBM complex for NF-κB activation |
| MALT1 | K63-linked chains [4] | Recruits and activates IKK complex through NEMO binding |
| BCL10 | K63 and K48 mixed chains [4] | Modulates CBM complex stability and signaling output |
| PKCθ | Potential K63 linkage | Less characterized ubiquitination status |
Experimental Protocol for T Cell Signaling Ubiquitination Studies:
- T Cell Activation: Use Jurkat T cells or primary human T cells stimulated with anti-CD3/anti-CD28 antibodies (1-10 μg/mL)
- Kinetic Analysis: Collect samples at 0, 5, 15, 30, 60 minutes post-stimulation
- Immunoprecipitation: Isolate specific T cell signaling proteins (CARMA1, BCL10, MALT1)
- Ubiquitin Detection: Use linkage-specific ubiquitin antibodies to determine chain topology
- Functional Assays: Correlate ubiquitination status with IL-2 production and NF-κB reporter activity
Research Reagent Solutions
Table 6: Essential Research Reagents for K63 and K27 Ubiquitination Studies
| Reagent Category | Specific Examples | Research Application |
|---|---|---|
| Linkage-Specific Antibodies | Anti-K63-linkage specific, Anti-K27-linkage specific (available but less validated) | Detect specific ubiquitin chain types in immunoblotting and immunofluorescence |
| Ubiquitin Mutants | Ubiquitin K63R, K27R, K63-only (all lysines except K63 mutated to arginine) | Determine linkage specificity in overexpression systems |
| E3 Ligase Inhibitors | SMAC mimetics (cIAP1/2), LUBAC inhibitor (HOIPIN-8) | Manipulate ubiquitination of specific substrates |
| DUB Inhibitors | PR-619 (pan-DUB inhibitor), specific USP inhibitors | Stabilize ubiquitinated proteins for detection |
| Activity Reporters | NF-κB luciferase, IFN-β luciferase, AP-1 reporter | Correlate ubiquitination with functional output |
| Proteasome Inhibitors | MG132, bortezomib | Distinguish between degradative and non-degradative ubiquitination |
Signaling Pathway Diagrams
The comparative analysis of TRIF, RIPK1, STING, and T cell signaling proteins reveals that K63-linked ubiquitination serves as a predominant regulatory mechanism across multiple immune signaling pathways, while evidence for K27-linked ubiquitination in these specific substrates remains limited. The well-established roles of K63 chains in facilitating protein-protein interactions and complex assembly contrast with the emerging understanding of K27 chains in immune regulation. This comparison highlights significant knowledge gaps and research opportunities, particularly in characterizing K27 ubiquitination and developing specific research tools. For drug discovery professionals, these distinctions offer potential pathways for therapeutic intervention, with K63-linked ubiquitination presenting more validated targets while K27 linkages represent emerging opportunities for future investigation.
Ubiquitination, the process by which ubiquitin proteins are attached to substrate proteins, is a critical post-translational modification that regulates nearly every aspect of cellular function. The specific linkages between ubiquitin molecules form distinct chains that function as molecular codes, directing diverse functional outcomes in immune signaling pathways [8] [7]. Among these, K63-linked ubiquitin chains have been extensively characterized as key regulators of inflammatory signaling, immune cell activation, and antiviral responses, primarily through non-proteolytic mechanisms that modulate protein activity, interactions, and subcellular localization [4] [27]. In contrast, K27-linked ubiquitin chains represent a more recently discovered and less understood linkage type that has emerged as a crucial regulator in innate antiviral immunity, particularly in the cGAS-STING DNA sensing pathway [54].
This comparison guide provides an objective assessment of the methodologies and technologies enabling functional characterization of these distinct ubiquitin linkages, with a focus on their roles in cytokine production, immune cell differentiation, and antiviral response. We present structured experimental data, detailed protocols, and pathway visualizations to equip researchers with the tools needed to decipher the specialized functions of K63 and K27 ubiquitin chains in immune regulation.
Functional Distinctions Between K63 and K27 Ubiquitin Linkages
Table 1: Comparative overview of K63 and K27 ubiquitin linkages in immune signaling
| Characteristic | K63-Linked Ubiquitination | K27-Linked Ubiquitination |
|---|---|---|
| Primary Immune Functions | Regulation of signal transduction, inflammatory signaling, immune cell activation [8] [4] | Regulation of antiviral signaling, particularly in DNA sensing pathways [54] |
| Key Signaling Pathways | NF-κB, MAPK, NLRP3 inflammasome, RLR pathways [8] [4] [27] | cGAS-STING pathway [54] |
| Representative E3 Ligases | TRAF6, TRAF2, cIAP1/2, XIAP [8] [4] | TRIM23, AMFR/gp98 [54] |
| Known Substrates | RIPK2, NEMO, RIPK1, IRAK1 [8] [4] | NEMO, STING [54] |
| Cellular Outcomes | NF-κB activation, proinflammatory cytokine production, inflammasome assembly [8] [4] | Type I IFN production, antiviral state establishment [54] |
| Experimental Evidence Level | Extensive functional characterization across multiple immune pathways [8] [4] [27] | Emerging evidence with limited substrate identification [54] |
Methodological Approaches for Linkage-Specific Ubiquitination Analysis
Tandem Ubiquitin Binding Entities (TUBEs) for High-Throughput Assessment
Protocol: Chain-specific TUBEs assay for linkage-dependent ubiquitination
- Cell Stimulation: Treat THP-1 cells (human monocytic cell line) with either:
- Cell Lysis: Lyse cells using optimized buffer (e.g., RIPA buffer with protease and deubiquitinase inhibitors) to preserve polyubiquitination [8] [16]
- Linkage-Specific Capture: Incubate cell lysates with:
- K63-TUBE coated plates (for K63-linked ubiquitination)
- K48-TUBE coated plates (for K48-linked ubiquitination)
- Pan-TUBE coated plates (for total ubiquitination capture)
- Wash and Detection: Wash plates thoroughly, then detect captured proteins using target-specific antibodies (e.g., anti-RIPK2) via standard ELISA protocols [8] [16] [10]
- Quantification: Measure signal intensity and normalize to appropriate controls
Experimental Validation: This approach has been successfully applied to demonstrate that L18-MDP stimulation induces K63 ubiquitination of RIPK2, which is captured specifically by K63-TUBEs and Pan-TUBEs but not K48-TUBEs. Conversely, RIPK2 PROTAC-mediated ubiquitination is captured specifically by K48-TUBEs and Pan-TUBEs but not K63-TUBEs [8] [16]. The technology enables rapid, quantitative assessment in a 96-well plate format with nanomolar affinity for polyubiquitin chains, representing a significant advancement over traditional Western blotting methods which are lower throughput and provide only semiquantitative data [8] [16] [10].
Mass Spectrometry-Based Approaches for Ubiquitin Linkage Characterization
Protocol: Ubiquitin Absolute Quantification (Ub-AQUA) for branched chain analysis
- Sample Preparation: Enrich ubiquitinated substrates from cellular lysates using affinity purification (e.g., TUBE-based enrichment) [55]
- Proteolytic Digestion: Digest samples with specific proteases (e.g., trypsin) to generate ubiquitin-derived peptides [55]
- Spike-in Standards: Add known quantities of synthetic, stable isotope-labeled ubiquitin peptides representing different linkage types [55]
- LC-MS/MS Analysis: Perform liquid chromatography coupled to tandem mass spectrometry to separate and analyze both endogenous and standard peptides [55]
- Quantitative Analysis: Calculate absolute amounts of specific ubiquitin linkages by comparing endogenous peptide signals to known standard quantities [55]
Application: This approach has been utilized to identify and quantify branched ubiquitin chains, revealing that K11/K48-branched chains account for a significant proportion of ubiquitin polymers and are preferentially recognized by the proteasome [55]. Similar methodology could be adapted specifically for K27 and K63 linkage quantification.
Table 2: Experimental platforms for ubiquitin linkage analysis
| Methodology | Throughput | Linkage Specificity | Key Applications | Limitations |
|---|---|---|---|---|
| Chain-Specific TUBEs | High (96-well format) [8] [16] | High (K63, K48, Pan-specific) [8] [16] | Target protein ubiquitination dynamics, PROTAC characterization [8] [16] [10] | Limited to characterized linkages, antibody-dependent detection |
| Ub-AQUA Mass Spectrometry | Medium | Comprehensive (all linkages) [55] | Discovery of novel linkages, branched chain analysis [55] | Technically demanding, requires specialized instrumentation |
| Linkage-Specific Antibodies | Medium-High | Variable (dependent on antibody quality) | Immunoblotting, immunofluorescence [55] | Specificity validation required, limited multiplexing capability |
| Mutant Ubiquitin Expression | Low-Medium | Defined by mutation | Functional studies in cellular contexts | May not reflect endogenous ubiquitin dynamics |
Signaling Pathways Regulated by K63 and K27 Ubiquitination
K63-Linked Ubiquitination in Innate Immune Signaling
K63-linked ubiquitination serves as a critical regulatory mechanism in multiple innate immune signaling pathways. In the NOD2-RIPK2 pathway, bacterial muramyl dipeptide (MDP) engagement of NOD2 triggers recruitment of E3 ligases (XIAP, cIAP1/2, TRAF2) that catalyze K63-linked ubiquitination of RIPK2 [8]. These K63 ubiquitin chains then serve as scaffolding platforms to recruit and activate the TAK1/TAB1/TAB2 and IKK complexes, ultimately leading to NF-κB activation and production of proinflammatory cytokines [8]. Similarly, in the RIG-I-MAVS pathway, K63-linked ubiquitination of RIG-I by TRIM25, Riplet, TRIM4, and MEX3C promotes its activation and subsequent signaling through MAVS to induce type I interferon production [54]. The TLR-IL-1R signaling pathways also depend on K63 ubiquitination, where TRAF6-mediated K63 chains activate TAK1 and IKK complexes through a mechanism involving Ubc13 and Uev1A [4].
K27-Linked Ubiquitination in Antiviral Defense
K27-linked ubiquitination has emerged as a specialized mechanism in the cGAS-STING DNA sensing pathway, which is essential for antiviral defense against DNA viruses. In this pathway, the E3 ligase AMFR/gp98 catalyzes K27-linked ubiquitination of STING (also known as MITA) in collaboration with an unknown E2 enzyme [54]. This modification is essential for the recruitment of TBK1 and IKKε to STING, leading to phosphorylation and activation of the transcription factor IRF3, which translocates to the nucleus to induce type I interferon gene expression [54]. Additionally, TRIM23 has been identified as another E3 ligase that mediates K27-linked ubiquitination of NEMO (IKKγ), contributing to the antiviral response [54]. Unlike the well-characterized K63 linkages, the molecular mechanisms and functional consequences of K27 ubiquitination remain less understood and represent an active area of research.
Research Reagent Solutions for Ubiquitin Signaling Studies
Table 3: Essential research reagents for studying K63 and K27 ubiquitin linkages
| Reagent Category | Specific Examples | Research Applications | Experimental Considerations |
|---|---|---|---|
| Chain-Specific TUBEs | K63-TUBE, K48-TUBE, Pan-TUBE [8] [16] [10] | Linkage-specific ubiquitin enrichment, high-throughput screening [8] [16] [10] | Nanomolar affinity, available in 96-well plate format for HTS [8] [16] |
| Ubiquitin Linkage Antibodies | Anti-K63-Ub, Anti-K48-Ub, Anti-K27-Ub (commercial) | Immunoblotting, immunofluorescence, immunoprecipitation | Variable specificity between vendors requires validation |
| E3 Ligase Inhibitors | Ponatinib (RIPK2 inhibitor) [8] [16] | Pathway perturbation studies, functional validation | Consider selectivity and off-target effects |
| Activity-Based Probes | Ubiquitin vinyl sulfones, HA-Ub-VS | Deubiquitinase activity profiling, mechanism studies | Can target multiple DUB families with varying selectivity |
| Cell Stimulators | L18-MDP (NOD2 agonist) [8] [16] | Induction of K63 ubiquitination in inflammatory signaling | Concentration and time optimization required |
| Mass Spectrometry Standards | Ub-AQUA quantification standards [55] | Absolute quantification of ubiquitin linkages | Requires specialized MS instrumentation and expertise |
Functional Outcomes in Immune Regulation
Cytokine Production
K63-linked ubiquitination directly regulates cytokine production through multiple mechanisms. In the NOD2-RIPK2 pathway, K63 ubiquitination of RIPK2 leads to NF-κB activation and subsequent production of proinflammatory cytokines including TNF-α, IL-6, and IL-1β [8]. Similarly, K63 ubiquitination events in the TLR signaling pathways activate NF-κB and MAPK pathways, amplifying cytokine gene expression [4]. The NLRP3 inflammasome activation also involves K63 ubiquitination, contributing to processing and secretion of IL-1β and IL-18 [8]. While less extensively characterized, K27-linked ubiquitination of STING in the DNA sensing pathway specifically regulates type I interferon production (IFN-α/β), which establishes an antiviral state in infected and neighboring cells [54].
Antiviral Response
Both K63 and K27 linkages play distinct but essential roles in antiviral immunity. K63-linked ubiquitination regulates multiple antiviral signaling pathways, including RIG-I-MAVS for RNA virus detection and TLR pathways for endosomal viral nucleic acid sensing [4] [56]. These pathways collectively induce type I interferons, proinflammatory cytokines, and interferon-stimulated genes (ISGs) that restrict viral replication. K27-linked ubiquitination provides a more specialized mechanism for DNA virus detection through the cGAS-STING pathway, inducing a robust type I interferon response [54]. Interestingly, many viruses have evolved strategies to directly target these ubiquitination events, highlighting their crucial role in host defense. For example, certain viral proteases can cleave ubiquitin chains or deubiquitinating enzymes can be hijacked to suppress antiviral signaling [56].
Therapeutic Targeting Opportunities
The strategic manipulation of specific ubiquitin linkages presents promising therapeutic opportunities. Targeting K63 ubiquitination has emerged as a potential strategy for modulating inflammatory responses in autoimmune diseases, with inhibitors of enzymes involved in K63 chain assembly (TRAF6, Ubc13, Mms2) showing promise in preclinical models of rheumatoid arthritis and colitis [8]. Additionally, deubiquitinases that specifically cleave K63-linked chains, such as A20 and CYLD, provide another therapeutic avenue for inflammatory conditions [8] [4]. The PROTAC (Proteolysis Targeting Chimeras) technology leverages K48-linked ubiquitination to induce targeted protein degradation, but assessment of PROTAC efficiency now benefits from linkage-specific ubiquitination monitoring using TUBE-based approaches [8] [16]. While therapeutic targeting of K27 linkages remains exploratory, better understanding of its role in antiviral immunity may yield novel antiviral strategies.
Overcoming Experimental Challenges in K63 and K27 Ubiquitin Chain Research
Ubiquitination is a crucial post-translational modification that regulates immune signaling pathways through diverse polyubiquitin chain architectures. Among the eight ubiquitin linkage types, lysine 63 (K63)-linked chains are well-established regulators of inflammatory and immune signaling, while lysine 27 (K27)-linked chains present unique research challenges due to their resistance to certain deubiquitinating enzymes (DUBs). This comparison guide examines the distinct properties, cellular functions, and research methodologies for these two ubiquitin chain types, with particular focus on overcoming the technical challenge of K27 chain DUB resistance in experimental settings.
The functional diversity of ubiquitin chains arises from the ability of ubiquitin moieties to form polymers through different linkage sites. K63-linked chains primarily function in non-proteolytic signaling processes, including innate immune activation and protein trafficking [8] [14]. In contrast, K27 linkages remain less characterized but appear to play important roles in immune regulation, with their unique biochemical properties necessitating specialized research approaches [57].
