K6-Linked Ubiquitination: Emerging Roles in DNA Damage Response and Therapeutic Targeting
This article synthesizes current knowledge on the non-degradative roles of lysine 6 (K6)-linked ubiquitin chains in the DNA damage response (DDR).
K6-Linked Ubiquitination: Emerging Roles in DNA Damage Response and Therapeutic Targeting
Abstract
This article synthesizes current knowledge on the non-degradative roles of lysine 6 (K6)-linked ubiquitin chains in the DNA damage response (DDR). While historically less characterized than K48 or K63 linkages, recent research reveals K6 ubiquitination as a critical signal in managing specific DNA lesions and associated RNA damage. We explore its foundational mechanisms, including the identification of the E3 ligase RNF14 in resolving formaldehyde-induced RNA-protein crosslinks (RPCs) and its interplay with other DDR pathways. The content details advanced methodologies for studying K6 chains, addresses common research challenges, and validates its functions through comparative analysis with other ubiquitin linkages. Aimed at researchers and drug development professionals, this review highlights the potential of targeting K6 ubiquitination for novel cancer therapeutics and biomarkers.
Unraveling the Basics: K6 Ubiquitin Linkages in Genome Integrity
Ubiquitin and the Expanding Ubiquitin Code
Protein ubiquitylation is a crucial post-translational modification that regulates virtually every aspect of cellular biology, including the DNA damage response (DDR) [1]. The process involves a sequential enzymatic cascade whereby ubiquitin—a 76-amino acid protein—is activated by an E1 enzyme, transferred to an E2 conjugating enzyme, and finally attached to substrate proteins via E3 ubiquitin ligases [1]. The remarkable functional diversity of ubiquitylation stems from the ability of ubiquitin itself to form various polymer chains through its seven lysine residues (K6, K11, K27, K29, K33, K48, K63) or N-terminal methionine (M1) [1] [2].
For decades, the primary focus remained on K48-linked polyubiquitin chains that target proteins for proteasomal degradation and K63-linked chains involved in signaling pathways [1] [2]. However, recent research has revealed the existence and importance of "atypical" ubiquitin chains, including those linked through K6, K11, K27, K29, and K33, which play specialized regulatory roles in cellular processes [2] [3]. These atypical linkages expand the "ubiquitin code" and offer sophisticated mechanisms for fine-tuning cellular pathways, particularly in the response to genotoxic stress [1] [4].
Table 1: Ubiquitin Chain Linkages and Their Primary Functions
| Linkage Type | Primary Functions | Representative Roles in DNA Damage Response |
|---|---|---|
| K48-linked | Proteasomal degradation [1] | Termination of DDR signaling; removal of repair factors [1] |
| K63-linked | Signal transduction; DNA repair [1] [2] | Recruitment of repair machinery to damage sites [1] |
| K6-linked | DNA repair [1] [4]; stress response [5] | UV-induced DNA damage response [4] [6] |
| K11-linked | Cell cycle regulation [1] | Cell cycle-dependent degradation |
| K27/K29-linked | Lysosomal degradation [1]; signaling | Less characterized in DDR |
| M1-linked (linear) | NF-κB signaling; immunity [2] | Innate immune response to damage |
K6-Linked Ubiquitin Chains: An Emerging Atypical Player in DNA Damage Response
Among the atypical ubiquitin chains, K6-linked ubiquitylation has recently emerged as a significant regulator of the cellular response to DNA damage. Although all lysine linkages are detectable in yeast and mammalian cells, K6-linkages have been indirectly linked to DNA repair processes since the BRCA1/BARD1 ubiquitin ligase complex was found to assemble K6-linked polyubiquitin on itself and its substrates [7].
Research has demonstrated that K6-linked ubiquitylation is specifically induced by ultraviolet (UV) light radiation but not by other types of DNA damage such as double-strand breaks, crosslinks, or replication stress [4]. This specificity suggests a unique role for K6-linked chains in the cellular response to UV-induced lesions. Quantitative proteomic studies have confirmed a 1.5-fold UV-induced increase in K6-linked ubiquitin chains, while the abundance of other linkage types remained largely unaffected [6].
Table 2: Experimental Evidence for K6-Linked Ubiquitylation in DNA Damage Response
| Experimental System | Key Finding | Methodology | Reference |
|---|---|---|---|
| Human cells (UV irradiation) | K6-linked ubiquitylation increases 1.5-fold after UV damage | Quantitative di-Gly proteomics (LC-MS/MS) | [6] |
| Histone H1 ubiquitylation | HUWE1 mediates UV-dependent histone H1 ubiquitylation at multiple lysines | SILAC-based quantitative proteomics; immunopurification of di-Gly peptides | [6] |
| BRCA1/BARD1 complex | Assembles K6-linked polyubiquitin on itself and substrates | In vitro ubiquitylation assays with single-lysine ubiquitin mutants | [7] |
| Formaldehyde-induced stress | K6-linked ubiquitylation marks RNA-protein crosslinks for resolution | Quantitative proteomics; ribosome profiling; ubiquitin linkage mapping | [5] |
Methodologies for Studying K6-Linked Ubiquitin Chains
Biochemical and Proteomic Approaches
The investigation of K6-linked ubiquitin chains relies on specialized biochemical and mass spectrometry-based techniques:
Linkage-Specific Deubiquitinase (DUB) Restriction Analysis: This approach uses DUBs with defined linkage preferences as "ubiquitin chain restriction enzymes" to characterize chain architecture. OTUD3 has been identified as a DUB with strong preference for K6-linkages, while OTUB1 specifically cleaves K48-linkages [7]. Sequential treatment with these enzymes allows dissection of heterotypic chains containing both K6 and K48 linkages.
Quantitative Di-Gly Proteomics: This method involves tryptic digestion of ubiquitylated proteins, which generates peptides containing a Lys-ϵ-Gly-Gly (di-Gly) remnant—the two C-terminal glycine residues of ubiquitin covalently attached to modified lysines. These peptides are enriched using di-Gly-specific antibodies and analyzed by liquid chromatography-tandem mass spectrometry (LC-MS/MS) [6]. Stable isotope labeling with amino acids in cell culture (SILAC) enables quantitative comparison of ubiquitylation sites between different conditions.
Experimental Protocol: Di-Gly Proteomics for K6-Linkage Detection
- Culture two populations of U2OS cells in light (K0R0) and heavy (K6R10) SILAC media
- Treat heavy-labeled cells with UV irradiation (16 J/m²); keep light-labeled cells as mock control
- Harvest cells and combine populations in 1:1 ratio
- Perform protein extraction and tryptic digestion
- Immunopurify di-Gly-modified peptides using anti-Lys-ϵ-Gly-Gly antibody
- Analyze peptides by LC-MS/MS using MaxQuant software for identification and quantification
- Validate specific K6-linkage increase by monitoring di-Gly-modified ubiquitin peptides at K6 position
Enzymatic Tools for K6-Linked Chain Synthesis
The bacterial effector E3 ligase NleL from enterohaemorrhagic Escherichia coli O157:H7 has been exploited to generate large quantities of K6-linked ubiquitin chains in vitro [7]. NleL assembles both K6- and K48-linked ubiquitin polymers, with preference for generating heterotypic chains containing both linkage types. This enzyme has proven invaluable for biochemical and structural studies of K6-linked chains.
Diagram 1: Enzymatic Cascade for K6-Linked Ubiquitin Chain Assembly
Molecular Functions of K6-Linked Ubiquitylation in DNA Damage Response
Histone H1 Ubiquitylation in UV Damage Response
A prominent pathway involving K6-linked ubiquitylation in the DDR centers on histone H1. Quantitative di-Gly proteomics identified histone H1 as one of the most significantly UV-induced ubiquitylated proteins, with multiple UV-induced ubiquitylation sites across various H1 variants (H1.0, H1.1, H1.2, H1.4) [6]. This UV-dependent histone H1 ubiquitylation is mediated by the E3 ligase HUWE1, which appears to "prime" histone H1 with ubiquitin modifications that are subsequently extended by RNF8 to form K63-linked chains [6]. This priming function is critical for efficient recruitment of downstream DDR factors, as HUWE1-depleted cells show reduced recruitment of RNF168 and 53BP1 to DNA damage sites, while upstream factors like MDC1 remain unaffected [6].
Beyond Nuclear DNA Damage: Cytoplasmic Stress Responses
Recent research has revealed that K6-linked ubiquitylation also functions in resolving formaldehyde-induced RNA-protein crosslinks (RPCs) in the cytoplasm [5]. Formaldehyde, produced by cellular metabolism or alcohol consumption, induces RPCs that stall ribosomes and inhibit translation. These RPCs are marked by atypical K6-linked ubiquitylation catalyzed by the RING-in-between-RING (RBR) E3 ligase RNF14 and subsequently resolved by the ubiquitin-dependent unfoldase VCP in a GCN1-RNF14-VCP-dependent pathway [5]. This mechanism represents an evolutionary conserved formaldehyde-induced stress response pathway that protects cells against RPC accumulation.
Diagram 2: K6-Linked Ubiquitylation in RPC Resolution Pathway
Structural Insights into K6-Linked Ubiquitin Chains
Biophysical studies of K6-linked ubiquitin chains have revealed unique structural properties that distinguish them from other linkage types. Crystallographic and NMR spectroscopy analyses show that K6-linked diubiquitin adopts a compact conformation characterized by an asymmetric interface between Ile44 and Ile36 hydrophobic patches of neighboring ubiquitin moieties [7]. This interface differs significantly from the closed conformation of K48-linked chains and the extended conformation of K63-linked chains.