Structural and Functional Comparisons
Key Characteristics of K63 and K27 Ubiquitin Chains
Table 1: Fundamental properties of K63 and K27 ubiquitin chains
| Property | K63-Linked Ubiquitin Chains | K27-Linked Ubiquitin Chains |
|---|---|---|
| Primary Functions | Non-degradative signaling, inflammatory pathways, protein trafficking, DNA repair | Immune signaling, proteasomal degradation (context-dependent) |
| DUB Sensitivity | Highly sensitive to various DUB families | Resistant to specific DUBs (e.g., UCHL3) |
| Chain Formation Enzymes | TRAF6, Ubc13/Mms2, cIAPs | RNF185, TRIM10, AMFR-GP78/INSIG1 complex |
| Chain Recognition Proteins | NEMO, Optineurin, ABIN proteins | NEMO (weaker affinity) |
| Role in NF-κB Signaling | Direct activation through RIP1, RIP2, and NEMO recruitment | Context-dependent modulation |
| Research Tools | Well-established: TUBEs, linkage-specific antibodies, DUB probes | Limited: synthetic ubiquitin tools, specialized MS methods |
Quantitative Experimental Data Comparison
Table 2: Experimental data from key studies on K63 and K27 ubiquitin chains
| Experimental Parameter | K63-Linked Chains | K27-Linked Chains |
|---|---|---|
| NEMO binding affinity | High (direct recruitment to signaling complexes) | Lower affinity compared to linear and K63 chains [46] |
| DUB inhibition constant (Ki) for UCHL3 | Not inhibited | Strong inhibition (K27Ub2 acts as natural UCHL3 inhibitor) [57] |
| Inflammatory signaling outcome | Strong activation of NF-κB and MAPK pathways | Variable effects depending on cellular context |
| Chain abundance in cells | Second most abundant linkage type | Less abundant, more specialized functions |
| Proteasomal targeting efficiency | Generally not targeted for degradation | Can target substrates for degradation in specific contexts |
DUB Resistance Mechanisms: The K27 Challenge
The primary challenge in studying K27-linked ubiquitin chains stems from their unique resistance to certain deubiquitinating enzymes, particularly UCHL3. Research has revealed that K27-linked diubiquitin (K27Ub2) acts as a natural inhibitor of UCHL3 through an unusual kinetic trap mechanism [57].
Molecular Basis of DUB Resistance
K27Ub2 inhibits UCHL3 through dual mechanisms:
- Covalent inhibition: K27Ub2 binds to the catalytic cysteine of UCHL3
- Allosteric inhibition: K27Ub2 locks the catalytic loop of UCHL3 in a fixed position
This dual inhibition mechanism represents a unique regulatory relationship in the ubiquitin system, suggesting that UCHL3 and K27Ub chains may function as a stimulus-sensor pair in cellular environments [57]. The resistance profile necessitates specialized experimental approaches distinct from those used for K63 chains, which are readily processed by many DUB families.
Experimental Methodologies for Chain Differentiation
Linkage-Specific Ubiquitin Binding Entities (TUBEs)
Tandem Ubiquitin Binding Entities (TUBEs) with nanomolar affinities for specific polyubiquitin chains enable differentiation of linkage-specific ubiquitination in high-throughput formats [8]. The application of chain-specific TUBEs represents a powerful methodology for capturing context-dependent ubiquitination events.
Table 3: TUBE-based differentiation of K63 and K48 linkages in RIPK2 signaling
| Experimental Condition | K63-TUBE Capture | K48-TUBE Capture | Pan-TUBE Capture |
|---|---|---|---|
| L18-MDP stimulation | Strong signal | No appreciable signal | Strong signal |
| RIPK2 PROTAC treatment | No appreciable signal | Strong signal | Strong signal |
| Ponatinib pre-treatment | Inhibited signal | Not applicable | Inhibited signal |
DUB-Based Linkage Differentiation Protocols
Protocol 1: UbiCRest Assay for Linkage Verification
- Synthesize ubiquitin chains of interest using linkage-specific E2 enzymes
- Treat with K48-specific DUB (OTUB1) and K63-specific DUB (AMSH)
- Analyze cleavage patterns by immunoblotting or MS
- K27 chains will show resistance to UCHL3 cleavage, providing differentiation from sensitive linkages [57] [3]
Protocol 2: In Vitro DUB Susceptibility Profiling
- Generate ubiquitin chains using native enzymatic synthesis
- Incubate with panel of DUBs (UCHL3, OTUD5, USP11, etc.)
- Monitor cleavage kinetics by gel electrophoresis or FRET-based assays
- K27 chains show characteristic resistance to UCHL3 while remaining susceptible to other DUBs
Mass Spectrometry Approaches
Advanced mass spectrometry techniques, including UbiSite technology and Ub-AQUA/PRM, enable precise mapping of ubiquitination sites and linkage types [58] [59]. These methods are particularly valuable for studying K27 linkages, as they can distinguish them from more abundant K48 and K63 chains based on unique spectral signatures.
Signaling Pathway Diagrams
K63-Linked Ubiquitination in Inflammatory Signaling
K27 Chain DUB Resistance Mechanism
Experimental Workflow for Linkage Differentiation
Research Reagent Solutions
Table 4: Essential research reagents for studying K27 and K63 ubiquitin chains
| Reagent Category | Specific Examples | Research Application | Considerations |
|---|---|---|---|
| Linkage-Specific TUBEs | K63-TUBE, K48-TUBE, Pan-TUBE | Capture endogenous ubiquitinated proteins with linkage specificity | K27-specific TUBEs not yet commercially available [8] |
| DUB Inhibitors | PR619 (cysteine DUBs), N-ethylmaleimide (NEM), Chloroacetamide (CAA) | Preserve ubiquitination patterns during lysis | NEM can perturb NEMO binding to K63 chains; CAA more specific [3] |
| Linkage-Specific DUBs | OTUB1 (K48-specific), AMSH (K63-specific) | UbiCRest assay for linkage verification | K27 chains resistant to UCHL3 cleavage [57] [3] |
| Synthetic Ubiquitin Tools | K27-linked diubiquitin, K63-linked tetraubiquitin | Structural and biochemical studies | K27Ub2 commercially available for research [57] |
| E3 Ligase Modulators | RIPK2 PROTACs, L18-MDP, Ponatinib | Pathway-specific ubiquitination induction | Enable context-dependent ubiquitination studies [8] |
The differentiation of K27-linked ubiquitin chains from other linkage types, particularly K63 chains, presents distinct methodological challenges primarily due to the unique DUB resistance profile of K27 linkages. While K63 chains function as well-characterized signaling scaffolds in immune pathways, K27 chains represent a more specialized regulatory mechanism with context-dependent functions. Successful experimental approaches require combined methodologies including chain-specific TUBEs, DUB resistance profiling, advanced mass spectrometry, and careful selection of DUB inhibitors. The continuing development of K27-specific research tools will enhance our understanding of this unique ubiquitin linkage and its role in immune regulation.
In the realm of intracellular signaling, ubiquitination stands as a versatile post-translational modification that extends far beyond its canonical role in targeting proteins for proteasomal degradation. The specific linkage type within polyubiquitin chains dictates their functional outcomes, creating a complex "ubiquitin code" that cells utilize to regulate diverse processes [8] [60]. While K48-linked ubiquitin chains predominantly serve as recognition signals for proteasomal degradation, K63-linked chains primarily regulate non-proteolytic functions including inflammatory signaling, protein trafficking, and DNA repair [8] [60]. More recently, K27-linked chains have emerged as significant players in innate immune responses, particularly in the formation of signalosomes that activate downstream inflammatory pathways [60].
The challenge of accurately distinguishing these chain types in complex biological assays represents a significant technical hurdle in ubiquitin research. This difficulty is compounded by the existence of branched ubiquitin chains—heterogeneous polymers containing multiple linkage types—which can confer unique properties and decoding specificities not present in homotypic chains [2] [61] [6]. For instance, K48-K63 branched chains have been shown to regulate NF-κB signaling by modulating deubiquitinase susceptibility while maintaining recognition by specific binding proteins [61]. This review comprehensively compares contemporary methodological approaches for dissecting this complex ubiquitin landscape, with particular emphasis on their application in immune signaling pathways.
Methodological Comparison: Linkage-Specific Assessment Technologies
Tandem Ubiquitin Binding Entities (TUBEs)
Chain-specific TUBEs represent a cutting-edge affinity reagent technology that enables precise capture and analysis of endogenous proteins modified with specific ubiquitin linkages. These specialized binding entities with nanomolar affinities for polyubiquitin chains can differentiate context-dependent linkage-specific ubiquitination of endogenous proteins in high-throughput screening formats [8].
Experimental Protocol for TUBE-Based Ubiquitination Assay:
- Cellular Stimulation: Treat cells (e.g., THP-1 human monocytic cells) with specific agonists. For K63 ubiquitination, stimulate with 200-500 ng/ml L18-MDP (a NOD2 receptor agonist) for 30-60 minutes. For K48 ubiquitination, utilize PROTAC molecules like RIPK2 degrader-2 [8].
- Cell Lysis: Lyse cells in specialized buffer (e.g., RIPA buffer supplemented with 1% SDS, 5 mM N-ethylmaleimide, and complete protease inhibitors) optimized to preserve polyubiquitination by inhibiting deubiquitinases [8] [2].
- Affinity Capture: Incubate cell lysates with linkage-specific TUBE-coated 96-well plates (K48-TUBE, K63-TUBE, or pan-selective TUBE) for 2-4 hours at 4°C [8].
- Washing: Wash plates extensively with wash buffer to remove non-specifically bound proteins.
- Target Detection: Detect captured ubiquitinated proteins by immunoblotting with target-specific antibodies (e.g., anti-RIPK2 for inflammatory signaling studies) [8].
Key Application: Research demonstrates that L18-MDP-stimulated K63 ubiquitination of RIPK2 is captured by K63-TUBEs and pan-selective TUBEs but not K48-TUBEs. Conversely, RIPK2 PROTAC-mediated K48 ubiquitination is captured by K48-TUBEs and pan-selective TUBEs but not K63-TUBEs, confirming the linkage specificity of this approach [8].
Ubiquitin Interactor Pull-Down with Mass Spectrometry
This methodology employs immobilized ubiquitin chains of defined linkages and lengths as bait to enrich for specific ubiquitin-binding proteins (UBPs) from cell lysates, with subsequent identification by liquid chromatography-mass spectrometry (LC-MS) [2] [3].
Experimental Protocol for UBP Pull-Down:
- Ubiquitin Chain Preparation: Synthesize and purify homotypic chains (K48 or K63 Ub2/Ub3) and heterotypic branched chains (K48-K63 BrUb3) using linkage-specific E2 enzymes (e.g., CDC34 for K48, Ubc13/Uev1a for K63) [2] [3].
- Immobilization: Conjugate ubiquitin chains to streptavidin resin via a serine/glycine linker containing a single cysteine residue and maleimide-biotin chemistry [2].
- Deubiquitinase Inhibition: Pre-treat HeLa or yeast cell lysates with deubiquitinase inhibitors—either chloroacetamide (CAA) or N-ethylmaleimide (NEM)—to prevent chain disassembly during incubation [2] [3].
- Enrichment: Incubate inhibited lysates with immobilized ubiquitin chains for 1-2 hours at 4°C.
- Washing and Elution: Wash resin thoroughly and elute bound proteins using mild acid or SDS-containing buffer.
- Identification: Analyze eluates by LC-MS to identify enriched UBPs [2] [3].
Key Application: This approach has identified novel branch-specific ubiquitin interactors, including PARP10, UBR4, and HIP1 (validated by surface plasmon resonance), and revealed chain-length preferences for interactors like CCDC50, FAF1, and DDI2, which prefer Ub3 over Ub2 chains [2] [3].
Linkage-Specific Immunoblotting and Mutant Ubiquitin Approaches
Traditional methods for assessing ubiquitin linkage types have relied heavily on linkage-specific antibodies or expression of mutant ubiquitin proteins where specific lysine residues are mutated to arginine to prevent chain formation through that site [8] [7].
- Limitations: Mass spectrometry remains labor-intensive with limited sensitivity for rapid endogenous ubiquitination changes. Mutant ubiquitin approaches may not accurately represent modifications involving wild-type ubiquitin and can disrupt normal ubiquitin pool dynamics [8]. Linkage-specific antibodies vary considerably in their specificity and may not reliably detect heterotypic or branched chains [2].
Quantitative Comparison of Method Performance Characteristics
Table 1: Performance comparison of methodologies for distinguishing ubiquitin chain linkages
| Method | Throughput | Linkage Specificity | Sensitivity | Ability to Detect Branched Chains | Key Applications |
|---|---|---|---|---|---|
| Chain-Specific TUBEs | High (HTS compatible) | High (K48, K63, pan-specific) | High (detects endogenous proteins) | Limited | PROTAC characterization, inflammatory signaling (e.g., RIPK2, NEMO) [8] |
| UBP Pull-Down + MS | Medium | High (defined chain structures) | Medium | Excellent (identifies branch-specific binders) | Ubiquitin interactome mapping, branch-specific decoder identification [2] [3] |
| Linkage-Specific Antibodies | Medium | Variable (antibody-dependent) | Medium-High | Limited | Initial linkage assessment, immunoblotting, immunofluorescence [8] |
| Mutant Ubiquitin Expression | Low-Medium | Indirect inference | Low-Medium | Limited | Functional studies of specific linkage requirements [8] |
Functional Roles of Ubiquitin Linkages in Immune Signaling Pathways
K63-Linked Ubiquitin Chains
K63-linked polyubiquitination serves as a critical regulatory mechanism in multiple immune signaling pathways without targeting substrates for degradation:
- NF-κB Activation: K63 chains formed on RIPK2 following NOD2 receptor activation by MDP serve as scaffolding platforms that recruit and activate the TAK1/TAB1/TAB2/IKK kinase complexes, leading to NF-κB activation and proinflammatory cytokine production [8]. Multiple E3 ligases including XIAP, cIAP1, cIAP2, and TRAF2 contribute to K63 ubiquitination of RIPK2 in this pathway [8].
- Inflammasome Regulation: K63 ubiquitination contributes to NLRP3 inflammasome activation and subsequent pro-inflammatory cytokine production [8].
- Therapeutic Targeting: Enzymes in the K63 ubiquitination cascade (TRAF6, Ubc13, Mms2) and linkage-specific deubiquitinases represent potential therapeutic targets for inflammatory diseases [8].
K27-Linked Ubiquitin Chains
While less characterized than K63 linkages, K27-linked chains play significant roles in immune regulation:
- Innate Immunity: K27 linkages play roles in innate immune responses, with RNF168 catalyzing K27 ubiquitination of H2A/H2A.X to promote recruitment of DNA damage response proteins to damage foci [60].
- Transcriptional Regulation: MKRN3-mediated K27 ubiquitination of MDB3 promotes epigenetic silencing of GNRH1 transcription [60].
Branched Ubiquitin Chains in Immune Regulation
Branched K48-K63 ubiquitin chains represent a specialized regulatory mechanism in inflammatory signaling:
- NF-κB Signaling Amplification: In response to IL-1β, the E3 ligase HUWE1 generates K48 branches on K63 chains formed by TRAF6. These branched chains permit recognition by TAB2 while protecting K63 linkages from CYLD-mediated deubiquitination, thereby amplifying NF-κB signaling [61].
- Apoptotic Regulation: During apoptotic responses, branched K48/K63 chains on TXNIP are produced through sequential action of ITCH (K63-specific) and UBR5 (K48-specific), resulting in proteasomal degradation of this pro-apoptotic regulator [6].
Visualizing Ubiquitin-Dependent Immune Signaling Pathways
Diagram 1: K63 vs K48 ubiquitination in RIPK2-mediated immune signaling. K63 chains (red) initiate non-proteolytic inflammatory signaling, while K48 chains (red) target RIPK2 for proteasomal degradation.
Experimental Workflow for Comprehensive Ubiquitin Analysis
Diagram 2: Experimental workflows for ubiquitin chain analysis. Parallel approaches using TUBE-based affinity capture (top) and ubiquitin interactor pull-down with mass spectrometry (bottom).
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key research reagents for studying ubiquitin linkages
| Research Tool | Specific Examples | Function and Application | Key Characteristics |
|---|---|---|---|
| Linkage-Specific TUBEs | K48-TUBE, K63-TUBE, Pan-TUBE | Capture and detect endogenous proteins modified with specific ubiquitin linkages; HTS compatible [8] | Nanomolar affinity, linkage-specific binding, preserves endogenous ubiquitination |
| Deubiquitinase Inhibitors | N-ethylmaleimide (NEM), Chloroacetamide (CAA) | Prevent ubiquitin chain disassembly during experiments by inhibiting cysteine proteases [2] [3] | NEM more potent but broader reactivity; CAA more cysteine-specific |
| Defined Ubiquitin Chains | Homotypic K48/K63 Ub2/Ub3, K48-K63 BrUb3 | Bait for interactor pull-down assays; specificity controls; structural studies [2] [3] | Enzymatically synthesized with native isopeptide bonds; precisely defined linkages |
| Linkage-Specific Antibodies | Anti-K48 ubiquitin, Anti-K63 ubiquitin | Immunoblotting, immunofluorescence for initial linkage assessment [8] | Variable specificity; may not detect branched chains effectively |
| E3 Ligase Modulators | L18-MDP (K63 inducer), PROTACs (K48 inducers) | Experimental manipulation of specific ubiquitination pathways [8] | L18-MDP induces K63-RIPK2 via NOD2; PROTACs recruit E3 ligases for degradation |
The evolving methodological landscape for analyzing ubiquitin chain linkages has significantly enhanced our capacity to decipher the complex ubiquitin code in immune signaling. Chain-specific TUBEs offer unprecedented capability for studying endogenous protein ubiquitination in high-throughput formats, while ubiquitin interactor screens with defined chain architectures continue to reveal novel decoding mechanisms for both homotypic and branched chains. The critical challenge of distinguishing non-proteolytic signaling from degradative ubiquitination requires researchers to strategically employ complementary approaches that can address the increasing recognition of heterogeneous and branched ubiquitin chains as specialized regulatory signals. As these technologies continue to mature, they promise to accelerate both fundamental understanding of ubiquitin signaling and therapeutic development targeting ubiquitin pathway components in inflammatory diseases and cancer.
Ubiquitin chains, particularly those involving lysine 63 (K63) and lysine 27 (K27) linkages, represent critical signaling entities in immune regulation. While K63-linked chains are well-established mediators of inflammatory and innate immune signaling, K27-linked chains constitute a more enigmatic component of the ubiquitin code with emerging roles in immune homeostasis. The complexity of these signals is magnified when they form branched architectures, where a single ubiquitin molecule is modified at two different acceptor sites. This comparison guide examines the current experimental understanding of branched ubiquitin chains containing K63 or K27 linkages, providing researchers with structured data and methodologies to navigate this challenging research landscape.
Structural and Functional Comparison of K63 and K27 Linkages
Table 1: Fundamental characteristics of K63 and K27-linked ubiquitin chains
| Characteristic | K63-Linked Chains | K27-Linked Chains |
|---|---|---|
| Primary Immune Functions | NF-κB signaling, T-cell activation, innate immunity pathways, autophagy [4] [27] [14] | Mitochondrial quality control, potential immune regulation [19] [7] |
| Structural Features | Extended, open conformation [2] | Compact conformation with unique dynamics [19] |
| Deubiquitinase (DUB) Sensitivity | Cleaved by specific DUBs (AMSH, others) [19] [2] | Resistant to most deubiquitinases (USP2, USP5, Ubp6) [19] |
| Chain Architecture in Immune Context | Forms homotypic chains and heterotypic branched chains (e.g., with K11, K48) [6] [62] | Structural characterization incomplete; branching potential not fully explored [19] |
| Known Branched Partners | K11, K48 [6] [62] | Limited experimental evidence for specific branched architectures |
Table 2: Experimental approaches for studying K63 and K27 chain functions
| Methodology | Application to K63 Chains | Application to K27 Chains |
|---|---|---|
| Linkage-Specific Ubiquitin Interactor Screens | Identified branch-specific binders (PARP10, UBR4, HIP1) [2] [3] | Limited data available due to reagent and tool limitations |
| Structural Analysis | SANS, NMR, computational modeling [2] | NMR, small-angle neutron scattering, in silico ensemble modeling [19] |
| Immune Signaling Assays | Well-established in TLR, TCR, cytokine signaling pathways [4] [27] | Emerging role in mitochondrial immunity pathways [19] |
| Chain Synthesis Methods | Enzymatic synthesis using Ubc13-Uev1a complex [4] [2] | Non-enzymatic assembly using orthogonal protection strategies [19] |
Key Signaling Pathways and Experimental Findings
K63 Linkages in Branched Architectures
Branched ubiquitin chains containing K63 linkages have been documented in several immune contexts. A seminal study revealed that major histocompatibility complex class II (MHC II) in murine antigen-presenting cells is modified with branched K63/K11-linked ubiquitin chains, with the E3 ligase MARCH1 and E2 enzymes UBE2D2/3 responsible for this modification [62]. This branched architecture regulates MHC II intracellular trafficking and surface expression, with implications for adaptive immunity.
K63 linkages also form branched structures with K48 linkages, creating complex signals that can enhance NF-κB signaling or trigger proteasomal degradation depending on cellular context [2] [6]. These K48/K63 branched chains comprise approximately 20% of all K63 linkages in cells and function as specialized scaffolds for recruiting specific effector proteins [2].
K27 Linkages: Unique Properties and Unknown Branching Potential
K27-linked ubiquitin chains exhibit distinctive biochemical properties that set them apart from other linkage types. Structural analyses reveal that K27-linked di-ubiquitin (K27-Ub2) exhibits no noncovalent interdomain contacts and unique conformational dynamics [19]. Functionally, K27 linkages are remarkably resistant to deubiquitination by most deubiquitinases, including linkage-nonspecific enzymes such as USP2, USP5, and Ubp6 [19]. This DUB resistance may prolong K27-mediated signaling events or create competitive inhibition of DUB activity toward other ubiquitin chain types.
While direct evidence for K27-containing branched chains in immune signaling remains limited, K27 linkages have been implicated in mitochondrial quality control through the regulation of Miro1 degradation [19]. This pathway represents a potential intersection between mitochondrial health and immune signaling that merits further investigation.
Experimental Protocols and Methodologies
Ubiquitin Interactor Screening for Branched Chain Recognition
Comprehensive identification of proteins that specifically recognize branched ubiquitin chains requires carefully designed pulldown experiments:
Chain Synthesis and Immobilization: Synthesize homotypic K63, K27 (where possible), and heterotypic branched chains (e.g., K63/K48, K63/K11) using enzymatic or chemical methods. For K63-containing chains, employ Ubc13-Uev1a complex for K63 linkage formation [2]. For K27 linkages, non-enzymatic assembly using orthogonal protecting groups (Alloc and Boc) has been successful [19].
Immobilization: Add a serine/glycine linker with a single cysteine residue after the C-terminus of the proximal ubiquitin, then conjugate biotin using cysteine-maleimide chemistry for streptavidin resin immobilization [2].
Deubiquitinase Inhibition: During pulldown from cell lysates, use deubiquitinase inhibitors to preserve chain integrity. Both chloroacetamide (CAA) and N-ethylmaleimide (NEM) are effective, though each has distinct off-target effects that should be considered in experimental design [2] [3].
Interactor Identification: Incubate immobilized chains with cell lysates, followed by washing, protein elution, and identification by liquid chromatography-mass spectrometry (LC-MS) [2].
Validation: Confirm specific interactions using complementary techniques such as surface plasmon resonance (SPR) [3].
Structural Characterization of Ubiquitin Chains
For structural analysis of K27 linkages, researchers have employed an integrated approach:
NMR Spectroscopy: Collect 1H-15N NMR spectra separately for each ubiquitin unit within the chain. Quantify amide chemical shift perturbations (CSPs) to identify regions involved in noncovalent interactions or structural changes [19].
Small-Angle Neutron Scattering (SANS): Utilize SANS to obtain low-resolution structural information in solution, complementing high-resolution NMR data [19].
Computational Ensemble Modeling: Integrate experimental data with in silico modeling to generate structural ensembles that represent the dynamic behavior of ubiquitin chains in solution [19].
Visualization of K63 and K27 Linkage Signaling Pathways
K63 vs K27 Linkage Comparison
MHC II K63/K11 Branched Ubiquitination
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key research reagents for studying branched ubiquitin chains
| Reagent / Tool | Function / Application | Specific Examples / Notes |
|---|---|---|
| Linkage-Specific E2 Enzymes | Catalyze formation of specific ubiquitin linkages | Ubc13-Uev1a for K63 linkages [4] [2]; Limited specific E2s for K27 |
| E3 Ubiquitin Ligases | Mediate substrate specificity and chain formation | MARCH1 for MHC II ubiquitination [62]; TRAF6 for K63 chains [4] |
| Deubiquitinase Inhibitors | Preserve ubiquitin chains during experiments | Chloroacetamide (CAA), N-ethylmaleimide (NEM) [2]; Consider off-target effects |
| Branched Chain Synthesis Systems | Generate defined ubiquitin architectures | Enzymatic synthesis with specific E2 combinations [2] [3]; Chemical synthesis for K27 [19] |
| Ubiquitin-Binding Domains (UBDs) | Detect and characterize specific ubiquitin signals | Linkage-specific UBDs for pull-down assays; Limited tools for K27 linkages |
| Mass Spectrometry Platforms | Identify ubiquitin linkages and interactors | LC-MS for interactor screens [2] [3]; Proteomics for endogenous chain analysis |
The landscape of branched ubiquitin chains containing K63 or K27 linkages presents both challenges and opportunities for researchers. While K63-containing branched architectures are increasingly recognized as important signaling entities in immune regulation, K27 linkages remain structurally and functionally enigmatic. The experimental approaches outlined here provide a framework for investigating these complex post-translational modifications, with particular emphasis on the need for specialized reagents and methodologies tailored to each linkage type. As tool development advances, particularly for K27 linkages, our understanding of how branched ubiquitin chains fine-tune immune signaling will undoubtedly expand, potentially revealing new therapeutic targets for immune-related diseases.
The study of ubiquitin chains, particularly K63 and K27-linked varieties, represents a critical frontier in understanding immune signaling regulation. These post-translational modifications employ identical ubiquitin monomers but generate functionally distinct signals through different isopeptide linkages. K63-linked ubiquitin chains are established as major non-proteolytic signaling scaffolds in immune and inflammatory pathways, regulating receptors including TLR, TNF-R, IL-1R, and RIG-I-like receptors [4]. In contrast, K27-linked chains remain more enigmatic but are increasingly implicated in regulation of innate immunity and exhibit unique biochemical properties that distinguish them from all other ubiquitin linkages [19] [7]. This comparison guide objectively analyzes experimental approaches for differentiating these chain types, providing validated methodologies for researchers investigating ubiquitin-dependent immune signaling mechanisms.
Fundamental Comparison of K63 and K27 Ubiquitin Chains
Table 1: Core Biochemical and Functional Properties of K63 and K27 Ubiquitin Chains
| Property | K63-Linked Ubiquitin Chains | K27-Linked Ubiquitin Chains |
|---|---|---|
| Primary Immune Function | Scaffold for immune signalosome assembly; activates NF-κB, MAPK, IRF pathways [4] | Regulator of innate immunity; implicated in mitochondrial trafficking and damage response [19] [7] |
| Structural Characteristics | Adopts extended conformation with minimal non-covalent interdomain contacts [19] | Unique structural ensemble; exhibits largest NMR spectral perturbations among all linkages [19] |
| Proteasomal Targeting | Non-proteolytic; does not target proteins for degradation [4] [27] | Non-proteolytic; slows proteasomal degradation of substrates like Miro1 [19] |
| Chain Assembly Enzyme | Ubc13-Uev1a E2 complex with various E3 ligases (TRAF6, cIAPs) [4] | Linkage-specific E3 ligases not fully characterized; requires specialized assembly methods [19] |
| Cellular Abundance | Highly abundant; rapidly accumulates during oxidative stress and immune activation [4] [11] | Lower abundance; challenging to study due to technical limitations [19] |
Table 2: Deubiquitinase (DUB) Specificity Profiles
| DUB Enzyme | K63 Linkage Response | K27 Linkage Response | Experimental Utility |
|---|---|---|---|
| USP5 (IsoT) | Effectively cleaved [19] | Resistant to cleavage [19] | Differential cleavage distinguishes K27 from K63 |
| USP2 | Cleaved efficiently [19] | Minimal cleavage activity [19] | Validates K27 identity through resistance |
| Ubp6 | Processed effectively [19] | Highly resistant [19] | Confirms K27 chains in complex mixtures |
| AMSH | Selective cleavage [19] | Not a preferred substrate [19] | K63-specific validation |
| OTUB1 | Minimal cleavage [19] | Not a preferred substrate [19] | K48-specific control |
| Cezanne | Not a preferred substrate [19] | Not a preferred substrate [19] | K11-specific control |
| UCHL3 | Standard processing [57] | Potent inhibition [57] | Unique K27 sensor through inhibition profiling |
Orthogonal Methodologies for Chain Validation
Deubiquitinase Profiling Assay
Protocol Objective: To distinguish K63 and K27 linkages through differential sensitivity to deubiquitinase enzymes.
Experimental Workflow:
- Sample Preparation: Isolate ubiquitinated proteins or synthesize ubiquitin chains of interest using non-enzymatic methods for K27 linkages [19] or enzymatic assembly with Ubc13 for K63 linkages [4].
- DUB Incubation: Incubate ubiquitin chains (0.5-1 μg) with individual DUBs (0.1-0.5 μg) in appropriate reaction buffers for 30-60 minutes at 37°C.
- Reaction Termination: Add SDS-PAGE loading buffer with 50mM DTT and heat at 95°C for 5 minutes.
- Analysis: Resolve products by SDS-PAGE (12-15% gels) followed by Western blotting with ubiquitin-specific antibodies or fluorescent staining.
Key Validation Metrics:
- K27 linkages demonstrate resistance to multiple DUBs including USP5, USP2, and Ubp6 [19]
- K63 linkages show specific cleavage by AMSH and processing by linkage-nonspecific DUBs [19]
- K27-Ub2 can serve as a competitive inhibitor of DUB activity toward other linkages [19]
DUB Profiling Workflow for K63 vs K27 Discrimination
Linkage-Specific Ubiquitinomics
Protocol Objective: To precisely identify and quantify K63 ubiquitination sites during immune activation using the Ub-DiGGer method.
Experimental Workflow:
- Cell Culture & Treatment: Culture yeast or mammalian cells in SILAC media with light/heavy isotopes. Treat with immune stimuli (e.g., 0.6mM H₂O₂ for oxidative stress, IL-1β, or LPS) for 30 minutes [11].
- Protein Extraction: Lyse cells in Tris-HCl buffer (pH 7.5) with chloroacetamide, protease inhibitors, and FLAG-tagged K63-TUBE for linkage-specific preservation.
- Sequential Enrichment:
- Primary enrichment: Anti-FLAG resin to capture K63-ubiquitinated proteins
- Elution with glycine (pH 2.5), neutralization, and tryptic digestion
- Secondary enrichment: Anti-diGly lysine beads to capture ubiquitinated peptides [11]
- Mass Spectrometry Analysis: LC-MS/MS with Q-Exactive instrument, 75-min gradient; database search with GlyGly(K) as variable modification.
Key Applications:
- Identified 1,100+ K63 ubiquitination sites in yeast during oxidative stress [11]
- Revealed ribosome ubiquitination as key mechanism in stress response [11]
- Enables quantitative tracking of K63 ubiquitination dynamics in immune signaling
Structural Analysis by NMR and SANS
Protocol Objective: To characterize unique structural and dynamic properties of K27 vs. K63 linkages.
Experimental Workflow:
- Chain Preparation: Synthesize homogeneous K27-Ub2 using non-enzymatic methods with mutually orthogonal removable amine-protecting groups (Alloc and Boc) [19]. Prepare K63-Ub2 enzymatically using chain-terminating mutations.
- NMR Spectroscopy: Collect ¹H-¹⁵N NMR spectra for each Ub unit separately. Quantify amide chemical shift perturbations (CSPs) between distal/proximal Ub and monoUb.
- Small-Angle Neutron Scattering (SANS): Analyze chain conformation in solution under native conditions.
- Computational Modeling: Generate conformational ensembles using in silico approaches informed by experimental data [19].
Key Structural Insights:
- K27-Ub2 exhibits no noncovalent interdomain contacts in distal Ub [19]
- K27-Ub2 shows largest CSPs in proximal Ub among all linkages [19]
- K27 linkages display unique conformational ensembles distinct from K63 extended conformation
Signaling Pathway Analysis
K63 Ubiquitination in Immune Activation
K63-linked ubiquitination serves as a master regulator in multiple immune signaling pathways. In TNFR1 signaling, K63 ubiquitination of RIPK1 by the TRAF2-cIAP1-Ubc13-UbcH5 complex facilitates formation of Complex I, which activates NF-κB and MAPK-driven transcriptional activation of prosurvival genes [4]. In the TLR3 pathway, TRIF adaptor-TRAF3-mediated K63 ubiquitination activates the TBK1-IRF3-IFN-β pathway for antiviral response [4]. Additionally, B cell receptor activation involves TRAF2-mediated K63 ubiquitination of cIAP1/2, leading to K48-linked degradation of TRAF3 and subsequent NIK stabilization that activates alternative NF-κB signaling [4].