In K6-linked polymers, interactions through the Ile36 patch can displace Leu8 from the Ile44 patch, leading to marked structural perturbations of ubiquitin [7]. This distinct topology likely facilitates recognition by specific ubiquitin-binding domains and contributes to the non-proteolytic functions of K6-linked chains in DNA damage signaling.
Research Reagent Solutions for K6-Linked Ubiquitylation Studies
Table 3: Essential Research Reagents for Investigating K6-Linked Ubiquitylation
| Reagent/Tool | Function/Application | Example/Source |
|---|---|---|
| Linkage-specific DUBs | Cleavage and analysis of specific ubiquitin linkages | OTUD3 (K6-specific); OTUB1 (K48-specific) [7] |
| Single-lysine ubiquitin mutants | Determining linkage specificity of E3 ligases | Ub K6R; Ub K48R; Ub K6R/K48R [7] |
| Bacterial E3 ligase NleL | Enzymatic synthesis of K6-linked ubiquitin chains | Enterohaemorrhagic E. coli O157:H7 [7] |
| Di-Gly remnant antibody | Immunoaffinity enrichment of ubiquitylated peptides | Commercial anti-Lys-ϵ-Gly-Gly antibodies [6] |
| E1 and E2 enzymes | In vitro ubiquitylation assays | UBE1 (E1); UBE2L3/UbcH7 (E2) [7] |
| Mass spectrometry standards | Quantitative proteomics | SILAC labeling reagents (K0R0, K6R10) [6] |
Future Perspectives and Therapeutic Implications
The investigation of K6-linked ubiquitin chains in the DNA damage response represents an emerging field with significant potential for therapeutic development. As atypical ubiquitin chains are increasingly recognized as important regulatory modifications, understanding their specific functions in genome maintenance may reveal new targets for cancer therapy, particularly in tumors with defects in DNA repair pathways.
Current research efforts focus on identifying the complete repertoire of E3 ligases that assemble K6-linked chains, the specific readers that recognize this modification, and the full spectrum of biological processes regulated by this atypical ubiquitin linkage. The development of chemical probes that specifically target K6-linked ubiquitylation events may offer new avenues for modulating the DNA damage response in human diseases.
K6-linked ubiquitin chains represent one of the more enigmatic forms of ubiquitination, increasingly recognized for their specialized roles in cellular stress response pathways, particularly the DNA damage response (DDR). Unlike the well-characterized K48 and K63 linkages, K6-linked ubiquitination employs a dedicated enzymatic cascade that orchestrates critical functions in genome maintenance. This technical guide comprehensively details the specific E1 activating enzymes, E2 conjugating enzymes, E3 ligases, and deubiquitinating enzymes that govern K6 ubiquitination dynamics. We synthesize current mechanistic insights into K6 linkage formation and recognition, with emphasis on its emerging role in DDR pathways including DNA damage tolerance and the resolution of transcription-replication conflicts. The document provides detailed experimental methodologies for investigating K6-specific ubiquitination and catalogs essential research tools, facilitating advanced research into this atypical ubiquitin signaling pathway.
Ubiquitination is a crucial post-translational modification that regulates virtually all cellular processes in eukaryotes. The versatility of ubiquitin signaling stems from its ability to form diverse chain topologies through eight distinct linkage types: M1, K6, K11, K27, K29, K33, K48, and K63 [8] [1]. Among these, K6-linked ubiquitin chains have remained one of the least characterized, often referred to as 'atypical' ubiquitin linkages [8]. However, emerging research has established that K6-linked ubiquitination serves as a critical signaling mechanism in the cellular response to genotoxic stress, playing specialized roles in DNA damage repair and maintenance of genomic integrity [8] [1] [9].
The K6 linkage is formed through an isopeptide bond between the C-terminal glycine of one ubiquitin molecule and the lysine at position 6 (K6) of another ubiquitin molecule [8]. Early evidence for K6 ubiquitination emerged from studies of DNA damage response, where K6-linked chains were found to accumulate at sites of DNA damage [1]. More recent work has demonstrated that K6 ubiquitination participates in a ubiquitin-mediated degradation system for resolving formaldehyde-induced RNA-protein crosslinks (RPCs), which are toxic when they block protein synthesis [10]. This pathway represents a specialized branch of the DNA damage response that protects against aldehyde-induced genotoxicity.
Table 1: Key Characteristics of K6-Linked Ubiquitin Chains
| Property | Characteristics | Biological Context |
|---|---|---|
| Chain Structure | Atypical linkage topology | Less characterized than K48/K63 |
| Primary Functions | DNA damage response, protein degradation | RPC resolution, proteasomal targeting |
| Cellular Abundance | Lower abundance compared to K48/K63 | Induced under genotoxic stress |
| Chain Recognition | Specific UBD-containing proteins | p97/VCP recruitment |
| Regulatory Complexity | Forms homotypic and branched chains | Can be combined with K48 in branched chains |
The K6-Specific Enzymatic Cascade
The formation of K6-linked ubiquitin chains follows the canonical three-step enzymatic cascade but employs specific enzyme combinations that determine linkage specificity.
E1 Activating Enzymes
The ubiquitination cascade initiates with a single E1 ubiquitin-activating enzyme, which primes ubiquitin for transfer through an ATP-dependent reaction. The E1 enzyme forms a thioester bond between its active-site cysteine and the C-terminal glycine of ubiquitin, creating a high-energy E1~Ub intermediate [8] [9]. This E1~Ub complex then engages with specific E2 conjugating enzymes. While the E1 enzyme is shared across all ubiquitination types, its interaction with specific E2 partners represents the first branching point toward K6 linkage specificity.
E2 Conjugating Enzymes
E2 conjugating enzymes serve as the central determinants of ubiquitin chain linkage specificity. These enzymes receive activated ubiquitin from E1 via transthiolation and collaborate with E3 ligases to dictate the topology of the resulting ubiquitin chain [11]. For K6-linked ubiquitination, the E2 enzyme UBE2A (RAD6A) has been implicated in DNA damage-associated K6 chain formation [1]. UBE2A functions in conjunction with the E3 ligase BRCA1 as part of the BRCA1-A macromolecular complex, which accumulates at DNA double-strand breaks and facilitates the DNA damage response through K6-linked ubiquitination [9]. The specific pairing of UBE2A with K6-directed E3 ligases ensures the faithful formation of K6 isopeptide linkages during the ubiquitin transfer process.
E3 Ligases for K6 Linkage
E3 ubiquitin ligases provide substrate specificity and directly catalyze ubiquitin transfer to target proteins. Several E3 ligases have been identified as capable of synthesizing K6-linked ubiquitin chains:
BRCA1-BARD1 Complex: The BRCA1-BARD1 heterodimer represents a well-characterized E3 ligase complex that generates K6-linked ubiquitin chains both in vitro and in vivo [9]. This RING-type E3 ligase complex plays an essential role in DNA damage repair, particularly in the homologous recombination pathway. BRCA1-BARD1-mediated K6 ubiquitination occurs at DNA damage sites and contributes to the recruitment of DNA repair factors.
Parkin: This RBR-type E3 ligase, often mutated in early-onset Parkinson's disease, has been demonstrated to synthesize branched K6/K48 chains [11]. While Parkin's primary functions relate to mitochondrial quality control, its ability to form K6 linkages suggests potential roles in DNA damage response, particularly given the emerging connections between mitochondrial dysfunction and genomic instability.
HUWE1: A HECT-domain E3 ligase that has been associated with K6 ubiquitination in the context of DNA damage response [8]. HUWE1 can collaborate with other E3 ligases to form branched ubiquitin chains containing K6 linkages, expanding the regulatory potential of this modification.
* Bacterial E3 Ligases*: The bacterial HECT-like E3 ligase NleL has been reported to assemble branched K6/K48 chains [11], demonstrating that K6 linkage formation is an evolutionarily conserved capability among certain E3 ligase families.
Table 2: E3 Ligases Implicated in K6-Linked Ubiquitination
| E3 Ligase | Type | Cellular Function | K6 Linkage Context |
|---|---|---|---|
| BRCA1-BARD1 | RING | DNA damage repair, homologous recombination | Primary linkage in DNA damage response |
| Parkin | RBR | Mitochondrial quality control, mitophagy | Forms branched K6/K48 chains |
| HUWE1 | HECT | Apoptosis regulation, p53 modulation | Collaborates with TRAF6 in NF-κB signaling |
| NleL | HECT-like | Bacterial infection pathogenesis | Branched K6/K48 chain formation in vitro |
Deubiquitinases for K6 Linkage
Deubiquitinating enzymes (DUBs) counterbalance ubiquitin ligase activity by removing ubiquitin chains, thereby providing temporal control over ubiquitin signaling. The identity of K6-specific DUBs remains an active area of investigation, but several DUB families have demonstrated activity toward K6 linkages:
USP Family DUBs: Certain ubiquitin-specific proteases exhibit broad linkage specificity and may process K6-linked chains. Their ability to recognize multiple linkage types suggests they might serve as general regulators of ubiquitin signaling homeostasis rather than K6-specific editors.
OTU Family DUBs: Several ovarian tumor proteases display linkage specificity and may contain K6-linkage selective members, though these remain to be fully characterized in the context of DNA damage response.
The identification of DUBs with precise specificity for K6 linkages would provide important insights into the regulation of this modification pathway and represents a critical gap in our current understanding of K6 ubiquitination dynamics.
K6 Linkages in DNA Damage Response
K6-linked ubiquitination has emerged as a significant regulator of genome maintenance pathways, with specialized functions in distinct DNA damage contexts.
DNA Damage Recognition and Signaling
The initial recognition of DNA damage involves the rapid accumulation of ubiquitin signaling at lesion sites, with K6 linkages contributing to these early signaling events. The BRCA1-BARD1 complex localizes to DNA double-strand breaks where it catalyzes the formation of K6-linked ubiquitin chains on surrounding histones and potentially on itself or other repair factors [9]. This K6 ubiquitination creates a platform that facilitates the assembly of DNA repair complexes, serving as a recruitment signal for additional repair factors through ubiquitin-binding domains. The presence of K6 linkages at DNA damage sites within minutes of damage induction highlights its role as an early responder in the DNA damage signaling cascade.
Resolution of Transcription-Replication Conflicts
Recent research has revealed a specialized function for K6 ubiquitination in resolving RNA-protein crosslinks (RPCs) induced by formaldehyde exposure [10]. Formaldehyde endogenously arises during metabolic processes and can induce toxic DNA-protein and RNA-protein crosslinks that block essential cellular processes. Two recent studies demonstrate that K6-linked ubiquitin chains mobilize the segregase p97/VCP and the proteasome to resolve formaldehyde-induced RPCs, representing a dedicated pathway for managing this specific type of DNA damage [10]. This pathway highlights the specialization of K6 ubiquitination in dealing with transcription-associated DNA damage that arises from aldehyde exposure.
Proteasomal Targeting via K6 Linkages
While K48-linked chains represent the canonical proteasomal degradation signal, emerging evidence suggests that K6 linkages can also target proteins for degradation, particularly in the context of DNA damage. Branched ubiquitin chains containing K6/K48 linkages have been reported to enhance proteasomal targeting efficiency compared to homotypic K48 chains [11]. In the context of DNA damage response, K6/K48-branched chains may facilitate the removal of damaged proteins or regulatory factors that must be rapidly cleared during the DNA repair process. The partnership between K6 and K48 linkages in branched chains represents an emerging paradigm for understanding how atypical ubiquitin linkages collaborate with canonical degradation signals to fine-tune protein stability.
Experimental Methods for K6 Linkage Analysis
Linkage-Specific Ubiquitin Enrichment
The analysis of K6-linked ubiquitination requires specialized tools that can distinguish this linkage type among the complex landscape of cellular ubiquitination.
Tandem Ubiquitin Binding Entities (TUBEs): Recent methodological advances have enabled the specific enrichment of linkage-defined ubiquitin chains using engineered TUBEs with linkage specificity [12]. Pan-selective TUBEs can capture all ubiquitinated proteins, while linkage-specific TUBEs (e.g., K48-TUBEs or K63-TUBEs) allow for selective isolation of particular chain types. The development of K6-specific TUBEs would represent a significant advance for the field, though current approaches typically rely on linkage-nonspecific enrichment followed by mass spectrometric analysis to identify K6 linkages.
Ubiquitin Mutants: A common experimental approach involves expression of ubiquitin mutants in which all lysine residues except K6 are mutated to arginine (Ub-K6-only) [12]. This strategy permits the selective formation of K6-linked chains when co-expressed with enzymes of interest, enabling functional studies of K6 ubiquitination without competition from other linkage types. Similarly, ubiquitin in which K6 is mutated to arginine (Ub-K6R) serves as a critical control to demonstrate K6 linkage dependence in functional assays.
Mass Spectrometric Identification
Mass spectrometry represents the gold standard for definitive identification of K6-linked ubiquitination sites. Middle-down and proteomic approaches using di-glycine remnant antibodies (e.g., K-ε-GG antibody enrichment) enable system-wide identification of ubiquitination sites, with linkage type determined by mapping the precise ubiquitination sites within ubiquitin chains themselves [12]. Advanced spectral libraries and targeted mass spectrometry methods have improved the sensitive detection of K6 linkages, though their relatively low abundance compared to K48 and K63 linkages continues to present analytical challenges.
Functional Validation Assays
In Vitro Ubiquitination Assays: Reconstruction of the K6 ubiquitination cascade using purified E1, E2, and E3 components provides definitive evidence of K6 linkage formation capability [11]. These assays typically include ATP, ubiquitin, and the relevant enzymes, with reaction products analyzed by western blotting using linkage-specific antibodies or mass spectrometry.
Cell-Based DNA Damage Sensitivity Assays: Functional validation of K6 ubiquitination in DNA damage response typically involves assessing cellular sensitivity to DNA damaging agents (e.g., formaldehyde, ionizing radiation, or crosslinking agents) following manipulation of K6 pathway components [10]. Resistance or sensitivity phenotypes in these assays provide functional readouts of K6 pathway activity.
Table 3: Experimental Approaches for K6 Linkage Analysis
| Method | Application | Key Reagents | Technical Considerations |
|---|---|---|---|
| TUBE Enrichment | Affinity purification of ubiquitinated proteins | Pan-selective or linkage-specific TUBEs | Limited commercial availability of K6-specific TUBEs |
| Ubiquitin Mutants | Selective formation of specific linkage types | Ub-K6-only, Ub-K6R mutants | May not fully replicate endogenous chain complexity |
| Mass Spectrometry | Definitive identification of linkage types | Di-glycine remnant antibodies, spectral libraries | Sensitivity challenges due to low K6 abundance |
| In Vitro Reconstitution | Direct demonstration of enzymatic activity | Purified E1, E2, E3 enzymes | May lack cellular regulatory context |
Research Reagent Solutions
The investigation of K6-linked ubiquitination requires specialized reagents designed to probe this specific pathway within the complex ubiquitin landscape.
Table 4: Essential Research Reagents for K6 Linkage Studies
| Reagent Category | Specific Examples | Research Application | Key Features |
|---|---|---|---|
| Linkage-Specific Antibodies | Anti-K6-linkage specific antibodies | Immunodetection of endogenous K6 chains | Variable commercial availability; require extensive validation |
| Ubiquitin Expression Plasmids | Ub-K6-only, Ub-K6R, Ub-noK | Selective chain formation in cellular contexts | Enable functional studies of specific linkage types |
| Recombinant Enzymes | Purified BRCA1-BARD1 complex, UBE2A | In vitro ubiquitination assays | Confirm direct enzymatic activity toward K6 linkages |
| Activity-Based Probes | K6-linkage selective DUB probes | DUB activity profiling | Identify K6-specific deubiquitinases |
| TUBE Reagents | K6-TUBEs (developmental) | Affinity enrichment of K6-ubiquitinated proteins | Highly specific isolation of K6-linked substrates |
Visualization of K6 Ubiquitination Pathway
K6 Ubiquitination in DNA Damage Response
K6 Linkage Analysis Workflow
The enzymatic cascade governing K6-linked ubiquitination represents a specialized branch of the ubiquitin-proteasome system with particular relevance to DNA damage response and genome maintenance pathways. While significant progress has been made in identifying the key E3 ligases, particularly BRCA1-BARD1 and Parkin, that generate K6 linkages, substantial knowledge gaps remain regarding the complete complement of E2 conjugating enzymes and deubiquitinating enzymes that establish and edit these chains. The development of more specific research tools, particularly K6-linkage selective TUBEs and antibodies, will accelerate our understanding of this pathway. As research continues to elucidate the precise mechanisms of K6 ubiquitination in DNA damage response, particularly in the resolution of transcription-replication conflicts, new therapeutic opportunities may emerge for targeting this pathway in diseases characterized by genomic instability, including cancer and certain neurodegenerative disorders.
Formaldehyde (FA) induces toxic RNA-protein crosslinks (RPCs) that stall translation, activating a stress response pathway where the E3 ligase RNF14 marks crosslinked proteins with atypical K6-linked ubiquitin chains. This modification targets RPCs for resolution by VCP, revealing a cytoplasmic quality control mechanism critical for managing aldehyde toxicity. This review details the molecular mechanisms, experimental methodologies, and significance of K6-linked ubiquitylation in the DNA damage response field.
Reactive aldehydes like formaldehyde are abundant endogenous metabolites produced by cellular processes such as demethylation reactions and one-carbon metabolism [13]. While aldehyde-induced DNA damage and repair mechanisms have been extensively studied, their impact on RNA has remained less explored. Formaldehyde efficiently induces covalent RNA-protein crosslinks (RPCs), which were historically considered methodological tools but are now recognized as significant lesions that contribute to cellular stress [14] [13]. These RPCs in messenger RNA (mRNA) stall elongating ribosomes, inhibit translation, and trigger collision with trailing ribosomes, ultimately activating multiple stress response pathways [13]. Recent discoveries identify RPCs as physiological substrates for the poorly understood K6-linked ubiquitin modification, positioning this atypical ubiquitination within the DNA damage response landscape as a critical quality control mechanism [14].
Molecular Mechanisms of K6-Linked Ubiquitylation in RPC Resolution
The resolution of formaldehyde-induced RPCs follows a coordinated pathway initiated when translating ribosomes encounter crosslinked mRNA-binding proteins. This ribosomal collision activates a multi-step quality control process:
- Ribosome Stalling and Collision: RPCs in mRNA stall elongating ribosomes, leading to collisions with trailing ribosomes.
- E3 Ligase Activation: Collisions activate the RING-in-between-RING (RBR) E3 ligase RNF14, and potentially RNF25.
- Atypical Ubiquitin Signaling: RNF14 catalyzes the attachment of K6- and K48-linked ubiquitin chains to the crosslinked protein adduct.