K63 Ubiquitin Signaling in Immune Pathways
Emerging Roles of K27 Linkages in Immune Regulation
While less characterized than K63 chains, K27 linkages exhibit distinct regulatory patterns in immune signaling. K27 chains are observed on mitochondrial protein Miro1, regulating mitochondrial trafficking and potentially influencing immune cell metabolism [19]. K27-linked ubiquitination is implicated in regulation of innate immunity through pathways yet to be fully elucidated [7]. Notably, K27-Ub2 demonstrates unique inhibitory activity against deubiquitinase UCHL3 through both covalent and allosteric mechanisms, suggesting a regulatory role in controlling DUB activity in immune cells [57].
Research Reagent Solutions
Table 3: Essential Research Tools for K63 and K27 Ubiquitin Studies
| Reagent Category | Specific Examples | Research Application | Supplier/Generation Method |
|---|---|---|---|
| Linkage-Specific Ubiquitin Chains | K27-Ub2 (non-enzymatic synthesis); K63-Ub2 (enzymatic assembly) | Structural studies; DUB specificity profiling; in vitro reconstitution | Non-enzymatic synthesis with orthogonal protecting groups [19] |
| E2 Enzyme Systems | Ubc13-Uev1a complex | K63 chain assembly in biochemical assays | Recombinant expression and purification [4] |
| DUB Panels | USP5, USP2, Ubp6, AMSH, OTUB1, Cezanne, UCHL3 | Linkage validation through differential cleavage | Commercial sources or recombinant expression [19] [57] |
| Mass Spectrometry Reagents | SILAC amino acids; Anti-FLAG resin; Anti-diGly beads | Ubiquitinomics site mapping | Commercial vendors (e.g., Thermo Scientific, Cell Signaling) [11] |
| K63-Specific Inhibitors | GPS2 (G-Protein Pathway Suppressor 2) | Inhibition of Ubc13-mediated K63 ubiquitination | Recombinant expression; study mitochondrial translation regulation [63] |
| Linkage-Specific Antibodies | Anti-K63 ubiquitin (EMD Millipore) | Western blot detection of K63 chains | Commercial vendors [11] |
| Branched Chain Reagents | K48-K63 branched ubiquitin chains | NF-κB signaling studies; CYLD protection assays | In vitro assembly using HUWE1 and TRAF6 [61] |
Integrated Validation Strategy
A robust validation strategy for K63 versus K27 ubiquitin chains requires orthogonal method integration:
Primary Confirmation: Begin with DUB profiling panels to exploit the unique K27 resistance to multiple deubiquitinases [19]. This provides initial linkage assignment with standard laboratory techniques.
Secondary Validation: Employ linkage-specific ubiquitinomics (Ub-DiGGer) for comprehensive mapping of K63 sites in cellular contexts [11]. This approach delivers system-wide quantification and identification of specific modified residues.
Tertiary Structural Analysis: Utilize NMR and SANS for challenging specimens or when novel linkages are suspected [19]. These methods provide biophysical validation of chain architecture.
Functional Correlation: Implement pharmacological inhibitors like GPS2 for K63-specific functional studies [63] or utilize branched chain analysis where K48-K63 hybrid chains regulate NF-κB signaling [61].
This multi-layered approach ensures accurate chain-type identification while contextualizing results within relevant immune signaling pathways, providing researchers with a comprehensive framework for ubiquitin linkage validation.
The ubiquitin system, particularly K63-linked ubiquitination, serves as a central regulator of immune signaling pathways, controlling processes from inflammatory response activation to interferon production. Numerous pathogens, including SARS-CoV-2 and other viruses, have evolved sophisticated mechanisms to exploit this system for immune evasion. This review comprehensively examines how viral proteins directly target and disrupt K63 ubiquitination events to subvert host innate immunity. We compare these immune evasion strategies across different viruses, detail experimental methodologies for studying these interactions, and provide key resources for researchers investigating ubiquitin-pathogen interactions. Understanding these mechanisms provides critical insights for developing targeted antiviral therapies that counteract pathogen-mediated ubiquitin subversion.
Ubiquitination is a crucial post-translational modification that governs numerous cellular processes through the covalent attachment of ubiquitin to target proteins. Among the various ubiquitin linkage types, K63-linked ubiquitination stands out as a major non-proteolytic signaling mechanism that regulates immune pathway activation [8] [4]. Unlike K48-linked chains that primarily target proteins for proteasomal degradation, K63 linkages function as scaffolding elements that facilitate protein-protein interactions and complex assembly in key immune signaling pathways [8] [6].
The biological significance of K63 ubiquitination in immunity is profound. This modification regulates signal transduction through multiple receptors of innate and adaptive immunity, including Toll-like receptors (TLRs), RIG-I-like receptors (RLRs), Nod-like receptors (NLRs), and cytokine receptors [4] [64]. K63 ubiquitination is catalyzed by specific E2 enzymes, notably Ubc13 in complex with its non-enzymatic cofactors Uev1a or Mms2, which work in concert with various E3 ligases to build K63-linked chains on target substrates [4] [17]. These chains then serve as docking platforms for recruiting downstream signaling components through ubiquitin-binding domains, ultimately leading to the activation of transcription factors NF-κB and IRF3 that drive inflammatory and antiviral responses [8] [4].
The critical positioning of K63 ubiquitination in immune activation makes it a prime target for viral interference. Pathogens have consequently evolved mechanisms to disrupt these signaling cascades, with SARS-CoV-2 representing a particularly sophisticated example of ubiquitin system exploitation [65] [4] [66].
SARS-CoV-2 Interference with K63 Ubiquitination
SARS-CoV-2 employs multiple viral proteins to systematically disrupt K63 ubiquitination-dependent immune signaling at various levels. The table below summarizes the primary viral mechanisms and their specific targets:
Table 1: SARS-CoV-2 proteins targeting K63 ubiquitination pathways
| Viral Protein | Target/Mechanism | Functional Consequence | Reference |
|---|---|---|---|
| NSP5 (3C-like protease) | Inhibits K63 polyubiquitination of RIG-I | Blocks RIG-I activation and downstream IFN signaling | [65] |
| ORF9b | Inhibits K63-linked ubiquitination of NEMO | Suppresses IKK complex formation and NF-κB activation | [65] |
| NSP13 | Binds to TBK1 and TBKBP1 | Inhibits TBK1 phosphorylation and IRF3 activation | [65] |
| M (membrane) protein | Blocks MAVS aggregation and TRAF3-TANK-TBK1-IKKε complex formation | Disrupts RLR signaling complex assembly | [65] |
| N (nucleocapsid) protein | Inhibits RIG-I ubiquitination and STAT1/2 phosphorylation | Suppresses both IFN induction and signaling | [65] |
These coordinated attacks on the ubiquitin system allow SARS-CoV-2 to effectively delay and dampen the host interferon response, creating a window of opportunity for viral replication and establishment of infection [65] [66]. The delayed interferon response is a hallmark of COVID-19 pathology and contributes to both viral persistence and the hyperinflammatory state observed in severe cases [66].
The molecular interactions between SARS-CoV-2 proteins and host ubiquitin machinery represent promising targets for therapeutic intervention. For instance, the inhibition of K63 ubiquitination of RIG-I by NSP5 prevents the formation of signaling complexes necessary for interferon induction, effectively blinding cells to viral presence during early infection [65]. Similarly, ORF9b's targeting of NEMO ubiquitination disrupts a critical node in multiple immune signaling pathways, providing broad-spectrum suppression of antiviral defenses [65].
Comparative Viral Interference Strategies
Beyond SARS-CoV-2, other viruses similarly target K63 ubiquitination pathways to evade immune surveillance. Recent research on Senecavirus A (SVA) reveals a sophisticated mechanism where the virus destabilizes the interaction between TRIM25 and RIG-I, thereby reducing K63-linked ubiquitination of RIG-I and dampening innate immune activation [67] [68]. This strategy mirrors SARS-CoV-2's approach of targeting early pathogen recognition events.
The TRIM25-RIG-I axis represents a common vulnerability exploited by multiple viruses. TRIM25 catalyzes K63-linked ubiquitination of RIG-I, which is essential for its activation and subsequent signal transduction leading to interferon production [67] [68]. Beyond directly interfering with RIG-I ubiquitination, some viruses additionally modulate metabolic pathways to further suppress immune signaling. For instance, TRIM25 also promotes the K48-linked ubiquitination and degradation of platelet-type phosphofructokinase (PFKP), a key glycolytic enzyme, thereby inhibiting virus-induced glycolysis that would otherwise support viral replication [68].
Table 2: Comparative viral interference strategies targeting K63 ubiquitination
| Virus | Primary Target | Interference Mechanism | Outcome |
|---|---|---|---|
| SARS-CoV-2 | Multiple (RIG-I, NEMO, MAVS) | Viral proteins (NSP5, ORF9b, M) directly inhibit K63 ubiquitination | Delayed IFN response, enhanced replication |
| Senecavirus A | TRIM25-RIG-I interaction | Destabilizes TRIM25-RIG-I complex | Reduced K63 ubiquitination of RIG-I, immune evasion |
| Multiple RNA viruses | RIG-I/MDA5 activation | Sequestration of ubiquitination sites or E3 ligases | Blunted antiviral signaling |
These comparative observations highlight conserved strategies across different viral families, suggesting evolutionary convergence on critical regulatory nodes in the ubiquitin system. The recurring targeting of RIG-I and MAVS signaling components underscores their pivotal role in antiviral defense and the selective advantage gained by viruses that successfully disrupt their function.
Experimental Approaches for Studying K63 Ubiquitination
Investigating pathogen-mediated disruption of ubiquitination requires specialized methodologies that can capture these transient, linkage-specific modifications. Several key experimental approaches have been developed to address these challenges:
Tandem Ubiquitin Binding Entities (TUBEs)
TUBEs are engineered affinity reagents with nanomolar affinities for polyubiquitin chains that can be designed to be either pan-selective or linkage-specific [8] [16]. These reagents protect polyubiquitin chains from deubiquitinase (DUB) activity during cell lysis and processing, preserving labile ubiquitination events for analysis. In recent applications, K63-selective TUBEs have successfully captured inflammatory stimulus-induced K63 ubiquitination of RIPK2, while K48-selective TUBEs specifically captured PROTAC-induced K48 ubiquitination of the same protein [8] [16]. This specificity enables precise dissection of linkage-specific ubiquitination events in response to different cellular stimuli or pathogenic insults.
The typical TUBE-based ubiquitination assay involves several key steps:
- Cell stimulation (e.g., with L18-MDP for RIPK2 ubiquitination) or viral infection
- Cell lysis with specialized buffers optimized to preserve polyubiquitination
- Affinity enrichment using TUBE-coated plates or magnetic beads
- Immunoblotting with target-specific antibodies to detect ubiquitinated proteins
This approach has been successfully adapted to high-throughput screening formats, enabling quantitative assessment of linkage-specific ubiquitination in response to potential therapeutic compounds or pathogenic factors [8].
Mass Spectrometry-Based Ubiquitinomics
Advanced mass spectrometry techniques allow system-wide identification and quantification of ubiquitination sites and linkage types. These approaches typically involve:
- DiGly remnant enrichment following tryptic digestion to identify ubiquitination sites
- Linkage-specific antibodies to immunopurify particular chain types
- Absolute quantification using stable isotope-labeled ubiquitin
While powerful, these methods require sophisticated instrumentation and can be limited in sensitivity for capturing rapid, transient changes in endogenous protein ubiquitination [8].
Signaling Pathways and Experimental Workflows
The diagram below illustrates the core innate immune signaling pathway targeted by viruses and the experimental workflow for studying its disruption:
Figure 1: Immune signaling pathway targeted by viruses and experimental workflow for studying K63 ubiquitination
Essential Research Tools and Reagents
The table below summarizes key reagents and methodologies used in ubiquitination research, particularly for studying pathogen interference:
Table 3: Research reagent solutions for studying K63 ubiquitination
| Reagent/Method | Specific Application | Key Features | Example Use Cases |
|---|---|---|---|
| K63-TUBEs | Selective enrichment of K63-linked polyubiquitin chains | Linkage-specific nanomolar affinity; protects from DUBs | Studying inflammatory signaling; viral disruption of K63 chains [8] |
| K48-TUBEs | Selective enrichment of K48-linked chains | Specifically captures degradation-targeted ubiquitination | PROTAC mechanism studies; viral protein degradation [8] |
| Pan-TUBEs | Broad ubiquitin chain capture | General ubiquitination assessment when linkage unknown | Initial screening of ubiquitination events [8] |
| Linkage-specific Antibodies | Immunoblot/immunofluorescence detection | Validates chain specificity; spatial localization | Confirming TUBE specificity; cellular localization [8] |
| Ubiquitin Mutants (K63R) | Dissecting linkage-specific functions | Prevents K63 chain formation while preserving other linkages | Defining K63-specific signaling roles [8] |
| DUB Inhibitors | Preserving ubiquitination signatures | Stabilizes labile ubiquitination events during processing | Enhancing detection of transient ubiquitination [4] |
These tools have enabled significant advances in understanding pathogen interactions with the ubiquitin system. For instance, applying K63-TUBEs to study SARS-CoV-2 infected cells could reveal specific defects in RIG-I or MAVS ubiquitination that contribute to immune evasion. Similarly, combining TUBE-based enrichment with mass spectrometry provides a powerful approach for unbiased identification of viral targets within the ubiquitin system.
The systematic targeting of K63 ubiquitination by SARS-CoV-2 and other viruses highlights the critical importance of this modification in antiviral immunity. Understanding these interference mechanisms provides not only insights into viral pathogenesis but also reveals novel opportunities for therapeutic intervention. Future research directions should focus on:
- Developing specific inhibitors of viral proteins that disrupt K63 ubiquitination
- Engineering stabilized ubiquitin variants that resist viral deubiquitinases
- Exploiting branched ubiquitin chains that may be more resistant to viral interference
- Developing high-throughput screening platforms to identify compounds that restore proper ubiquitin signaling during infection
The continuing evolution of tools to study ubiquitination, particularly linkage-specific approaches like TUBE technology, will enable increasingly precise dissection of how pathogens manipulate host signaling networks. These advances will ultimately inform the development of next-generation antivirals that target the delicate interplay between viruses and the host ubiquitin system.
Direct Functional Comparison: K63 vs K27 Ubiquitination in Specific Immune Pathways
Ubiquitination, a fundamental post-translational modification, regulates diverse cellular processes through the formation of polyubiquitin chains. Among the various linkage types, lysine 63 (K63) and lysine 27 (K27)-linked ubiquitin chains have emerged as critical regulators of immune signaling pathways [8] [4] [19]. While both linkage types perform non-degradative functions, they exhibit remarkable differences in their structural conformations, dynamics, and interactions with receptor proteins. This comparative analysis examines the distinct structural properties of K63 and K27-linked ubiquitin chains and their specialized roles in immune signal transduction, providing researchers with experimental frameworks for studying these complex ubiquitin signals.
Structural Conformations and Dynamics
Quaternary Structure and Conformational Ensembles
K63-linked ubiquitin chains exhibit exceptional conformational plasticity, sampling multiple quaternary states in their ligand-free form. Paramagnetic relaxation enhancement (PRE) NMR studies reveal that free K63-diubiquitin (K63-Ub2) exists as a dynamic ensemble comprising multiple closed and open quaternary states [69]. This inherent flexibility enables K63-Ub2 to engage with diverse binding partners through a conformational selection mechanism, whereby target proteins select and stabilize one of the preexisting conformational states [69]. The equilibrium between these states can be modulated by point mutations, directly affecting binding affinities for various K63-Ub2 ligands [69].
In contrast, K27-linked chains display unique structural characteristics that distinguish them from all other ubiquitin linkages. Comprehensive structural analysis using NMR spectroscopy and small-angle neutron scattering demonstrates that K27-Ub2 exhibits no noncovalent interdomain contacts in the distal ubiquitin unit [19]. Surprisingly, while the distal ubiquitin shows minimal chemical shift perturbations, the proximal ubiquitin displays the most widespread and pronounced chemical shift perturbations among all ubiquitin linkages, indicating substantial structural modulation around the K27 linkage site [19].
Table 1: Comparative Structural Properties of K63 vs K27-linked Diubiquitin
| Structural Property | K63-linked Diubiquitin | K27-linked Diubiquitin |
|---|---|---|
| Conformational States | Multiple open and closed states [69] | Unique structural ensemble [19] |
| Interdomain Contacts | Transient and dynamic [69] | Minimal in distal Ub [19] |
| Chemical Shift Perturbations | Moderate and localized [69] | Extensive in proximal Ub [19] |
| Structural Dynamics | High quaternary flexibility [69] | Unique dynamics around linkage site [19] |
| Deubiquitinase Susceptibility | Cleaved by linkage-specific DUBs [19] | Resistant to most DUBs [19] |
Deubiquitinase Resistance and Structural Implications
A remarkable functional property of K27-linked chains is their exceptional resistance to deubiquitination. When screened against six deubiquitinases representing different DUB families, K27-Ub2 demonstrated complete resistance to cleavage by linkage non-specific DUBs including USP2, USP5, and Ubp6 [19]. K27 was the only linkage that resisted cleavage by USP5 (IsoT), and K27-Ub2 also resisted disassembly by the reconstituted proteasome lid core complex containing Rpn11 [19]. This exceptional stability likely influences the signaling lifetime of K27-linked ubiquitination events in immune pathways and contributes to its competitive inhibition of DUB activity toward other linkages [19].
Immune Signaling Pathways and Functions
K63-linked Ubiquitination in Immune Regulation
K63-linked ubiquitin chains serve as master regulators in multiple immune signaling pathways. Ubc13-catalyzed K63 ubiquitination controls critical immune functions including chronic inflammation, pathogen responses, lymphocyte activation, and regulatory signaling [4]. In the NF-κB pathway, K63 ubiquitination regulates signaling adaptors including cellular inhibitor of apoptosis proteins (cIAPs), TNF receptor-associated factors (TRAFs), and receptor-interacting serine/threonine-protein kinase 2 (RIPK2) [8] [4].
A key mechanism involves NOD2 receptor activation by muramyldipeptide (MDP), which recruits RIPK2 and E3 ligases including XIAP, leading to K63 ubiquitination of RIPK2 [8]. These K63 chains serve as signaling scaffolds to recruit and activate the TAK1/TAB1/TAB2/IKK kinase complexes, ultimately driving NF-κB activation and production of proinflammatory cytokines [8].
Figure 1: K63-linked Ubiquitination in NOD2/RIPK2-mediated Immune Signaling
K27-linked Ubiquitination in Immune Regulation
While less extensively characterized than K63 linkages, K27-linked ubiquitin chains contribute to the regulation of innate immune responses [19]. K27 chains are implicated in mitochondrial regulation through modification of mitochondrial trafficking protein Miro1, where they slow down proteasomal degradation and act as markers of mitochondrial damage [19]. This mechanism potentially links K27 ubiquitination to mitochondrial-dependent immune signaling pathways.
The unique structural properties of K27 chains, particularly their resistance to deubiquitinases, suggest they may form stable signaling platforms in immune pathways where transient signals are undesirable. Their unique conformational ensemble may also facilitate recognition by specialized receptor proteins that discriminate K27 linkages from other ubiquitin chain types [19].
Table 2: Functional Roles in Immune Signaling Pathways
| Functional Aspect | K63-linked Chains | K27-linked Chains |
|---|---|---|
| Primary Immune Functions | NF-κB activation, inflammasome regulation, lymphocyte signaling [4] | Innate immunity regulation, mitochondrial damage response [19] |
| Signaling Mechanism | Scaffold for kinase complex assembly [8] | Stable signaling platforms [19] |
| Regulatory Dynamics | Dynamic and reversible [4] | Stable due to DUB resistance [19] |
| Key Substrates | RIPK2, NEMO, TRAF6 [8] [4] | Miro1, mitochondrial proteins [19] |
| Pathogen Targeting | SARS-CoV-2 immune evasion targets [4] | Emerging role in pathogen response [19] |
Experimental Methodologies
Chain-Specific Ubiquitination Detection
The development of tandem ubiquitin binding entities (TUBEs) has enabled specific detection and analysis of linkage-specific ubiquitination events. Recent advances employ chain-specific TUBEs with nanomolar affinities for particular polyubiquitin chains in high-throughput screening assays [8]. This methodology allows researchers to investigate ubiquitination dynamics of endogenous proteins like RIPK2 in response to specific stimuli.
For studying K63 ubiquitination, inflammatory agent L18-MDP stimulation induces K63 ubiquitination of RIPK2, which can be specifically captured in 96-well plates coated with K63-TUBEs or pan-selective TUBEs, but not with K48-TUBEs [8]. Conversely, PROTAC-induced K48 ubiquitination is captured using K48-TUBEs and pan-selective TUBEs, while K63-TUBEs show minimal capture of PROTAC-induced signals [8]. This specificity enables precise dissection of context-dependent ubiquitination events.
Figure 2: Experimental Workflow for Chain-Specific TUBE Assay
Structural Analysis Techniques
NMR Spectroscopy for Conformational Dynamics
Solution NMR spectroscopy provides atom-specific information about ubiquitin chain conformations and dynamics. Key approaches include:
- Chemical shift perturbation analysis: Mapping interaction surfaces and conformational changes by comparing chemical shifts between diubiquitin and monoUb [19] [69]
- Paramagnetic relaxation enhancement (PRE): Detecting transient interdomain contacts and visualizing quaternary arrangements between ubiquitin subunits [69]
- Relaxation dispersion measurements: Characterizing conformational dynamics at microsecond to millisecond timescales relevant for biological function [70]
For K63-Ub2, PRE NMR with strategically placed paramagnetic probes at N25C or K48C sites has revealed the coexistence of open and closed states in the free form [69]. This technique is particularly valuable for characterizing dynamic ensembles and transient states that evade conventional structural methods.
Structural Ensemble Modeling
Complementary techniques including small-angle neutron scattering (SANS) and computational modeling provide insights into conformational ensembles [19]. For K27-Ub2, combined NMR, SANS, and in silico ensemble modeling have revealed unique structural properties that explain its functional behavior, particularly its resistance to deubiquitinases [19].
Research Reagent Solutions
Table 3: Essential Research Tools for Studying Ubiquitin Chain Interactions
| Research Tool | Specific Example | Function and Application |
|---|---|---|
| Chain-specific TUBEs | K63-TUBEs, K48-TUBEs, Pan-TUBEs [8] | Selective capture of linkage-specific polyubiquitinated proteins from cell lysates |
| Linkage-specific DUBs | OTUB1 (K48-specific), AMSH (K63-specific), Cezanne (K11-specific) [19] | Validation of chain linkage identity; cleavage specificity controls |
| NMR Isotope Labels | 15N-labeled ubiquitin, 13C/15N-labeled ubiquitin [19] [69] | NMR studies of chain conformation and dynamics |
| Paramagnetic Probes | MTSL probe conjugation at N25C or K48C sites [69] | PRE NMR measurements of inter-subunit distances and dynamics |
| Activity-Based Probes | DUB activity probes with K27-Ub2 [19] | Monitoring DUB resistance and enzymatic activity |
| E2/E3 Enzyme Pairs | Ubc13-Uev1a (K63-specific), undefined for K27 [4] [19] | In vitro reconstitution of specific ubiquitin linkages |
Discussion and Research Implications
The structural and functional specialization of K63 and K27-linked ubiquitin chains demonstrates how ubiquitin topology encodes biological information. K63 chains function as dynamic signaling scaffolds in well-defined immune pathways, with their conformational plasticity enabling recognition by multiple effector proteins [8] [4] [69]. In contrast, K27 chains appear to form stable signaling platforms where DUB resistance extends signaling duration, potentially in pathways requiring sustained signals [19].
From a drug discovery perspective, the unique structural features of both chain types present attractive targeting opportunities. For K63 chains, inhibiting specific E2-E3 partnerships like Ubc13-Uev1a could modulate overactive immune signaling in inflammatory diseases [4]. The resistance of K27 chains to DUBs suggests potential for developing stabilized ubiquitin signals for therapeutic purposes [19].
Future research should focus on identifying specialized receptor proteins that specifically recognize K27 linkages, elucidating the complete signaling pathways regulated by K27 ubiquitination, and developing additional tools to manipulate specific ubiquitin linkages in cellular environments. The continued refinement of chain-specific detection methods and structural analysis techniques will be essential for advancing our understanding of how ubiquitin chain architecture dictates functional outcomes in immune signaling and beyond.
Innate immune signaling represents the host's first line of defense against pathogenic invasion, relying on pattern recognition receptors (PRRs) to detect conserved pathogen motifs and initiate protective responses. The TLR/TRIF, RIG-I, and STING pathways constitute three critical signaling arms that coordinate antiviral and inflammatory responses. Beyond receptor-ligand interactions, post-translational modifications (PTMs), particularly ubiquitination, serve as essential regulatory mechanisms that dictate the intensity, duration, and outcome of immune signaling. Among ubiquitin linkages, K63-linked chains have been extensively characterized as key mediators of signal transduction in innate immunity, while emerging research highlights K27-linked chains as specialized regulators with unique functional properties. This review comprehensively compares these three pivotal innate immune pathways, with particular emphasis on how K63 and K27 ubiquitin linkages confer specificity to immune signaling cascades and outcomes.
Pathway Architectures and Ubiquitin-Dependent Signaling Mechanisms
RIG-I/MAVS Pathway Architecture
Retinoic acid-inducible gene-I (RIG-I) and melanoma differentiation-associated gene 5 (MDA5) constitute the RIG-I-like receptor (RLR) family that detects viral RNA in the cytoplasm [71]. These receptors contain two N-terminal caspase recruitment domains (CARDs), a central DExD/H-box helicase domain, and a C-terminal domain. Upon viral RNA recognition, RIG-I and MDA5 engage the critical adaptor protein mitochondrial antiviral signaling protein (MAVS) through CARD-CARD interactions, triggering MAVS aggregation into prion-like filaments on mitochondrial membranes [72] [71]. This activated MAVS platform then recruits tumor necrosis factor receptor-associated factors (TRAFs), particularly TRAF3 and TRAF6, initiating downstream signaling that culminates in type I interferon and pro-inflammatory cytokine production.
Ubiquitin Regulation: The RIG-I/MAVS pathway is extensively regulated by ubiquitination, with K63-linked chains playing well-established activating roles and K27-linked chains emerging as important non-proteolytic regulators [72]. MAVS undergoes at least three distinct ubiquitination patterns: K63-linked ubiquitylation promotes MAVS aggregation and signal activation; K48-linked ubiquitylation mediates proteasomal degradation; and K27-linked ubiquitylation primarily facilitates autophagic degradation of MAVS, representing a unique regulatory mechanism [72]. Specific E3 ubiquitin ligases including TRIM31 catalyze K63-linked polyubiquitination of MAVS at Lys10, Lys311, and Lys461, enhancing cellular antiviral responses [72]. Conversely, E3 ligases MARCH8, RNF34, and RNF5 recruit K27-linked ubiquitin chains to MAVS, prompting its autophagic degradation and attenuation of innate immune signaling [72].
Figure 1: RIG-I/MAVS Pathway Architecture and Ubiquitin Regulation. Viral RNA detection triggers MAVS aggregation and downstream signaling. K63-linked ubiquitination promotes activation, while K27-linked chains facilitate autophagic degradation.
STING Pathway Architecture
The stimulator of interferon genes (STING) pathway constitutes the primary cytoplasmic DNA sensing mechanism in mammalian cells [73] [74]. Cyclic GMP-AMP synthase (cGAS) serves as the central DNA sensor that catalyzes synthesis of the second messenger 2'3'-cyclic GMP-AMP (cGAMP) upon DNA binding. cGAMP then activates STING (also known as TMEM173, MITA, ERIS, or MPYS), an endoplasmic reticulum membrane protein containing four transmembrane domains at its N-terminus, followed by a dimerization domain and C-terminal cytoplasmic domain [73]. Activated STING undergoes profound conformational changes, oligomerizes, and translocates from the ER to the Golgi apparatus, where it recruits and activates TANK-binding kinase 1 (TBK1). TBK1 subsequently phosphorylates interferon regulatory factor 3 (IRF3), leading to IRF3 dimerization, nuclear translocation, and initiation of type I interferon gene expression [74].
Ubiquitin Regulation: STING activation is tightly controlled by ubiquitination, though current research has primarily established roles for K63-linked chains in STING activation, while the functions of K27-linked chains in this pathway remain less characterized [74]. The E3 ubiquitin ligase TRIM56 catalyzes K63-linked ubiquitination of STING following double-stranded DNA stimulation, inducing STING dimerization and enhancing antiviral responses [74]. Additionally, recent research identifies ZC3HAV1 as a critical regulator that facilitates STING oligomerization and trafficking, though its connection to specific ubiquitin linkages requires further investigation [74]. The regulatory potential of K27 linkages in STING signaling represents an important area for future research, particularly given their established roles in related pathways.
Figure 2: STING Pathway Architecture and Ubiquitin Regulation. Cytosolic DNA detection triggers cGAS-STING signaling. K63-linked ubiquitination promotes STING activation, while ZC3HAV1 facilitates oligomerization.
TLR/TRIF Pathway Architecture
Toll-like receptors (TLRs) represent a conserved family of transmembrane PRRs that detect pathogen-associated molecular patterns (PAMPs) in extracellular and endosomal compartments [71]. Among TLR adaptors, TIR-domain-containing adapter-inducing interferon-β (TRIF) mediates signaling downstream of TLR3 and TLR4, driving interferon regulatory factor 3 (IRF3) activation and type I interferon production [71]. TRIF activation leads to recruitment of TRAF family members and subsequent TBK1 and IKKε kinase activation, which phosphorylate IRF3 and NF-κB components to initiate antiviral and inflammatory gene expression programs.
Ubiquitin Regulation: The TLR/TRIF pathway extensively employs K63-linked ubiquitin chains for signal propagation, while research on K27-linked chains in this pathway remains limited [71]. TRAF6-mediated K63-linked ubiquitination creates docking platforms for TAK1 and IKK complex activation through ubiquitin binding domains, enabling downstream NF-κB and MAPK signaling [71]. The potential involvement of K27 linkages in TRIF pathway regulation represents an open research question with significant implications for understanding pathway-specific ubiquitin coding.