- Crosslink Resolution: The ubiquitin- and ATP-dependent unfoldase VCP (p97) recognizes and resolves the ubiquitylated RPC.
- Proteasomal Degradation: The ubiquitylated protein adduct is ultimately degraded by the proteasome [14] [13].
This pathway represents an evolutionary conserved formaldehyde-induced stress response that protects cells against RPC accumulation in the cytoplasm [14].
Visualizing the RPC Resolution Pathway
The following diagram illustrates the sequential mechanism of RPC-induced translation stress and their resolution via K6-linked ubiquitylation:
Ribosome Stalling and RNF14 Activation
When translating ribosomes encounter formaldehyde-induced RPCs, they stall and collide with trailing ribosomes. These collisions form a specific structural interface recognized by the ubiquitin ligase RNF14 [13]. RNF14, along with RNF25, belongs to a specialized pathway that responds to ribosomal stalling caused by bulky mRNA lesions. While RNF25 initially ubiquitylates the ribosomal subunit protein eS31, this modification promotes RNF14-dependent poly-ubiquitylation of the entrapped protein adduct [13]. The RBR-type E3 ligase RNF14 provides the crucial specificity for marking RPCs with atypical ubiquitin linkages.
Atypical Ubiquitin Chain Formation
RNF14 catalyzes the formation of K6-linked ubiquitin chains on proteins covalently crosslinked to RNA [14]. This atypical ubiquitin linkage is distinct from the canonical K48-linked chains that typically target substrates for proteasomal degradation. K6-linked ubiquitin chains were previously associated with DNA damage response and mitophagy but now emerge as a specific signal for RPC resolution [15] [16]. The modification likely creates a unique "ubiquitin code" that is specifically recognized by downstream effectors in the resolution pathway, particularly VCP [14].
VCP-Mediated Resolution and Proteasomal Degradation
The ubiquitin-dependent segregase VCP (also known as p97/Cdc48) recognizes the K6-ubiquitylated RPCs and utilizes ATP hydrolysis to unfold the crosslinked protein or remodel the RPC complex [14] [10]. This activity is essential for making the crosslink accessible to the final degradation step. Following VCP-mediated remodeling, the K6- and K48-linked ubiquitin chains ultimately target the crosslinked protein for degradation by the 26S proteasome, thereby clearing the lesion and restoring translational capacity [13].
Experimental Models and Methodologies for Studying RPCs
Modeling RNA Crosslinking Damage
Studying formaldehyde-induced RPCs presents challenges because FA causes pleiotropic damage including DNA-protein crosslinks (DPCs) that confound analysis. Researchers have developed specific model systems to isolate RNA damage:
PAR-CL (Photoactivatable Ribonucleoside-Enhanced Crosslinking)
- Principle: Metabolic labeling of cellular RNA with 4-thiouridine (4-SU) followed by UVA irradiation (365 nm) to induce specific RNA-protein crosslinks without DNA damage [13].
- Validation: PAR-CL induces RPCs with 78% similarity (561 of 719 proteins) to FA-induced RPCs, primarily affecting RNA-binding proteins and ribosomal constituents [13].
- Advantage: Provides a clean model for studying RNA crosslinking damage without confounding DNA lesions.
Formaldehyde Exposure
- Experimental Conditions: Typically 0.5 mM FA for 1 hour in human cell lines (HAP1, HeLa, HCT116) [13].
- Relevance: Mimics endogenous aldehyde stress while inducing complex damage including RPCs and DPCs.
Detecting and Quantifying RPCs
Several specialized techniques enable RPC detection and quantification:
XRNAX (Protein-X-Linked RNA Extraction)
- Purpose: Purifies crosslinked RNA-protein complexes under denaturing conditions [13].
- Workflow: Cell lysis in denaturing buffer, oligo(dT) pulldown of polyadenylated RNA complexes, and proteomic/transcriptomic analysis.
- Applications: Identification of crosslinked proteins via mass spectrometry and quantification of RPC burden.
KCl-SDS Precipitation Assay
- Principle: Quantifies protein-DNA and protein-RNA crosslinks based on differential solubility [13].
- Procedure: Incubate lysates with KCl and SDS, precipitate crosslinked complexes by centrifugation, and quantify pellets.
- Utility: Distinguishes between RPCs and DPCs; PAR-CL shows RPCs without DPCs.
Visualizing Experimental Workflow for RPC Analysis
The following diagram outlines the key methodological approaches for studying formaldehyde-induced RPCs:
Quantitative Analysis of RPC Formation and Stress Responses
RPC Formation Across Experimental Conditions
Table 1: Quantitative Profiling of RPC Formation Using XRNAX and Mass Spectrometry
| Experimental Condition | Total Crosslinked Proteins Identified | Common RPC Proteins | Notable Protein Categories | Crosslink Efficiency |
|---|---|---|---|---|
| Formaldehyde (0.5 mM) | 719 proteins | 561 proteins (78% overlap) | RNA-binding proteins, ribosomal structural constituents | High (similar viability impact to PAR-CL) |
| PAR-CL (5 μM 4-SU + 6 kJ/m² UVA) | Similar profile to FA | 561 shared proteins | RNA-binding proteins, ribosomal structural constituents | High (mimics FA-induced RPCs) |
| Control (No Treatment) | Baseline levels | N/A | N/A | Minimal |
Signaling Pathway Activation Metrics
Table 2: Quantitative Signaling Responses to RPC-Induced Translation Stress
| Signaling Pathway | Key Phosphorylation Events | Activation Kinetics | Genetic Requirements | Functional Consequences |
|---|---|---|---|---|
| Ribotoxic Stress Response (RSR) | p38 MAPK phosphorylation (T180/Y182) | Rapid (30-60 min post-exposure) | ZAKα kinase dependent | Cell cycle arrest, inflammation |
| Integrated Stress Response (ISR) | eIF2α phosphorylation (S51) | Concurrent with RSR | GCN2 kinase dependent | Global translation inhibition |
| Ubiquitin Signaling | K6-linked ubiquitin chain formation | Dependent on ribosome collisions | RNF14 E3 ligase essential | RPC resolution via proteasome |
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagents for Studying K6-Linked Ubiquitylation in RPC Resolution
| Reagent/Category | Specific Examples | Function/Application | Experimental Use |
|---|---|---|---|
| E3 Ligase Tools | RNF14 inhibitors/knockout | Target identification | Validate RNF14 requirement in K6-UBQ |
| Ubiquitin Probes | K6-linkage specific antibodies | Ubiquitin chain typing | Detect atypical K6 chains on RPCs |
| Crosslinking Models | 4-thiouridine (4-SU) | PAR-CL system | Induce specific RPCs without DNA damage |
| Detection Reagents | XRNAX protocol components | RPC purification | Isolate crosslinked complexes for analysis |
| Cell Models | ZAKα KO cells, RNF14 KO | Pathway dissection | Define genetic requirements |
| Chemical Inhibitors | VCP/p97 inhibitors | Block resolution phase | Confirm functional pathway |
Discussion: Implications for DNA Damage Response and Disease
K6 Ubiquitylation in the DNA Damage Response Landscape
While K6-linked ubiquitin chains were previously associated with mitochondrial quality control and DNA damage response, their role in RPC resolution represents a significant expansion of their functional repertoire [16]. In the DNA damage context, K6-linked auto-ubiquitination occurs in the BRCA1-BARD1 complex, and K6 chains form during replication stress and double-strand break repair [15] [16]. The discovery that the same atypical ubiquitin linkage resolves formaldehyde-induced RPCs suggests parallel quality control mechanisms for different types of crosslinking damage across cellular compartments.
Pathophysiological Relevance in Neurodegeneration and Cancer
Endogenous formaldehyde accumulates in the aging brain due to dysregulation of FA-metabolizing enzymes like ALDH2 and ADH5, reaching concentrations up to 300 μM in Alzheimer's disease patients [17] [13]. This accumulation correlates with cognitive decline and neurodegeneration. FA-induced RPCs likely contribute to this pathology by disrupting neuronal translation and proteostasis [17]. Similarly, in cancer biology, defects in aldehyde clearance mechanisms correlate with bone marrow failure and leukemia, suggesting RPC accumulation may contribute to genomic instability and cellular transformation [13].
Therapeutic Implications and Future Directions
The RNF14-K6 ubiquitin-VCP pathway represents a promising therapeutic target for conditions characterized by aldehyde stress. Enhancing this pathway could mitigate RPC toxicity in neurodegenerative contexts, while inhibiting it might sensitize cancer cells to aldehyde-generating chemotherapeutics. Future research should focus on:
- Developing specific modulators of RNF14 E3 ligase activity
- Engineering improved tools for quantifying K6-linked ubiquitin chains in physiological contexts
- Investigating cross-talk between RPC resolution and classical DNA damage response pathways
- Exploring tissue-specific differences in RPC susceptibility and resolution capacity
Formaldehyde-induced RPCs represent a significant yet underappreciated component of aldehyde toxicity, with dedicated resolution mechanisms centered on atypical K6-linked ubiquitylation. The RNF14-K6 ubiquitin-VCP pathway protects cells against RPC accumulation by detecting stalled translation complexes and marking crosslinked proteins for proteasomal degradation. This pathway expands the functional repertoire of non-canonical ubiquitin signaling in maintaining proteostasis and represents a promising target for therapeutic intervention in neurodegeneration, cancer, and other conditions characterized by aldehyde stress. As research methodologies advance, particularly in mass spectrometry and genome editing, our understanding of this quality control pathway will continue to illuminate its significance in cellular homeostasis and disease.
The K6-linked ubiquitin chain, one of the least understood forms of polyubiquitination, has recently emerged as a critical signal in the cellular response to specific proteotoxic stresses. This whitepaper delineates the specialized axis comprising the E3 ubiquitin ligase RNF14 and the AAA+ ATPase VCP/p97 that collaboratively targets K6-ubiquitylated substrates for resolution. Mounting evidence establishes that this RNF14-VCP pathway is indispensable for managing translation-coupled damage, particularly the resolution of formaldehyde-induced RNA-protein crosslinks (RPCs) that threaten proteostasis and cell viability. Within the broader context of DNA damage response research, understanding this non-canonical ubiquitin signaling module provides crucial insights into how cells leverage the ubiquitin code to maintain genomic and transcriptomic integrity.
Protein ubiquitylation represents a versatile post-translational modification that regulates virtually every cellular process. The diversity of ubiquitin signaling stems from the capacity of ubiquitin to form polymer chains through eight different linkage types—via its N-terminal methionine (M1) or seven internal lysine residues (K6, K11, K27, K29, K33, K48, K63). Among these, the K6-linked ubiquitin chain has remained particularly enigmatic, with its functions only beginning to be elucidated in recent years [1] [18].
K6-linked ubiquitin chains occupy a unique position in the ubiquitin landscape. Unlike the well-characterized K48-linked chains that predominantly target substrates for proteasomal degradation, or K63-linked chains that typically serve as non-proteolytic signaling devices, K6 linkages appear to function in specialized stress response pathways. Structural analyses reveal that K6-linked diubiquitin (K6-Ub2) can be recognized by specific ubiquitin-binding domains, such as the NZF domain of TAB2, which also binds K63 linkages, suggesting potential crosstalk between inflammatory signaling and damage response pathways [19]. The discovery that the E3 ligase HUWE1 generates K6-linked chains in response to mitochondrial damage further expanded the functional repertoire of this modification [18].
Within the DNA damage response (DDR) field, the identification of specific E3 ligases that generate K6 linkages and the reader proteins that interpret this signal has represented a significant knowledge gap. Recent research has now positioned the RNF14-VCP axis as a central player in this space, specifically in response to transcription- and translation-associated damage.
The RNF14-VCP Axis: Core Components and Mechanisms
RNF14: A specialized E3 ligase for translation-associated damage
RNF14 is a RING-type E3 ubiquitin ligase that has recently been identified as a key component of the cellular response to translational stress. This ligase functions within a network that includes RNF25, with both enzymes collaborating to ubiquitylate specific substrates in response to ribosome stalling [20]. The foundational study by Zhao et al. (2023) demonstrated that RNF14 is activated when ribosomes stall at covalent RNA-protein crosslinks (RPCs), which are particularly toxic lesions induced by endogenous formaldehyde and other reactive aldehydes [13].
Mechanism of activation: RNF14 is recruited to stalled ribosomes through its interaction with GCN1, a sensor of collided ribosomes. This interaction positions RNF14 to ubiquitylate the crosslinked protein adducts that are obstructing translation. The ligase specifically modifies these protein adducts with atypical ubiquitin chains containing both K6 and K48 linkages [13].
Genetic evidence: CRISPRi screening experiments have validated the essential nature of RNF14 in this pathway. Knockdown of RNF14 abrogates the degradation of crosslinked proteins and sensitizes cells to formaldehyde toxicity, confirming its non-redundant function in this stress response pathway [20].
VCP/p97: The segregase for K6-marked substrates
VCP (also known as p97 or Cdc48) is a ubiquitous AAA+ ATPase that functions as a segregase, extracting ubiquitylated proteins from cellular structures such as chromatin, membranes, and protein complexes. In the context of RPC resolution, VCP is recruited to K6/K48-ubiquitylated crosslinks to catalyze their dislodgment from RNA [10].
Mechanical extraction: VCP utilizes ATP hydrolysis to generate mechanical force that physically extracts the crosslinked protein from the RNA molecule. This activity is particularly crucial for RPCs because these crosslinks create covalent linkages that cannot be resolved through conventional enzymatic mechanisms.
Proteasome coordination: Following extraction by VCP, the ubiquitylated protein adducts are delivered to the proteasome for degradation. This coordinated action between the extraction and degradation machinery ensures complete clearance of the damaging crosslinks [10] [13].
Collaborative mechanism of the RNF14-VCP axis
The RNF14-VCP axis operates through a sequential mechanism that detects, marks, and resolves translation-blocking lesions:
- Collision detection: When a translating ribosome stalls at an RPC, a trailing ribosome collides with it, forming a disome structure.
- Sensor recruitment: The collision sensor GCN1 recognizes this disome and recruits RNF14.
- Ubiquitin conjugation: RNF14, in collaboration with RNF25, modifies the crosslinked protein with K6/K48-linked ubiquitin chains.
- Substrate extraction: VCP recognizes the K6 ubiquitin signal and extracts the ubiquitylated protein from the RNA.
- Proteasomal degradation: The extracted protein is degraded by the proteasome, while the RNA component is presumably recycled [13].
This pathway represents a translation-coupled quality control mechanism that specifically targets covalent RNA-protein crosslinks for resolution, highlighting the sophisticated manner in which cells leverage the ubiquitin-proteasome system to maintain proteostasis.
Diagram Title: RNF14-VCP Axis: RPC Resolution Pathway
Experimental Evidence and Key Findings
Formaldehyde-induced RPCs activate the RNF14-VCP pathway
Formaldehyde (FA) is an endogenous metabolite that readily induces covalent crosslinks between proteins and nucleic acids. While its genotoxic effects through DNA-protein crosslinks (DPCs) have been extensively studied, recent evidence demonstrates that RNA-protein crosslinks (RPCs) constitute a significant component of formaldehyde toxicity.
Zhao et al. (2023) established that formaldehyde-induced RPCs cause translation stress by stalling elongating ribosomes, which subsequently leads to collisions with trailing ribosomes [13]. This collision event serves as the activating signal for the RNF14-VCP pathway. The key experimental evidence includes:
PAR-CL system development: Researchers developed a photoactivatable ribonucleoside-enhanced crosslinking (PAR-CL) system using 4-thiouridine (4-SU) labeling and UVA irradiation to specifically induce RPCs without confounding DNA damage. This clean model system enabled the specific study of RPC consequences [13].
Proteomic identification: Using protein-x-linked RNA extraction (XRNAX) coupled with quantitative mass spectrometry, the study identified 719 proteins crosslinked to RNA by formaldehyde, with the majority (561) also crosslinked by the PAR-CL system [13].
Viability assays: Dose-dependent toxicity was observed with PAR-CL treatment, mirroring the effects of formaldehyde exposure, confirming the physiological significance of RPC-induced damage [13].
Quantitative assessment of pathway components
Table 1: Quantitative Effects of RNF14-VCP Pathway Disruption on RPC Resolution
| Experimental Condition | RPC Clearance Efficiency | p38 Phosphorylation | Cell Viability | Reference |
|---|---|---|---|---|
| Wild-type cells | ~85% clearance at 6h | Strong induction | ~70% survival | [13] |
| RNF14 knockout | ~25% clearance at 6h | Abrogated | ~30% survival | [13] [20] |
| VCP inhibition | ~20% clearance at 6h | Sustained activation | ~25% survival | [10] [13] |
| Proteasome inhibition | ~15% clearance at 6h | Sustained activation | ~20% survival | [13] |
| GCN1 depletion | ~30% clearance at 6h | Reduced activation | ~35% survival | [13] [20] |
The data demonstrate that disruption of any component in the RNF14-VCP pathway severely compromises RPC resolution and dramatically reduces cell viability following formaldehyde exposure, underscoring the essential nature of this axis in managing crosslinking damage.
K6 ubiquitin linkage specificity
The RNF14-dependent ubiquitin chains incorporated both K6 and K48 linkages, representing an atypical hybrid ubiquitin code. Several lines of evidence support the functional importance of the K6 linkage:
Chain linkage analysis: Mass spectrometry of ubiquitin chains attached to crosslinked proteins revealed significant enrichment of K6 linkages alongside canonical K48 linkages [13].
Structural insights: The NZF domain of TAB2 can recognize both K6- and K63-linked diubiquitin, suggesting that K6 linkages can serve as specific recognition signals for ubiquitin-binding domains, though the precise readers in the RNF14-VCP pathway remain to be fully elucidated [19].
Functional specialization: The presence of K6 linkages distinguishes this pathway from canonical proteasomal targeting pathways that typically employ homogeneous K48-linked chains, suggesting potential specialized recognition by VCP or associated cofactors [10] [13].
Table 2: K6-Linked Ubiquitin Chain Characteristics in RPC Resolution
| Characteristic | Details | Experimental Evidence |
|---|---|---|
| Chain composition | Mixed K6 and K48 linkages | Mass spectrometry analysis of purified RPCs [13] |
| E2 requirements | Not fully elucidated | In vitro ubiquitylation assays pending |
| Recognition by VCP | Direct or adaptor-mediated | Co-immunoprecipitation and recruitment studies [10] |
| Structural features | Distinct from K48 and K63 chains | TAB2-NZF structural studies [19] |
| Deubiquitylase regulation | Unknown | DUB screening experiments pending |
Research Methods and Protocols
PAR-CL: Specific induction of RNA-protein crosslinks
The Photoactivatable Ribonucleoside-enhanced Crosslinking (PAR-CL) protocol enables specific induction of RPCs without concomitant DNA damage, making it ideal for studying RPC-specific biology [13].