Comparative Analysis of Pathway Features and Ubiquitination Patterns
Table 1: Comparative Features of Innate Immune Signaling Pathways
| Feature | RIG-I/MAVS Pathway | STING Pathway | TLR/TRIF Pathway |
|---|---|---|---|
| Primary Ligand | Viral RNA (5'ppp RNA, dsRNA) | Cytosolic DNA (2'3'-cGAMP) | dsRNA (TLR3), LPS (TLR4) |
| Receptor Localization | Cytoplasmic | ER Membrane | Endosomal/Cell Membrane |
| Key Adaptor Protein | MAVS (Mitochondrial) | STING (ER-Golgi) | TRIF (Cytoplasmic) |
| Primary Kinase Complex | TBK1/IKKε | TBK1 | TBK1/IKKε |
| Transcription Factors | IRF3, IRF7, NF-κB | IRF3, NF-κB | IRF3, NF-κB |
| K63-Linked Ubiquitin Role | MAVS aggregation & activation | STING dimerization & trafficking | TRAF6-mediated TAK1/IKK activation |
| K27-Linked Ubiquitin Role | MAVS autophagic degradation | Not well characterized | Not well characterized |
| Key Regulatory E3 Ligases | TRIM31 (K63), MARCH8/RNF34/RNF5 (K27) | TRIM56 (K63) | TRAF6 (K63) |
Table 2: Experimental Evidence for Ubiquitin Linkage Functions
| Experimental Approach | K63-Linked Chain Evidence | K27-Linked Chain Evidence |
|---|---|---|
| Linkage-Specific Antibodies | Confirm involvement in MAVS, STING, TRIF signaling | Detect K27 chains on MAVS during viral infection |
| Ubiquitin Mutants (K→R) | K63R mutants impair signal transduction | K27R mutants block autophagic degradation |
| E3 Ligase Knockdown/KO | TRIM31 KD impairs RIG-I signaling; TRIM56 KD blocks STING activation | MARCH8/RNF34 KD enhances IFN production |
| Deubiquitinase Studies | OTUD4 suppresses K48-linked MAVS; USP19 removes K63 chains | Most DUBs cannot cleave K27 linkages [19] |
| Biophysical Analysis | SANS, NMR show open conformations enabling protein recruitment | NMR reveals unique compact conformation [19] |
| Functional Assays | Luciferase reporter assays; IFN-β ELISA; plaque assays | Competitive DUB inhibition; proteasome resistance |
Research Reagent Solutions for Ubiquitin Signaling Studies
Table 3: Essential Research Reagents for Innate Immunity and Ubiquitination Studies
| Reagent Category | Specific Examples | Research Applications |
|---|---|---|
| Linkage-Specific Ubiquitin Antibodies | Anti-K63-Ub, Anti-K27-Ub, Anti-K48-Ub | Immunoblotting, immunofluorescence to detect specific chain types |
| Ubiquitin Mutants | K63R, K27R, K48R ubiquitin constructs | Determine linkage-specific functions in reconstitution assays |
| E3 Ligase Modulators | TRIM31, TRIM56, MARCH8 expression/siRNA | Manipulate specific ubiquitination events |
| Deubiquitinase Inhibitors | USP-specific inhibitors | Probe ubiquitination dynamics and chain stability |
| Pathway Agonists | 2'3'-cGAMP (STING), poly(I:C) (TLR3/RIG-I), HSV-1 | Activate specific innate immune pathways |
| Reporter Systems | IFN-β luciferase, ISRE-luciferase reporters | Quantify pathway activation downstream of ubiquitination |
Key Methodologies for Studying Ubiquitin-Dependent Immune Regulation
Ubiquitin Chain Disassembly Assays
Deubiquitinase (DUB) resistance profiling provides critical insights into ubiquitin chain topology and function. Experimental Protocol: Incubate purified ubiquitinated proteins (e.g., MAVS or STING) with linkage-selective DUBs including OTUB1 (K48-specific), AMSH (K63-specific), and Cezanne (K11-specific). Notably, K27-linked di-ubiquitin exhibits unique resistance to most deubiquitinases, including linkage-non-specific enzymes USP2, USP5, and Ubp6 [19]. This biochemical property distinguishes K27 linkages from other chain types and contributes to their specialized signaling functions.
Structural Analysis of Ubiquitin Chains
NMR Spectroscopy and Small-Angle Neutron Scattering (SANS): Structural characterization reveals how different ubiquitin linkages dictate unique conformations that determine effector protein interactions. For K27-Ub2 chains, NMR chemical shift perturbations indicate minimal noncovalent interdomain contacts in the distal ubiquitin but significant perturbations in the proximal ubiquitin [19]. This distinctive structural architecture likely underlies the unique functional properties and receptor recognition patterns of K27 linkages compared to K48- and K63-linked chains.
Functional Immune Signaling Assays
Luciferase Reporter Assays: Cells are transfected with IFN-β or ISRE promoter-driven luciferase constructs together with ubiquitin mutants or E3 ligase expression vectors. After stimulation with pathway-specific agonists (e.g., HSV-1 for STING, SeV for RIG-I), luciferase activity quantifies pathway output.
Cytokine Production Measurement: ELISA or quantitative PCR analysis of type I interferons and inflammatory cytokines (IFN-β, TNF-α, IL-6) from primary macrophages or cell lines following genetic or pharmacological perturbation of ubiquitination components.
The TLR/TRIF, RIG-I/MAVS, and STING pathways represent distinct but interconnected arms of the innate immune system, each employing specialized ubiquitin-dependent mechanisms for signal regulation. While K63-linked ubiquitin chains serve well-established roles in signal activation across all three pathways, K27-linked chains emerge as specialized regulators, particularly within the RIG-I/MAVS pathway where they direct autophagic degradation of MAVS and modulate antiviral responses. The unique biochemical properties of K27 linkages—including their compact structure and remarkable resistance to deubiquitinases—enable non-proteolytic regulatory functions that expand the ubiquitin code beyond degradation signals. Future research delineating the full spectrum of K27 functions across innate signaling pathways, particularly in STING and TLR pathways, will enhance our understanding of immune homeostasis and identify novel therapeutic targets for inflammatory diseases, antiviral strategies, and cancer immunotherapy.
The adaptive immune system maintains a delicate balance between mounting effective responses against pathogens and preserving tolerance to self-tissues. Post-translational modifications, particularly ubiquitination, serve as crucial regulatory mechanisms in this equilibrium. Among the various ubiquitin linkage types, K63-linked and K27-linked chains have emerged as pivotal but functionally divergent regulators of immune signaling pathways [4] [5]. This review systematically compares how these specific ubiquitin modifications differentially control two fundamental aspects of adaptive immunity: T-cell receptor (TCR) signaling and the balance between regulatory T cells (Tregs) and T helper 17 cells (Th17). Understanding these distinct regulatory mechanisms provides critical insights into the molecular basis of immune homeostasis and identifies potential therapeutic targets for autoimmune diseases, cancer, and other immunological disorders.
Comparative Analysis of K63-Linked vs. K27-Linked Ubiquitin Chains
Table 1: Functional Comparison of K63-Linked vs. K27-Linked Ubiquitin Chains in Immune Regulation
| Characteristic | K63-Linked Ubiquitination | K27-Linked Ubiquitination |
|---|---|---|
| Primary Function | Scaffold for signalosome assembly; activation signaling | Balancing activation/inhibition; diverse outcomes |
| Chain Topology | Unanchored or anchored chains | Typically anchored to substrate |
| Key E2 Enzyme | Ubc13 (with Uev1a cofactor) | Multiple, less specialized |
| Representative E3 Ligases | TRAF6, TRAF2, cIAPs | TRIM23, Rhbdd3 complex |
| Deubiquitinases (DUBs) | A20, CYLD, OTUD7B | Undefined repertoire |
| Role in TCR Signaling | Positive regulation via PKCθ-IKK-NF-κB pathway | Not directly established in TCR |
| Role in Innate Immunity | Activates NF-κB, MAPK in PRR pathways | Activates NF-κB, IRF3 in RLR signaling |
| Effect on Treg/Th17 Balance | Promotes Th1/Th17 differentiation | Limits Th17 via inhibition of excessive NF-κB |
| Therapeutic Implications | Chronic inflammation, autoimmunity | Autoimmunity, inflammatory disorders |
K63-linked ubiquitination primarily functions as a scaffold for signal transduction, facilitating the assembly of multiprotein complexes that drive immune activation [4]. This linkage type does not target proteins for proteasomal degradation but instead creates platforms that recruit proteins containing ubiquitin-binding domains, thereby promoting downstream signaling events. In contrast, K27-linked ubiquitination exhibits more context-dependent functions, sometimes activating and sometimes inhibiting immune responses depending on the cellular context and specific substrates involved [5]. For instance, K27-linked chains on NEMO can serve as platforms for recruiting both activating and inhibitory components, allowing for precise regulation of the immune response magnitude.
The enzymatic machinery governing these ubiquitination types also differs substantially. K63-linked chains are uniquely synthesized by the Ubc13-Uev1a E2 complex, which specifically catalyzes K63-specific linkages [4]. This complex partners with various E3 ligases, including TRAF6 in TCR signaling and cIAPs in innate immune pathways. For K27-linked chains, the E2 enzymes are less specialized, with E3 ligases like TRIM23 playing prominent roles in immune signaling pathways [5]. The deubiquitinating enzymes that reverse these modifications similarly differ, with A20 and CYLD preferentially targeting K63-linked chains, while the DUBs for K27 chains remain less characterized.
Divergent Roles in T-Cell Receptor Signaling Pathways
K63-Linked Ubiquitination in TCR Proximal Signaling
TCR activation initiates a sophisticated signaling cascade that begins with the phosphorylation of immunoreceptor tyrosine-based activation motifs (ITAMs) in CD3 subunits by Src-family protein tyrosine kinases Lck and Fyn [75] [76]. This phosphorylation recruits ZAP-70, which subsequently phosphorylates adaptor proteins LAT and SLP-76, nucleating the formation of a multi-protein signalosome [76]. K63-linked ubiquitination enters this pathway primarily through the activation of PKCθ, which in turn stimulates the IKK-NF-κB pathway through a CBM complex (CARD11-BCL10-MALT1) [76].
The CBM complex recruits TRAF6, an E3 ubiquitin ligase that catalyzes the formation of K63-linked ubiquitin chains on various signaling components, including itself [4]. These chains serve as docking platforms for TAK1, which activates the IKK complex, ultimately leading to NF-κB activation and transcription of genes essential for T-cell activation, proliferation, and differentiation [77] [76]. This K63-dependent pathway is critical for translating TCR engagement into effective immune responses, and its dysregulation can lead to either immunodeficiency or autoimmunity.
Contrasting Functions of K27-Linked Ubiquitination
While K63-linked ubiquitination generally promotes TCR signaling, the role of K27-linked chains in direct TCR signaling is less established. However, in related immune pathways, K27-linked ubiquitination demonstrates dual regulatory functions that suggest potential modulatory roles in T-cell activation. In RIG-I-like receptor (RLR) signaling, TRIM23 catalyzes K27-linked auto-ubiquitination, leading to TBK1 activation and subsequent IRF3-mediated type I interferon production [5]. Simultaneously, K27-linked chains on NEMO can recruit Rhbdd3, which then brings the deubiquitinase A20 to signaling complexes, removing K63-linked chains from NEMO and preventing excessive NF-κB activation [5].
This balanced regulatory function of K27-linked ubiquitination suggests it might serve as a feedback mechanism to fine-tune TCR signaling intensity and duration, although direct evidence in TCR pathways requires further investigation. The ability of K27-linked chains to both activate and inhibit signaling pathways positions them as potential modulators of T-cell activation thresholds, which could influence T-cell differentiation fate decisions, including the Treg/Th17 balance.
Regulation of Treg/Th17 Balance Through Ubiquitin-Dependent Mechanisms
K63-Linked Ubiquitination in T-Cell Differentiation
The balance between regulatory T cells (Tregs) and T helper 17 cells (Th17) represents a crucial determinant in immune homeostasis, with dysregulation contributing to various autoimmune diseases [78]. K63-linked ubiquitination significantly influences this balance through multiple mechanisms. In Th17 cells, the IL-1b-TRAF6 signaling axis catalyzes K63-linked ubiquitination of YAP, leading to its stabilization and nuclear translocation, where it promotes expression of pro-inflammatory genes [27]. This mechanism illustrates how K63-linked chains can drive inflammatory T-cell responses.
Additionally, K63-linked ubiquitination regulates the differentiation and function of Tregs. Recent evidence suggests that Treg versus Th1/Th17 cell subset regulation involves K63 ubiquitination, positioning this modification as a key determinant in T-cell fate decisions [4]. The deubiquitinating enzyme CYLD, which specifically cleaves K63-linked chains, negatively regulates T-cell activation, with CYLD-deficient T cells exhibiting hypersensitivity to TCR stimulation [4]. This suggests that precise control of K63-linked ubiquitination is essential for maintaining appropriate T-cell activation thresholds and preventing excessive immune responses that could disrupt the Treg/Th17 balance.
K27-Linked Ubiquitination in Immune Balance
While less extensively studied in T-cell biology, emerging evidence indicates that K27-linked ubiquitination contributes to the maintenance of immune balance. The Rhbdd3-A20 axis, triggered by K27-linked ubiquitination on NEMO, limits excessive NF-κB activation and has been shown to control dendritic cell activation and limit Th17 cell-mediated colitis in mouse models [5]. This pathway represents a mechanism through which K27-linked ubiquitination can indirectly influence the Treg/Th17 balance by modulating antigen-presenting cell function and subsequent T-cell differentiation.
The tissue gut microenvironment, particularly gut microbiota-derived metabolites, represents another intersection point between ubiquitination and Treg/Th17 balance. Short-chain fatty acids (SCFAs) from gut microbiota restore Th17/Treg balance in rheumatoid arthritis through metabolic reprogramming via AMPK/mTOR signaling, epigenetic regulation by inhibiting HDAC, and modulation of cytokine cascades [79]. While not directly linked to ubiquitination in the available literature, these pathways may interface with ubiquitin-dependent regulatory mechanisms through shared signaling components.
Table 2: Experimental Models for Studying Ubiquitination in T-Cell Biology
| Experimental Approach | Key Reagents/Methods | Applications in T-Cell Research |
|---|---|---|
| Genetic Manipulation | Conditional knockout mice (TRAF6, A20, CYLD), CRISPR/Cas9 | Define in vivo functions of ubiquitination enzymes |
| Biochemical Analysis | Immunoprecipitation, ubiquitin pull-down assays, mass spectrometry | Identify ubiquitinated substrates and linkage types |
| Chemical Biology | Ubiquitin variant probes, linkage-specific antibodies, DUB inhibitors | Detect specific chain types and modulate ubiquitination |
| Imaging Techniques | FRET-based ubiquitin sensors, confocal microscopy, live-cell imaging | Visualize spatial and temporal dynamics of ubiquitination |
| Functional Assays | T-cell differentiation cultures, suppression assays, cytokine measurement | Determine effects on Treg/Th17 differentiation and function |
Experimental Approaches and Methodologies
Assessing Ubiquitination in TCR Signaling
Investigating ubiquitination in TCR signaling requires specialized methodologies to capture these dynamic post-translational modifications. Co-immunoprecipitation assays combined with ubiquitin linkage-specific antibodies allow researchers to identify specific ubiquitin chain types associated with TCR signaling components [5]. For example, to study K63-linked ubiquitination of PKCθ or components of the CBM complex, researchers can stimulate T-cells with anti-CD3/CD28 antibodies, followed by immunoprecipitation of the target protein and immunoblotting with K63 linkage-specific ubiquitin antibodies [4] [76].
Mass spectrometry-based proteomics provides a comprehensive approach for identifying ubiquitination sites and linkage types. Using di-glycine remnant antibodies to enrich ubiquitinated peptides, researchers can map specific ubiquitination sites on proteins involved in TCR signaling and T-cell differentiation [5]. Quantitative proteomics enables comparison of ubiquitination patterns under different stimulation conditions or between T-cell subsets, revealing dynamic changes in the ubiquitin landscape during T-cell activation.
Evaluating Treg/Th17 Differentiation and Function
To investigate how ubiquitination impacts the Treg/Th17 balance, researchers employ in vitro T-cell differentiation assays. Naïve CD4+ T-cells are isolated and cultured under Treg-polarizing conditions (TGF-β, IL-2) or Th17-polarizing conditions (TGF-β, IL-6, IL-1β, IL-23) in the presence of modulators of ubiquitination pathways, such as E3 ligase inhibitors or deubiquitinase targeting compounds [78] [80]. The resulting T-cell populations are analyzed by flow cytometry for signature transcription factors (FoxP3 for Tregs, RORγt for Th17) and cytokine production (IL-10 for Tregs, IL-17A for Th17).
Functional suppression assays determine the regulatory capacity of Tregs generated under different ubiquitination conditions. Tregs are co-cultured with responder T-cells at varying ratios, and proliferation of responder cells is measured by 3H-thymidine incorporation or CFSE dilution [80]. To assess the pathogenicity of Th17 cells, adoptive transfer models of autoimmune diseases such as experimental autoimmune encephalomyelitis (EAE) can be employed, where Th17 cells generated under specific ubiquitination conditions are transferred to recipient mice and disease progression is monitored.