Protocol steps:
- Metabolic labeling: Culture cells in medium containing 5 μM 4-thiouridine (4-SU) for 16 hours to incorporate the photoactivatable nucleoside into newly transcribed RNA.
- Wash and irradiation: Wash cells twice with PBS and irradiate with UVA light (365 nm) at 6 kJ/m² to induce crosslinks between 4-SU-labeled RNA and proximal proteins.
- Crosslink validation: Harvest cells and validate crosslinking efficiency by protein-RNA co-purification using the XRNAX protocol.
- Viability assessment: Measure cell viability 24 hours post-irradiation using ATP-based assays or clonogenic survival.
Critical considerations:
- Include controls for 4-SU-only (no UVA) and UVA-only (no 4-SU) treatments to account for non-specific effects.
- Optimize UVA dose based on cell type; excessive irradiation causes non-specific protein oxidation.
- For proteomic analyses, process samples immediately after crosslinking to minimize post-lysis artifacts.
XRNAX: Isolation and quantification of crosslinked complexes
The protein-x-linked RNA extraction (XRNAX) protocol enables proteome-wide identification of RPCs [13].
Protocol steps:
- Cell lysis: Lyse crosslinked cells in guanidinium isothiocyanate buffer to denature proteins and inactivate RNases.
- RNA-protein co-precipitation: Precipitate RNA and crosslinked proteins using isopropanol, followed by extensive washing with ethanol.
- Proteinase K digestion: Digest non-crosslinked proteins with proteinase K to isolate crosslinked RNA-protein complexes.
- RNA digestion: Treat samples with RNase to release crosslinked peptides for mass spectrometry analysis.
- Mass spectrometry: Analyze peptides by LC-MS/MS using TMT-based multiplexing for quantification.
Applications:
- Identification of RPC-prone proteins under different stress conditions
- Quantification of crosslinking efficiency across experimental conditions
- Assessment of RPC resolution kinetics in genetic mutants
Functional assessment of RNF14-VCP axis activity
Ribosome profiling: Monitor ribosome stalling and collision formation through sucrose density gradient centrifugation and disome/trisome quantification.
Ubiquitin chain linkage analysis: Use linkage-specific affimers or antibodies to detect K6-linked ubiquitin chains in response to RPC induction [18].
VCP activity assays: Measure ATPase activity of VCP in response to RPC formation and assess substrate extraction efficiency through chromatin fractionation.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Essential Research Reagents for Studying RNF14-VCP-K6 Biology
| Reagent/Category | Specific Examples | Function/Application | Source/Reference |
|---|---|---|---|
| E3 Ligase Tools | RNF14 knockout cells | Determine RNF14-specific functions | [20] |
| RNF25 inhibitors | Dissect RNF14-RNF25 network | [20] | |
| Recombinant RNF14 | In vitro ubiquitylation assays | [13] | |
| VCP/p97 Reagents | VCP inhibitors (CB-5083) | Assess VCP requirement in pathway | [10] |
| ATPase-deficient VCP mutants | Mechanism of action studies | [10] | |
| K6 Ubiquitin Tools | K6 linkage-specific affimers | Detect K6 chains in blotting/IF | [18] |
| K6-only ubiquitin mutants | Define linkage specificity | [19] | |
| TAB2-NZF domain | Structural studies of K6 recognition | [19] | |
| Crosslinking Systems | 4-thiouridine (4-SU) | PAR-CL induction of specific RPCs | [13] |
| Formaldehyde | Physiological RPC induction | [13] | |
| Analytical Methods | XRNAX protocol | System-wide RPC identification | [13] |
| Disome profiling | Ribosome collision detection | [13] [20] |
Discussion: Implications for DNA Damage Response Research
The elucidation of the RNF14-VCP axis for resolving K6-marked substrates represents a significant advancement in our understanding of how the ubiquitin system manages transcription- and translation-associated damage. Several key implications emerge for the broader DNA damage response field:
Non-proteolytic ubiquitin signaling: While K6 linkages eventually lead to proteasomal degradation in this pathway, their primary function appears to be in marking specific types of damage (RPCs) for processing by a specialized machinery. This expands the paradigm of ubiquitin signaling in the DDR beyond the canonical roles of K63 (signaling) and K48 (degradation).
Cross-talk between RNA and DNA damage responses: The RNF14-VCP pathway highlights the interconnected nature of different damage response systems. As transcription-blocking lesions affect both DNA and RNA metabolism, understanding how RPC resolution coordinates with classical DNA repair mechanisms represents an important future direction.
Therapeutic opportunities: Given the prevalence of formaldehyde and other crosslinking agents in the environment and their contribution to carcinogenesis, the RNF14-VCP axis presents potential therapeutic targets. Modulating this pathway could sensitize cancer cells to crosslinking chemotherapeutics or protect normal tissues from their side effects.
Unresolved questions: Several key questions remain unanswered, including the specific E2 enzymes that collaborate with RNF14 to assemble K6 linkages, the precise mechanism of VCP recognition of K6-marked substrates, and potential connections to neurodegenerative diseases where protein aggregation and RNA damage coexist.
The RNF14-VCP axis represents a specialized quality control system that leverages the unique properties of K6-linked ubiquitin chains to resolve transcription- and translation-blocking RNA-protein crosslinks. This pathway expands the functional repertoire of the ubiquitin code in managing proteotoxic stress and provides a mechanistic link between ribosome surveillance and the ubiquitin-proteasome system. As research continues to unravel the complexities of K6 ubiquitin signaling, this pathway promises to yield further insights into cellular stress response mechanisms and their implications for human disease.
Genomic instability is a fundamental hallmark of cancer and aging, traditionally viewed through the lens of DNA damage and repair deficiencies. However, emerging research has illuminated a crucial, previously underestimated source of genomic compromise: RNA damage. While RNA has long been considered transient and therefore less critical to genomic integrity, recent studies reveal that lesions on RNA molecules can directly and indirectly promote genomic instability. Central to this emerging pathway is an atypical post-translational modification: K6-linked ubiquitination.
Ubiquitination, the process of attaching ubiquitin proteins to substrate targets, regulates nearly every aspect of cellular function. Among the various polyubiquitin chain linkages, K6-linked chains remain one of the least characterized. Historically implicated in the DNA damage response [21], recent groundbreaking work has identified a specific, critical role for K6-linked ubiquitylation in resolving RNA-protein crosslinks (RPCs) induced by reactive aldehydes [5] [14]. This whitepaper examines the mechanistic connection between RNA damage and genomic instability, focusing specifically on how the K6-ubiquitin pathway protects cells from the genotoxic effects of formaldehyde-induced RPCs, and explores the implications for therapeutic intervention.
K6-Linked Ubiquitin: A Primer on an Atypical Modification
The Ubiquitin Code and Linkage Diversity
Protein ubiquitylation is a multistep process involving E1 (activating), E2 (conjugating), and E3 (ligating) enzymes that results in the covalent attachment of ubiquitin to target proteins [1] [21]. The complexity of this system arises from the ability of ubiquitin itself to form polymer chains through any of its seven lysine residues (K6, K11, K27, K29, K33, K48, K63) or its N-terminal methionine (M1) [1] [21]. Each linkage type creates a distinct structural topology that is recognized by specific effector proteins, forming a "ubiquitin code" that determines diverse biological outcomes ranging from proteasomal degradation (primarily K48-linked chains) to signaling complex assembly (K63-linked chains) [1] [21].
Distinct Properties of K6-Linked Chains
Unlike the well-characterized K48 and K63 linkages, K6-linked ubiquitin chains have remained enigmatic. Gene Ontology classifies protein K6-linked ubiquitination (GO:0085020) as a distinct biological process [22]. Prior to recent discoveries, K6-linkages had been weakly associated with DNA damage response pathways, notably found on the tumor suppressor BRCA1 and its substrates [21]. However, their specific functions, enzymatic regulators, and mechanistic roles were poorly defined. The discovery that K6-linked chains specifically mark formaldehyde-induced RPCs for processing has now positioned this atypical modification as a critical mediator of cytoplasmic quality control with direct implications for genome stability [5].
Table 1: Major Ubiquitin Linkage Types and Their Known Functions
| Linkage Type | Primary Functions | Cellular Processes |
|---|---|---|
| K48 | Targets substrates for proteasomal degradation | Protein turnover, cell cycle regulation |
| K63 | Signaling scaffold, protein-protein interactions | DNA damage repair, NF-κB signaling, endocytosis |
| K11 | Cell cycle regulation, proteasomal degradation | Mitotic progression, ER-associated degradation |
| K6 | DNA damage response, RPC resolution | Genome stability, formaldehyde stress response |
| K27 | DNA damage signaling | DSB repair mediator recruitment |
| M1 (linear) | NF-κB pathway activation | Immune and inflammatory signaling |
Formaldehyde-Induced RNA-Protein Crosslinks: A Source of RNA Damage
Reactive aldehydes, such as formaldehyde, are produced through normal cellular metabolism and accumulate after alcohol consumption [5]. These highly reactive compounds pose a significant threat to macromolecular integrity. When aldehyde clearance mechanisms are impaired, these compounds accumulate in human tissues and have traditionally been associated with toxicity through their ability to damage genomic DNA and inhibit transcription and replication [5] [14]. However, this traditional view has recently been expanded with the discovery that formaldehyde also induces substantial RNA damage.