Research Reagent Solutions for Ubiquitin and T-Cell Research
Table 3: Essential Research Reagents for Investigating Ubiquitination in T-Cell Signaling
| Reagent Category | Specific Examples | Research Applications |
|---|---|---|
| Linkage-Specific Antibodies | Anti-K63-ubiquitin, Anti-K27-ubiquitin, Anti-linear ubiquitin | Detect specific ubiquitin chain types in immunoassays |
| E3 Ligase Modulators | TRAF6 inhibitors, TRIM23 expression constructs, cIAP antagonists | Manipulate specific ubiquitination pathways |
| Deubiquitinase Reagents | A20 siRNA, CYLD inhibitors, OTUD7B expression plasmids | Reverse or prevent deubiquitination events |
| T-Cell Activation Reagents | Anti-CD3/CD28 antibodies, PMA/ionomycin, antigenic peptides | Stimulate TCR signaling under controlled conditions |
| T-cell Differentiation Kits | Treg/Th17 polarization kits, cytokine cocktails, signature antibodies | Generate and identify specific T-cell subsets |
| Ubiquitin Activity Probes | HA-Ub-VS, biotin-Ub-PA, linkage-specific ubiquitin binders | Detect active ubiquitination enzymes and characterize chains |
Signaling Pathway Diagrams
K63-Linked Ubiquitination in TCR-NF-κB Signaling
Figure 1: K63-Ubiquitination in TCR-NF-κB Signaling Pathway
Ubiquitin-Mediated Regulation of Treg/Th17 Balance
Figure 2: Ubiquitin-Mediated Regulation of Treg/Th17 Balance
The comparative analysis of K63-linked and K27-linked ubiquitin chains reveals their divergent yet complementary functions in regulating adaptive immune responses. K63-linked chains primarily drive activation pathways in TCR signaling and promote inflammatory T-cell responses, while K27-linked chains exhibit more nuanced regulatory functions that can both activate and inhibit immune signaling depending on cellular context. These differential roles position these ubiquitin linkage types as critical determinants of the Treg/Th17 balance, with implications for understanding autoimmune disease pathogenesis and developing targeted immunotherapies.
Future research directions should focus on elucidating the crosstalk between different ubiquitin linkage types in determining T-cell fate decisions. The development of more specific tools to manipulate distinct ubiquitination events in precise T-cell subsets will enable finer dissection of these regulatory mechanisms. Additionally, exploring the interface between ubiquitination pathways and other immune regulatory mechanisms, such as those mediated by gut microbiota-derived metabolites, may reveal integrated networks controlling immune homeostasis. From a therapeutic perspective, targeting specific ubiquitination events represents a promising but challenging approach for modulating pathological immune responses in autoimmune diseases while preserving protective immunity.
Ubiquitination, a crucial post-translational modification, regulates virtually all cellular processes, with different ubiquitin linkage types encoding distinct functional outcomes. Among these, K63-linked and K27-linked polyubiquitin chains have emerged as critical non-degradative signals in immune signaling pathways [7] [81]. While K48-linked chains predominantly target substrates for proteasomal degradation, K63 and K27 linkages function as sophisticated molecular scaffolds that regulate protein-protein interactions, subcellular localization, and enzymatic activity [7]. The E3 ubiquitin ligase TRIM21 sits at the crossroads of these pathways, demonstrating remarkable specificity in utilizing both K63 and K27 linkages to mediate seemingly disparate functions in cancer, autoimmunity, and antiviral immunity [82]. This review systematically compares the mechanisms and functional consequences of TRIM21-mediated K63 versus K27 ubiquitination within immune signaling pathways, providing researchers with experimental frameworks and reagent solutions for investigating this biologically significant dichotomy.
Structural and Functional Dichotomy of K63 and K27 Linkages
Table 1: Fundamental Characteristics of K63 vs. K27 Ubiquitin Linkages
| Characteristic | K63-Linked Ubiquitination | K27-Linked Ubiquitination |
|---|---|---|
| Primary Functions | Non-degradative signaling, intracellular trafficking, innate immune signaling, kinase activation [7] | Immune response regulation, inflammatory signaling, protein-protein interactions [82] |
| Chain Architecture | Extended, open conformation [83] | Compact, closed conformation (inferred) |
| Prototypical Role | Platform for assembly of signaling complexes [7] | Recruitment of specific signaling components [82] |
| TRIM21 Linkage Usage | PD-1 stabilization (K233) [82] | MAVS ubiquitination in viral infection [82] |
| Cellular Outcome | Protein stabilization, signal transduction [82] | Signal transduction, immune activation [82] |
| Deubiquitinases (DUBs) | USP53, USP54, ZUFSP, JAMM family [83] [7] | Limited information, OTUD1 (for K6) [7] |
The functional specialization of ubiquitin linkages stems from their distinct structural properties and decoding mechanisms. K63-linked chains adopt an extended, open conformation that serves as an ideal platform for assembling signaling complexes, particularly in innate immune pathways and kinase activation cascades [83] [7]. In contrast, K27-linked ubiquitination appears to facilitate more specific protein-protein interactions, though structural characterization remains limited [82]. This fundamental architectural difference enables these linkage types to recruit distinct ubiquitin-binding domain-containing proteins, thereby initiating unique downstream signaling events from similarly modified substrates.
TRIM21-Mediated K63 Ubiquitination in Cancer and Autoimmunity
Molecular Mechanism of PD-1 Stabilization via K63 Ubiquitination
Recent research has illuminated a critical role for TRIM21 in stabilizing programmed cell death-1 (PD-1) through K63-linked ubiquitination at residue K233 [82]. This modification creates a competitive dynamic with K48-linked ubiquitination, which typically targets PD-1 for proteasomal degradation. The K63 linkage stabilizes PD-1 by directly antagonizing K48-mediated degradation, leading to increased PD-1 expression on CD8+ T cells and subsequent impairment of anti-tumor immunity [82]. This mechanism represents a sophisticated example of how competing ubiquitin codes integrate to fine-tune critical immune checkpoint protein levels.
Figure 1: TRIM21 Stabilizes PD-1 via K63-Linked Ubiquitination
Functional Consequences in Cancer and Therapeutic Implications
The stabilization of PD-1 via TRIM21-mediated K63 ubiquitination has profound implications for cancer immunotherapy. Genetic ablation of Trim21 significantly decreases PD-1 expression and enhances activation of cytotoxic CD8+ T cells, sensitizing tumors to anti-CTLA-4 immunotherapy [82]. In preclinical models, Trim21 knockout anti-CD19 CAR-T cells exhibit significantly improved anti-tumor efficacy compared to their wild-type counterparts [82]. These findings position the TRIM21-PD-1 axis as a promising therapeutic target for overcoming resistance to existing immune checkpoint blockade therapies. Interestingly, TRIM21 demonstrates context-dependent roles in cancer, functioning as both an oncogene and tumor suppressor in different tissue types, potentially through its utilization of different ubiquitin linkages [82].
TRIM21-Mediated K27 Ubiquitination in Antiviral Immunity
MAVS Ubiquitination in Innate Immune Signaling
In contrast to its K63-linked ubiquitination activities, TRIM21 also catalyzes K27-linked polyubiquitination of mitochondrial antiviral-signaling protein (MAVS) during viral infections, leading to activation of interferon (IFN) signaling [82]. This modification occurs through a distinct molecular mechanism that facilitates the assembly of antiviral signaling complexes rather than stabilizing individual proteins. The K27 linkage on MAVS serves as a platform for recruiting downstream signaling components that ultimately drive type I interferon production, establishing a potent antiviral state in infected cells.
Figure 2: TRIM21 Activates Antiviral Signaling via K27-Linked Ubiquitination
Balancing Antiviral Defense and Autoimmunity
The TRIM21-MAVS K27 ubiquitination axis represents a crucial antiviral defense mechanism, but its dysregulation may contribute to autoimmune pathology. As an initial autoantigen identified in patients with autoimmune conditions including systemic lupus erythematosus and Sjögren's syndrome, TRIM21 sits at the intersection of antiviral immunity and loss of self-tolerance [82]. This connection suggests that proper regulation of TRIM21-mediated K27 ubiquitination is essential for maintaining immune homeostasis, with potential implications for understanding the molecular triggers of autoimmune diseases that frequently follow viral infections.
Experimental Approaches for Studying K63 and K27 Ubiquitination
Methodologies for Linkage-Specific Ubiquitination Analysis
Table 2: Key Experimental Protocols for K63 vs. K27 Ubiquitination Studies
| Methodology | Application to K63 Studies | Application to K27 Studies | Key Technical Considerations |
|---|---|---|---|
| Linkage-specific DUB assays | USP53/USP54 cleavage specificity confirms K63 linkage [83] | Limited tools available; requires linkage-specific antibodies | Use tetraubiquitin panels to confirm linkage specificity [83] |
| Mass spectrometry-based proteomics | Identify K63-specific interactors and substrates [82] | Map K27 modification sites and interacting proteins | Requires enrichment with linkage-specific ubiquitin-binding domains |
| Genetic knockout/knockdown | Validate physiological roles in PD-1 regulation [82] | Determine function in MAVS signaling and IFN activation [82] | Use CRISPR/Cas9 for complete knockout or siRNA for transient knockdown |
| In vitro ubiquitination assays | Characterize TRIM21 E3 ligase activity with specific E2 enzymes | Define E2 requirements for K27 linkage formation | Use ubiquitin mutants (K63R, K27R) to confirm linkage specificity |
| Activity-based probes | Profile DUB specificity using propargylamide-based probes [83] | Limited probe availability for K27-specific DUBs | HA-ubiquitin-PA probes can enrich active DUBs [83] |
Linkage-Specific Reagent Solutions
Table 3: Essential Research Reagents for K63 and K27 Ubiquitination Studies
| Reagent Category | Specific Examples | Research Application | Experimental Function |
|---|---|---|---|
| Ubiquitin Mutants | Ubiquitin-K63R, Ubiquitin-K27R, Ubiquitin-ΔGG [81] | Define linkage specificity in vitro and in cells | Eliminate specific linkage types to assess functional requirements |
| Linkage-Specific DUBs | USP53, USP54 (K63-specific) [83] | Validate K63 linkage formation in cellular assays | Cleave specific ubiquitin linkages to confirm chain type |
| Activity-Based Probes | HA-Ubiquitin-PA (propargylamide) [83] | Profile active deubiquitinases in cell lysates | Covalently label active DUBs for enrichment and identification |
| Linkage-Specific Antibodies | Anti-K63-Ub, Anti-K27-Ub | Detect endogenous chain types by immunoblot/IF | Recognize specific ubiquitin linkages in biological samples |
| Recombinant E2 Enzymes | UBE2N/UEV1A (K63-specific), Specific E2s for K27 | In vitro ubiquitination assays with TRIM21 | Determine E2 requirements for specific linkage formation |
| Tetrabiquitin Panels | K63-linked tetraUb, K48-linked tetraUb, etc. [83] | DUB linkage specificity profiling | Substrates for assessing enzymatic activity and specificity |
Integrated Analysis: Therapeutic Implications and Future Directions
The dichotomy between TRIM21-mediated K63 and K27 ubiquitination presents both challenges and opportunities for therapeutic intervention. In cancer, inhibiting TRIM21's K63-specific activity toward PD-1 could potentially enhance T cell function and overcome resistance to immune checkpoint blockade [82]. Conversely, modulating TRIM21's K27-linked ubiquitination activity might provide avenues for treating autoimmune conditions or enhancing antiviral responses. The development of linkage-specific inhibitors would represent a major advancement in the precision targeting of ubiquitin pathways. Emerging technologies such as proteolysis-targeting chimeras (PROTACs) could leverage the endogenous ubiquitin machinery to target pathogenic proteins for degradation, potentially exploiting specific ubiquitin linkages for enhanced specificity [84] [81].
Future research should focus on elucidating the structural determinants governing TRIM21's linkage specificity, identifying the specific E2 enzymes that collaborate with TRIM21 to form K27 linkages, and developing more sophisticated tools for monitoring these specific ubiquitination events in living cells. As our understanding of the ubiquitin code deepens, the therapeutic manipulation of specific linkage types offers exciting possibilities for treating cancer, autoimmune diseases, and viral infections with unprecedented precision.
Ubiquitination, a crucial post-translational modification, regulates myriad cellular processes, with different ubiquitin chain linkages conferring distinct functional outcomes. Among these, K63-linked chains have been extensively characterized as key regulators of immune signaling pathways, while K27-linked chains represent a more enigmatic linkage with emerging roles in immune regulation. The differential druggability of the enzymatic machinery governing these chains—including E2 conjugating enzymes, E3 ligases, and deubiquitinases (DUBs)—presents unique opportunities and challenges for therapeutic development. This comparison guide objectively evaluates the current landscape of K63 versus K27 enzymatic machinery as drug targets, focusing on structural characteristics, established biological functions, and the availability of experimental tools for target validation and therapeutic modulation.
Table 1: Fundamental Characteristics of K63 vs K27 Linked Ubiquitin Chains
| Characteristic | K63-Linked Chains | K27-Linked Chains |
|---|---|---|
| Primary Functions | Signal transduction, DNA repair, protein trafficking, immune activation [4] [27] | Mitochondrial quality control, immune regulation (emerging roles) [19] |
| Structural Features | Extended, open conformations | Compact, closed conformations (predicted) |
| Proteasomal Degradation | Generally non-proteolytic [27] | Non-protelytic (Miro1 stabilization) [19] |
| Chain Recognition | Specific UBDs in signaling complexes | Poorly characterized recognition mechanisms |
| Therapeutic Validation | Established in preclinical models | Limited direct validation |
Enzymatic Machinery and Target Validation
K63-Linked Ubiquitination Machinery
The synthesis of K63-linked ubiquitin chains is primarily mediated by the unique E2 enzyme complex Ubc13-Uev1A, which collaborates with specific E3 ligases such as TRAF6, TRAF2, and cIAP1/2 to conjugate K63-linked chains onto target proteins in immune signaling pathways [4] [85]. This enzymatic cascade has been rigorously validated through genetic and pharmacological studies. For instance, Ubc13 deficiency impairs NF-κB and MAPK activation downstream of multiple immune receptors, including TLRs, IL-1R, and antigen receptors [4]. Small molecule inhibitors targeting Ubc13 have shown efficacy in preclinical models of inflammatory diseases, demonstrating the druggability of this enzymatic complex [8].
The K63-specific deubiquitinases A20 (TNFAIP3) and CYLD provide negative regulation of this pathway, with their genetic ablation resulting in hyperinflammatory phenotypes and increased susceptibility to autoimmune pathologies [4] [86]. These DUBs have emerged as attractive therapeutic targets, with drug discovery efforts focused on developing activators of their enzymatic activity to suppress aberrant immune signaling.
K27-Linked Ubiquitination Machinery
In stark contrast to the well-defined K63 machinery, the enzymatic components responsible for K27-linked ubiquitination remain poorly characterized. No dedicated E2-E3 complex has been definitively identified for K27 chain formation, representing a significant knowledge gap in the field. Cellular studies indicate that K27 linkages are synthesized by unknown E2-E3 pairs that remain to be fully elucidated [19].
The most distinctive feature of K27-linked ubiquitin chains is their remarkable resistance to deubiquitination. Screening against multiple DUB families revealed that K27-Ub2 resists cleavage by most deubiquitinases, including linkage-nonspecific enzymes such as USP2, USP5, and Ubp6 that efficiently cleave other ubiquitin linkages [19]. This unique biochemical property suggests that K27 chains may function as stable signaling platforms within cells, potentially offering advantages for therapeutic targeting through stabilization rather than inhibition.
Table 2: Enzymatic Machinery for K63 vs K27 Linked Ubiquitination
| Enzymatic Component | K63-Linked Chains | K27-Linked Chains |
|---|---|---|
| E2 Enzymes | Ubc13 (with Uev1A cofactor) [4] | Not identified |
| E3 Ligases | TRAF6, TRAF2, cIAP1/2 [4] [85] | Not fully characterized |
| Regulatory DUBs | A20, CYLD, OTUD7B [4] [86] | Highly resistant to most DUBs [19] |
| Validation Tools | siRNA, small molecule inhibitors, knockout models | Limited pharmacological tools |
| Therapeutic Modulation | Inhibitors in preclinical development | No direct modulators reported |
Structural and Functional Insights
Structural Characterization and Dynamics
Comprehensive structural studies using NMR spectroscopy and small-angle neutron scattering have revealed that K27-Ub2 exhibits minimal noncovalent interdomain contacts, with the distal ubiquitin unit showing the smallest chemical shift perturbations among all ubiquitin linkages [19]. This structural feature may contribute to its unique biochemical behavior and resistance to deubiquitinases.
K27-Ub2 demonstrates unexpected binding preferences, specifically interacting with the K48-selective UBA2 domain from the proteasomal shuttle protein hHR23a, highlighting the structural versatility and potential cross-reactivity of polyubiquitin chains with different receptor proteins [19]. In contrast, K63-linked chains adopt more defined conformations that are specifically recognized by ubiquitin-binding domains in proteins involved in NF-κB signaling, DNA repair, and other signaling pathways [4] [14].