RPC Formation and Translational Arrest
Formaldehyde directly induces the formation of covalent RNA-protein crosslinks (RPCs) in human cells [5]. When these crosslinks occur in messenger RNA (mRNA), they are recognized by translating ribosomes, which stall at the site of damage [5]. This stalling leads to a profound inhibition of protein synthesis, representing a significant mechanism of cellular toxicity distinct from DNA damage [5] [10]. The accumulation of RPCs therefore constitutes a dual threat: immediate proteostatic stress through translation inhibition, and potential genomic instability through yet-undefined mechanisms.
The K6-Ubiquitin Pathway for RPC Resolution
Pathway Mechanism and Key Components
Recent research has elucidated an evolutionary conserved pathway that specifically targets formaldehyde-induced RPCs for resolution. This pathway involves sequential recognition, marking, and processing steps:
Figure 1: K6-Ubiquitin Pathway for RPC Resolution. Formaldehyde induces RPCs that stall ribosomes during translation. Stalled RPCs are recognized and marked with K6-linked ubiquitin chains by RNF14. VCP/p97 is then recruited to resolve the crosslink.
Recognition and K6-Linked Ubiquitylation
The pathway initiates when translating ribosomes encounter RPCs in mRNA molecules, leading to ribosomal stalling [5]. These stalled complexes are recognized by the RING-in-between-RING (RBR) E3 ligase RNF14, which catalyzes the attachment of atypical K6-linked ubiquitin chains to the crosslinked proteins [5] [14]. RNF14 thereby acts as the central "writer" of the K6-ubiquitin code in this specific stress response.
Recruitment of VCP/p97 and Resolution
The K6-linked ubiquitin chains serve as a specific recognition signal for the ubiquitin- and ATP-dependent unfoldase VCP (also known as p97) [5] [10]. VCP, together with its cofactors, is recruited to the marked RPCs and utilizes ATP hydrolysis to extract and unfold the crosslinked proteins from the RNA moiety [5]. This action resolves the crosslink, potentially allowing for clearance of the damaged RNA and restoration of translation.
Conservation and Coordination
This pathway is evolutionarily conserved and operates in a GCN1-RNF14-VCP-dependent manner [5]. GCN1 is implicated in the recognition of stalled ribosomes, connecting translational arrest to the ubiquitin-mediated resolution machinery. The coordinated action of these factors protects cells against RPC accumulation and suggests that RPCs contribute significantly to the cellular and tissue toxicity of reactive aldehydes [5] [14].
Quantitative Proteomic Analysis of RPC Marking
Key insights into this pathway were revealed through quantitative proteomics, which identified the specific proteins targeted by K6-linked ubiquitylation following formaldehyde exposure:
Table 2: Selected RNA-Binding Proteins Identified as K6-Ubiquitylated After Formaldehyde Treatment
| Protein Target | Function | Fold-Increase in K6-Ubiquitylation | Potential Role in RPC Formation |
|---|---|---|---|
| G3BP1 | Stress granule assembly, RNA binding | High | Forms crosslinks that disrupt mRNA metabolism |
| FUS | RNA processing, DNA repair | High | Crosslinking may impair both RNA and DNA maintenance |
| NONO | Splicing regulator, DNA repair | Moderate | Links transcription-coupled RNA damage |
| RPS3 | Ribosomal protein | High | Directly involved in stalled translation complexes |
| PABPC1 | Poly(A) binding, translation | Moderate | Crosslinks disrupt translation termination |
Experimental Approaches for Studying K6-Ubiquitin in RNA Damage
Key Methodologies and Workflows
The investigation of K6-linked ubiquitin in RPC resolution employs sophisticated biochemical, genetic, and proteomic techniques. The following workflow outlines a comprehensive experimental approach:
Figure 2: Experimental Workflow for K6-Ubiquitin Pathway Analysis. Comprehensive approach combines chemical induction of RPCs with proteomic identification, functional validation, and mechanistic studies.
Detailed Experimental Protocols
Induction and Validation of RPCs
Formaldehyde Treatment: Cells are typically treated with 0.1-1 mM formaldehyde for 1-4 hours in culture to induce RPC formation without immediate cytotoxicity [5]. Dose-response curves should be established for each cell type.
RPC Validation Methods:
- Ribosome Profiling: Ultracentrifugation through sucrose gradients to detect ribosomal stalling at the optical density of 80S monosomes and disomes [5].
- Metabolic Labeling: Click-chemistry-based detection of newly synthesized proteins using homopropargylglycine (HPG) or similar analogues to quantify global translation inhibition.
- Immunofluorescence for RPCs: Modified protein-RNA crosslink detection using specific antibodies against RNA-binding proteins with altered localization.
Detection of K6-Linked Ubiquitylation
K6-Ubiquitin Enrichment:
- Linkage-Specific Antibodies: Immunoprecipitation with validated K6-linkage specific antibodies (commercial availability remains limited).
- Tandem Ubiquitin Binding Entities (TUBEs): Use of engineered ubiquitin-binding domains with preference for K6-linkages in combination with linkage-specific deubiquitinases as negative controls.
- Di-Glycine Remnant Proteomics: Enrichment of ubiquitinated peptides using K-ε-GG antibody purification, though this approach does not distinguish linkage types without additional separation techniques.
Quantitative Proteomics:
- TMT or SILAC Labeling: Stable isotope labeling for quantitative comparison of ubiquitylation sites between treated and untreated samples.
- Mass Spectrometry Parameters: LC-MS/MS with higher-energy collisional dissociation (HCD) fragmentation; data analysis using specialized software (MaxQuant, Proteome Discoverer) with ubiquitin linkage-specific spectral libraries.
Functional Validation of Pathway Components
Genetic Manipulation:
- CRISPR-Cas9 Knockout: Generation of RNF14, VCP, and GCN1 knockout cell lines to validate essential pathway components.
- siRNA/shRNA Knockdown: Transient or stable knockdown of putative pathway members for functional testing.
- Dominant-Negative Expression: Use of ATPase-deficient VCP mutants (e.g., VCP-E305Q/E578Q) to block RPC resolution.
Functional Assays:
- Clonogenic Survival: Measure formaldehyde sensitivity in pathway-deficient cells compared to wild-type controls.
- Translation Recovery Assays: Monitor time-dependent recovery of protein synthesis after formaldehyde washout using metabolic labeling.
- RPC Clearance Quantification: Quantitative PCR-based detection of crosslinked RNA species over time after damage induction.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagents for Studying K6-Ubiquitin in RPC Resolution
| Reagent/Category | Specific Examples | Function/Application |
|---|---|---|
| Chemical Inducers | Formaldehyde, Acetaldehyde | Induce RPC formation for experimental study |
| Linkage-Specific Binders | K6-linkage specific antibodies (in development), TUBEs | Enrich and detect K6-linked ubiquitin chains |
| E3 Ligase Tools | RNF14 expression plasmids, RNF14 knockout cells | Manipulate and study the primary writer of K6 chains in RPCs |
| Proteasome Inhibitors | MG132, Bortezomib | Distinguish proteasome-dependent and independent functions |
| VCP/p97 Inhibitors | CB-5083, NMS-873 | Block the final step of RPC resolution |
| Ubiquitin Mutants | K6R ubiquitin, K6-only ubiquitin | Specifically disrupt or enforce K6-linkage formation |
| Translation Reporters | Puromycylation, HPG labeling, Luciferase-based reporters | Quantify translation inhibition and recovery |
Implications for Genome Stability and Disease
Connecting RNA Damage to Genomic Instability
The pathway connecting RNA damage to genomic instability through K6-ubiquitin involves several mechanistic links:
Replication-Transcription Conflicts: unresolved RPCs may cause collisions between the transcription and replication machineries, leading to DNA double-strand breaks and replication fork collapse [23].
R-Loop Mediated Instability: Compromised RNA metabolism due to RPC accumulation may promote the formation of RNA-DNA hybrids (R-loops), which are potent inducers of DNA damage and genomic rearrangements [23].
Dysregulation of DNA Repair Factors: Many RNA-binding proteins targeted by formaldehyde (e.g., FUS, NONO) have dual roles in DNA repair, suggesting their sequestration in RPCs could directly impair DNA repair capacity [5] [23].
Therapeutic Opportunities and Future Directions
The discovery of this specialized K6-ubiquitin pathway presents several promising therapeutic avenues:
Cancer Therapy Sensitization: Inhibitors of VCP or RNF14 could potentiate the effects of chemotherapeutic agents that generate aldehydes or other RNA-damaging compounds.
Neurodegeneration Applications: As neurodegenerative diseases often involve defects in RNA quality control and protein aggregation, modulating K6-ubiquitylation might help clear pathological RPC-like species.
Biomarker Development: Quantification of K6-ubiquitylated proteins in liquid biopsies might serve as a biomarker for aldehyde exposure or defects in RNA damage response.
Future research should focus on identifying the complete set of writers, readers, and erasers of the K6-ubiquitin code, understanding potential crosstalk with other ubiquitin linkages, and developing specific chemical probes to manipulate this pathway therapeutically.
The discovery that K6-linked ubiquitin chains mark formaldehyde-induced RNA-protein crosslinks for resolution represents a significant advancement in understanding the connections between RNA quality control and genome stability. This pathway reveals a previously unappreciated mechanism by which cells defend against the genotoxic effects of reactive aldehydes, with implications for cancer biology, chemical toxicology, and therapeutic development. As research in this area progresses, the K6-ubiquitin code will likely emerge as a crucial regulatory system integrated throughout the DNA damage response network, offering new targets for intervention in genome instability-associated diseases.