Immune Signaling Pathways
K63-linked ubiquitination serves as a master regulator of innate and adaptive immune signaling. It controls signal transduction downstream of pattern recognition receptors (TLRs, RLRs, NLRs), antigen receptors (TCR, BCR), and cytokine receptors (TNFR, IL-1R) [4] [85] [14]. Key substrates include RIP1, RIP2, TRAF6, NEMO, and AKT, where K63 chains facilitate the assembly of signaling complexes and activation of downstream kinases.
The roles of K27-linked ubiquitination in immune signaling are less defined but emerging. K27 chains are implicated in the regulation of mitochondrial trafficking protein Miro1, potentially linking them to mitochondrial quality control during immune responses [19]. Additionally, K27 linkages have been associated with regulation of innate immunity pathways, though the specific mechanisms remain incompletely characterized [19].
Figure 1: Comparative Immune Signaling Pathways of K63 and K27 Ubiquitin Chains. K63 linkages are installed by defined E3 ligases (TRAF6, TRAF2, cIAP1/2) downstream of specific immune receptors and activate well-characterized signaling pathways. K27 linkages are formed by unknown E3 ligases and their signaling outcomes are less defined.
Experimental Approaches and Research Tools
Methodologies for Studying Linkage-Specific Ubiquitination
Advanced methodologies have been developed to investigate linkage-specific ubiquitination events in cellular contexts. Tandem Ubiquitin Binding Entities (TUBEs) serve as powerful tools for capturing and quantifying specific ubiquitin linkages with nanomolar affinities [8]. These specialized affinity matrices enable researchers to distinguish between different ubiquitin chain types in response to physiological stimuli or pharmacological interventions.
The experimental workflow for assessing linkage-specific ubiquitination involves:
- Cell stimulation with specific agonists (e.g., L18-MDP for K63 ubiquitination of RIPK2) or treatment with PROTAC molecules to induce K48-linked ubiquitination
- Cell lysis under conditions that preserve labile ubiquitin modifications
- Linkage-specific capture using chain-selective TUBEs (K63-TUBE, K48-TUBE, or pan-TUBE) immobilized in multiwell plates
- Immunoblotting with target-specific antibodies to quantify captured ubiquitinated proteins
- Data analysis to determine stimulus-dependent or context-dependent ubiquitination patterns [8]
This approach has been successfully applied to demonstrate that inflammatory agent L18-MDP stimulates K63 ubiquitination of RIPK2, which is captured by K63-TUBEs but not K48-TUBEs, while RIPK2 PROTAC-mediated ubiquitination is captured by K48-TUBEs but not K63-TUBEs [8].
The Scientist's Toolkit: Key Research Reagents
Table 3: Essential Research Tools for Studying K63 and K27 Ubiquitination
| Research Tool | Specific Application | Utility in Target Validation |
|---|---|---|
| Chain-Selective TUBEs | Enrichment of specific ubiquitin linkages from cell lysates [8] | High-throughput screening for linkage-specific ubiquitination events |
| DUB Inhibitors (PR619) | Pan-DUB inhibition to study ubiquitin dynamics [58] | Assessing DUB sensitivity and turnover rates of different ubiquitin linkages |
| Proteasome Inhibitors (MG132, Bortezomib) | Blockade of proteasomal degradation [58] | Distinguishing proteolytic vs. non-proteolytic ubiquitin signals |
| Ubc13 Inhibitors | Specific blockade of K63-linked chain formation [8] | Validation of K63-dependent cellular phenotypes |
| Linkage-Specific Antibodies | Detection of endogenous K63 or K27 chains by immunoblotting | Monitoring chain accumulation in response to pathway modulation |
Figure 2: Experimental Workflow for Linkage-Specific Ubiquitination Analysis Using TUBE Technology. Cellular stimuli induce specific ubiquitination events that can be captured using linkage-selective TUBEs and quantified to determine pathway-specific regulation.
Comparative Druggability and Therapeutic Outlook
Current Therapeutic Landscape
The K63 ubiquitination machinery presents a more advanced but complex therapeutic landscape. Multiple components of the K63 pathway have been targeted pharmacologically, including:
- Ubc13 inhibitors that block K63 chain formation and show efficacy in preclinical inflammation models [8]
- DUB modulators targeting A20 and CYLD to enhance negative feedback in inflammatory pathways [86]
- TRAF6 inhibitors that disrupt E3 ligase activity and downstream NF-κB signaling
In contrast, therapeutic targeting of K27 linkages remains largely unexplored due to fundamental knowledge gaps in its enzymatic machinery and clearer pathological roles. The unique resistance of K27 chains to deubiquitinases suggests potential alternative targeting strategies focused on stabilizing rather than inhibiting these modifications once their physiological functions are better understood.
Challenges and Future Directions
Major challenges persist in targeting both ubiquitination pathways. For K63, the pleiotropic nature of its signaling functions raises concerns about on-target toxicity, requiring sophisticated targeting approaches such as tissue-specific delivery or context-dependent inhibition. For K27, the primary challenge remains basic target validation, including identification of the responsible E2-E3 pairs and clarification of its physiological functions in immune regulation.
Future research directions should prioritize:
- Comprehensive identification of the enzymatic machinery for K27 chain assembly
- Development of chemical probes specifically modulating K27 linkages
- Structural characterization of K27 chains in complex with potential receptors
- Animal models with targeted disruptions of K27 ubiquitination
- Exploration of branched ubiquitin chains containing K27 linkages and their functional implications [6]
The continued development of sophisticated tools for monitoring linkage-specific ubiquitination, such as the TUBE-based platforms and UbiSite technologies, will be essential for advancing both basic understanding and therapeutic targeting of these complex post-translational regulatory systems [8] [58].
Conclusion
K63 and K27-linked ubiquitin chains, while both central to non-proteolytic signaling, are functionally non-redundant regulators of immunity. K63 chains are established drivers of pro-inflammatory NF-κB and interferon responses, whereas emerging research confirms K27's unique role in fine-tuning these pathways, exemplified by its regulation of TRIF in TLR3/4 signaling. Future research must leverage advanced structural and chemical biology to develop precise linkage-specific tools and therapeutics. Targeting the enzymes that write, erase, or read these codes—such as Ubc13, TRAF6, or USP19—holds immense promise for novel immunotherapies in oncology, autoimmune disorders, and infectious diseases, paving the way for a new class of drugs that modulate the ubiquitin code.
References
- 1. Major histocompatibility class II in murine antigen ... [https://www.nature.com/articles/s41598-025-25817-4]
- 2. K48- and K63-linked ubiquitin chain interactome reveals ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11109483/]
- 3. K48- and K63-linked ubiquitin chain interactome reveals ... [https://www.life-science-alliance.org/content/7/8/e202402740]
- 4. K63 ubiquitination in immune signaling - PMC - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC8755460/]
- 5. The Role of Atypical Ubiquitin Chains in the Regulation ... [https://www.frontiersin.org/journals/cell-and-developmental-biology/articles/10.3389/fcell.2019.00392/full]
- 6. Emerging functions of branched ubiquitin chains [https://www.nature.com/articles/s41421-020-00237-y]
- 7. Beyond K48 and K63: non-canonical protein ubiquitination [https://cmbl.biomedcentral.com/articles/10.1186/s11658-020-00245-6]
- 8. Unraveling chain specific ubiquitination in cells using tandem ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12217038/]
- 9. Opposing regulation of the K63-linked polyubiquitination ... [https://www.nature.com/articles/s41467-025-62723-9]
- 10. Unraveling Chain specific Ubiquitination in Cells Using ... [https://lifesensors.com/unraveling-chain-specific-ubiquitination-in-cells-using-tandem-ubiquitin-binding-entities-in-a-high-throughput-assay/]
- 11. Site-specific K63 ubiquitinomics provides insights into ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6465141/]
- 12. Linkage via K27 Bestows Ubiquitin Chains with Unique ... [https://www.sciencedirect.com/science/article/pii/S0969212616000174]
- 13. The Role of Protein Ubiquitination in the Onset and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12248740/]
- 14. Ubiquitin signaling in immune responses | Cell Research [https://www.nature.com/articles/cr201640]
- 15. Engineering the orthogonal ubiquitin E1-E2 pairs for ... [https://www.sciencedirect.com/science/article/abs/pii/S0141813024086471]
- 16. Unraveling chain specific ubiquitination in cells using ... [https://www.nature.com/articles/s41598-025-07242-9]
- 17. Review K63 ubiquitination in immune signaling [https://www.sciencedirect.com/science/article/abs/pii/S1471490621002635]
- 18. Deubiquitinating enzymes and the proteasome regulate ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9117253/]
- 19. Linkage via K27 bestows ubiquitin chains with unique ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4787624/]
- 20. Ubc13: the Lys63 ubiquitin chain building machine [https://www.oncotarget.com/article/10948/text/]
- 21. E3 ubiquitin ligases: styles, structures and functions - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC8607428/]
- 22. Structural basis for the RING catalyzed synthesis of K63 ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4529489/]
- 23. Crystal Structure of the Mms2/Ubc13 Heterodimer [https://www.sciencedirect.com/science/article/pii/S0092867401003877]
- 24. Broad and diverse mechanisms used by deubiquitinase family ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5931765/]
- 25. CYLD, A20 and OTULIN deubiquitinases in NF-κB ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5520167/]
- 26. Article The Structure of the CYLD USP Domain Explains Its ... [https://www.sciencedirect.com/science/article/pii/S1097276508000154]
- 27. Role of K63-linked ubiquitination in cancer [https://www.nature.com/articles/s41420-022-01204-0]
- 28. K27‐linked ubiquitylation promotes p97 substrate processing ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9058539/]
- 29. Discovery and mechanism of K63-linkage-directed ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12037411/]
- 30. K27-linked ubiquitination of BRAF by ITCH engages ... [https://www.nature.com/articles/s41467-019-09844-0]
- 31. Deubiquitinase–based analysis of ubiquitin chain architecture using Ubiquitin Chain Restriction (UbiCRest) - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC5011418/]
- 32. Current methodologies in protein ubiquitination ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9373315/]
- 33. The Role of Atypical Ubiquitin Chains in the Regulation ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6987411/]
- 34. Deubiquitinase-based analysis of ubiquitin chain architecture using Ubiquitin Chain Restriction (UbiCRest) | Springer Nature Experiments [https://experiments.springernature.com/articles/10.1038/nprot.2015.018?code=654d73ce-5672-4632-bd9c-01e92d2d3cf0&error=cookies_not_supported]
- 35. Computationally Driven Top-Down Mass Spectrometry of ... [https://pubmed.ncbi.nlm.nih.gov/40766653/]
- 36. Advancements in the application of reporter gene cell lines ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12460214/]
- 37. The role of cGAS-STING pathway ubiquitination in innate ... [https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2025.1522200/full]
- 38. Reporter gene comparison demonstrates interference of ... [https://www.nature.com/articles/s41598-020-80451-6]
- 39. Two novel red-FRET ERK bosensors in the 670-720 nm range [https://jbioleng.biomedcentral.com/articles/10.1186/s13036-025-00541-9]
- 40. IRF3 in viral infections: more than just triggering the ... [https://pubmed.ncbi.nlm.nih.gov/40897839/]
- 41. TOB1 attenuates IRF3-directed antiviral responses by ... [https://www.nature.com/articles/s42003-022-03911-x]
- 42. Chemogenetic tuning reveals optimal MAPK signaling for ... [https://www.sciencedirect.com/science/article/pii/S2211124725009970]
- 43. K27-linked noncanonic ubiquitination in immune regulation [https://pubmed.ncbi.nlm.nih.gov/33857334/]
- 44. USP19 modulates autophagy and antiviral immune responses ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4972138/]
- 45. USP19 (ubiquitin specific peptidase 19) promotes TBK1 ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9037486/]
- 46. NEMO Family of Proteins as Polyubiquitin Receptors [https://pmc.ncbi.nlm.nih.gov/articles/PMC11854354/]
- 47. The Functional Deubiquitinating Enzymes in Control of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7816709/]
- 48. The role of ubiquitination in tumorigenesis and targeted ... [https://www.nature.com/articles/s41392-020-0107-0]
- 49. Updated insights into the molecular networks for NLRP3 ... [https://www.nature.com/articles/s41423-025-01284-9]
- 50. Ubiquitin-Mediated Regulation of RIPK1 Kinase Activity ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5823975/]
- 51. The Two Faces of Receptor Interacting Protein Kinase-1 - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC4254517/]
- 52. Necroptosis in cancer: insight from epigenetic, post ... [https://jhoonline.biomedcentral.com/articles/10.1186/s13045-025-01726-x]
- 53. An Integrated View of Deubiquitinating Enzymes Involved ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8542838/]
- 54. Regulation of Cellular Antiviral Signaling by Modifications ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5833123/]
- 55. Structural basis of K11/K48-branched ubiquitin chain ... [https://www.nature.com/articles/s41467-025-64719-x]
- 56. Ubiquitination in the Antiviral Immune Response - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC4774549/]
- 57. K27-Linked Diubiquitin Inhibits UCHL3 via an Unusual ... [https://www.sciencedirect.com/science/article/pii/S2451945620304347]
- 58. Deubiquitinating enzymes and the proteasome regulate ... [https://www.nature.com/articles/s41467-022-30376-7]
- 59. Combinatorial ubiquitin code degrades deubiquitylation- ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11933340/]
- 60. Non-proteolytic ubiquitylation in cellular signaling and human ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8826416/]
- 61. The K48-K63 Branched Ubiquitin Chain Regulates NF-κB ... [https://www.sciencedirect.com/science/article/pii/S1097276516305639]
- 62. Major histocompatibility class II in murine antigen... | Research Square [https://www.researchsquare.com/article/rs-6763007/v1]
- 63. Inhibition of K63 ubiquitination by G-Protein pathway ... [https://www.sciencedirect.com/science/article/pii/S1043661824002810]
- 64. K63 ubiquitination in immune signaling [https://pubmed.ncbi.nlm.nih.gov/35033428/]
- 65. Mechanisms of Antiviral Immune Evasion of SARS-CoV-2 - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC8457632/]
- 66. Innate immune evasion strategies of SARS-CoV-2 [https://www.nature.com/articles/s41579-022-00839-1]
- 67. TRIM25 potentiates innate immune response to ... [https://pubmed.ncbi.nlm.nih.gov/40651151/]
- 68. TRIM25 potentiates innate immune response to ... [https://www.sciencedirect.com/science/article/abs/pii/S037811352500269X]
- 69. Lys63-linked ubiquitin chain adopts multiple conformational ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4507786/]
- 70. Conformational Dynamics and Structural Plasticity Play ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2854419/]
- 71. Regulation of antiviral innate immune signaling and viral ... [https://www.nature.com/articles/s12276-021-00691-y]
- 72. Exploring the role of mitochondrial antiviral signaling protein ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11876050/]
- 73. STING-ΔN, a novel splice isoform of STING, modulates innate ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12181837/]
- 74. ZC3HAV1 facilitates STING activation and enhances ... [https://www.nature.com/articles/s42003-024-07116-2]
- 75. T cell receptor (TCR) signaling in health and disease [https://www.nature.com/articles/s41392-021-00823-w]
- 76. T cell Receptor Signal Transduction in T lymphocytes - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC3740441/]
- 77. Ubiquitin signaling in immune responses - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC4822134/]
- 78. Gut microbiota-derived metabolites modulate Treg/Th17 ... [https://pubmed.ncbi.nlm.nih.gov/41293163/]
- 79. Short-chain fatty acids from gut microbiota restore Th17 ... [https://www.sciencedirect.com/science/article/pii/S2589909025000516]
- 80. The role of regulatory T cells in preventing ... [https://pubmed.ncbi.nlm.nih.gov/40567182/]
- 81. Ubiquitination Enzymes in Cancer, Cancer Immune ... [https://www.mdpi.com/2073-4409/14/2/69]
- 82. Targeting the TRIM21-PD-1 axis potentiates immune ... [https://www.sciencedirect.com/science/article/abs/pii/S1525001625000863]
- 83. Discovery and mechanism of K63-linkage-directed ... [https://www.nature.com/articles/s41589-024-01777-0]
- 84. Ubiquitination in cancer: mechanisms and therapeutic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12479132/]
- 85. The role of ubiquitylation in immune defence and pathogen ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3864900/]
- 86. Deubiquitylating enzymes and drug discovery - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC7097658/]