Advanced Tools and Techniques for Profiling K6-Ubiquitin Signaling
The study of atypical ubiquitin linkages has been historically challenging due to a lack of high-affinity, linkage-specific reagents. This technical guide details the development and application of chain-specific Tandem Ubiquitin-Binding Entities (TUBEs) for the capture and analysis of endogenous K6-linked ubiquitination, with particular emphasis on their utility in DNA damage response (DDR) research. We provide comprehensive methodologies for employing K6-specific TUBEs in western blotting, immunoprecipitation, confocal microscopy, and mass spectrometry workflows. Furthermore, we present structured data on reagent performance, detailed experimental protocols, and essential research tools that enable researchers to overcome traditional barriers in ubiquitin research. The integration of these high-affinity probes into DDR studies is illuminating the critical functions of K6-linked ubiquitination in maintaining genomic integrity and coordinating cellular stress responses.
Protein ubiquitination, the covalent attachment of ubiquitin to substrate proteins, is a fundamental post-translational modification that regulates diverse cellular processes including protein degradation, DNA repair, and signal transduction [1] [24]. The versatility of ubiquitin signaling stems from the ability of ubiquitin molecules to form polymers (polyubiquitin chains) through eight different linkage types: M1 (linear), K6, K11, K27, K29, K33, K48, and K63 [11]. Among these, K48-linked chains predominantly target proteins for proteasomal degradation, while K63-linked chains and M1-linked chains play key roles in signaling pathways such as NF-κB activation and inflammatory responses [1] [19].
K6-linked ubiquitin chains belong to the group of "atypical" ubiquitin linkages that have remained less studied due to technical limitations in specific detection methods [25]. However, emerging evidence has established important functions for K6 linkages in cellular stress responses, particularly in the DNA damage response. Early studies linked K6 ubiquitination to the E3 ubiquitin ligase BRCA1, which is mutated in hereditary breast and ovarian cancers [25]. Additionally, K6 chain levels increase following DNA damage, suggesting a regulated role in genomic maintenance [25] [1]. The RBR E3 ubiquitin ligase Parkin assembles K6-linked chains during mitophagy, a process counteracted by the K6-selective deubiquitinase USP30 [25]. More recent work has identified HUWE1 as a major E3 ligase for K6 chains in cells, with specific substrates including mitofusin-2 (Mfn2) [25].
The molecular analysis of K6-linked ubiquitination has been hampered by the scarcity of tools capable of specifically recognizing this linkage type among the complex landscape of ubiquitin signals in cells. Traditional antibodies face challenges in distinguishing between different ubiquitin linkages, particularly for the less abundant atypical chains. This technical gap has limited our understanding of the dynamics, regulation, and functional consequences of K6 ubiquitination in DDR and other pathways.
The Development of K6-Linkage Specific Affinity Reagents
From Traditional Antibodies to Advanced Affimer Technology
Initial attempts to study linkage-specific ubiquitination relied on antibodies generated through phage display, which have been successfully developed for five of the eight ubiquitin linkage types (K11, K48, K63, M1) but remained unavailable for K6, K27, and K33 linkages [25]. The high conservation of ubiquitin across species makes conventional antibody generation challenging, as the immune system often fails to recognize subtle differences in linkage-specific epitopes.
Affimer technology provides an alternative approach using non-antibody protein scaffolds based on the stable cystatin fold (Figure 1A) [25]. These 12-kDa scaffolds offer several advantages over traditional antibodies:
- Enhanced specificity: Randomized surface loops can be engineered to recognize specific ubiquitin linkage conformations
- High-affinity binding: Affimers selected from large libraries (10¹⁰ variants) exhibit tight binding to cognate antigens
- Stability: The cystatin fold provides a robust scaffold resistant to denaturation
- Versatile conjugation: Site-specific biotinylation enables use in various detection methods
Table 1: Comparison of Ubiquitin Detection Technologies
| Technology | Linkage Specificity | Affinity | Applications | Limitations |
|---|---|---|---|---|
| Traditional Antibodies | Available for 5/8 linkages | Variable | WB, IF, IP | Difficult to generate for conserved epitopes |
| Affimer Proteins | Available for K6, K33/K11 | High (nM range) | WB, IF, IP, pull-downs, microscopy | Requires protein engineering |
| TUBE Technology | Linkage-specific variants | High (nM range) | Enrichment, proteomics, functional studies | Limited commercial availability |
Structural Basis of K6-Linkage Specificity
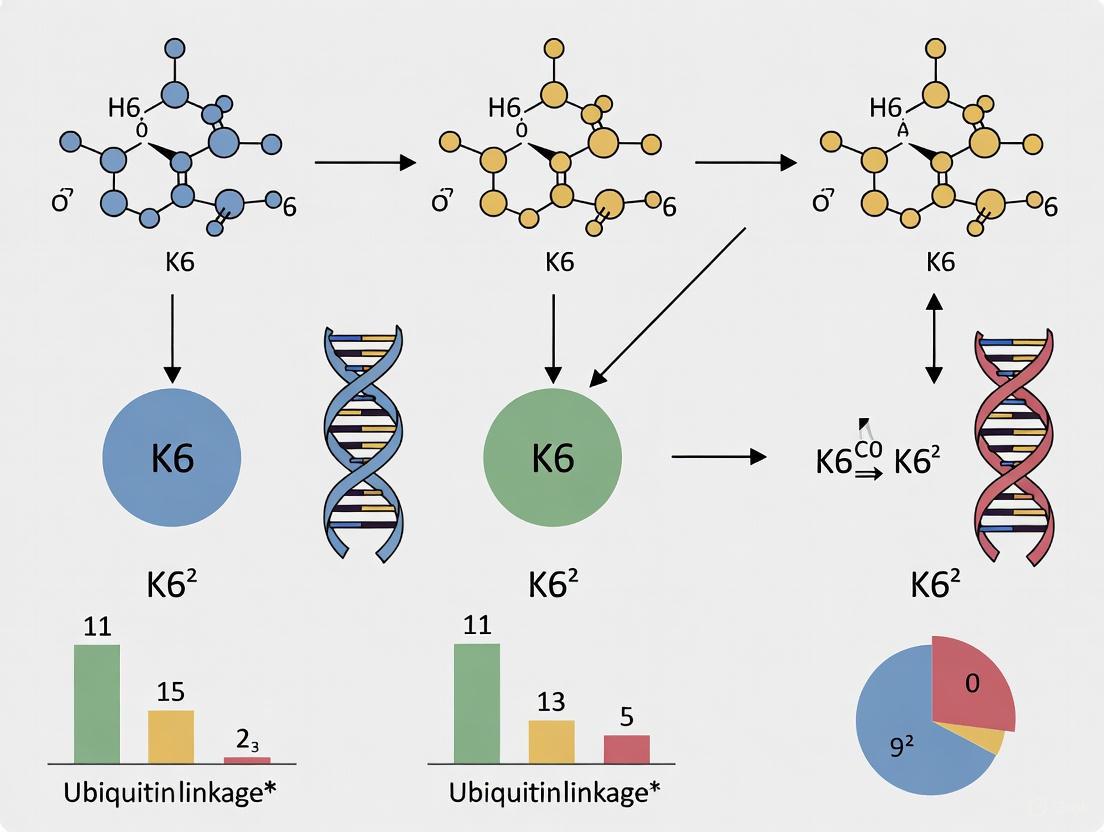
The molecular mechanism underlying K6-linkage specificity has been elucidated through X-ray crystallography of affimers bound to K6-linked diubiquitin (Figure 1B) [25]. The structures reveal that:
- Dimeric binding mode: Each affimer molecule binds one ubiquitin moiety, with affimer dimerization creating two binding sites for ubiquitin I44 patches with defined distance and orientation
- Linkage-specific recognition: The spatial arrangement of binding sites preferentially accommodates the geometry of K6-linked chains
- Variable loop interactions: Randomized loops in the affimer scaffold mediate both dimerization and ubiquitin recognition
This structural arrangement mimics naturally occurring ubiquitin-binding domains (UBDs) that provide multiple binding surfaces, where only the cognate linkage can simultaneously engage both sites, leading to enhanced specificity and affinity [25].
The K6-specific affimer exhibits high selectivity, with minimal cross-reactivity to other linkage types as demonstrated by isothermal titration calorimetry (ITC) and surface plasmon resonance (SPR) [25]. Qualitative kinetic analysis by SPR showed that linkage specificity is achieved through very slow off-rates specifically for K6-linked diubiquitin.
Figure 1: Structural basis of K6-linkage recognition. Affimer proteins dimerize to create two ubiquitin-binding sites that specifically accommodate the geometry of K6-linked diubiquitin.
Quantitative Characterization of K6-Specific Reagents
Binding Affinity and Specificity Profiles
Rigorous biochemical characterization of K6-specific affimers has provided quantitative data on their performance (Table 2). Isothermal titration calorimetry (ITC) measurements demonstrated tight binding to K6-linked diubiquitin with no detectable interaction to K33-linked chains [25]. The binding stoichiometry indicated formation of a 2:1 affimer:diUb complex (n = 0.46 for K6 affimer), consistent with the dimeric binding mode observed crystallographically [25].
Table 2: Quantitative Binding Parameters of K6-Specific Affimers
| Parameter | Value | Measurement Method | Experimental Conditions |
|---|---|---|---|
| Binding Stoichiometry (n) | 0.46-0.44 | Isothermal Titration Calorimetry (ITC) | 5 μM protein concentration |
| K6 diUb Specificity | High (no cross-reactivity with K33) | ITC, Western Blot | Various buffer conditions |
| Off-target Recognition | Minimal with diUb, increased with tetraUb | Western Blot | 50 nM reagent concentration |
| Detection Limit | ~50 nM | Western Blot | Chemiluminescent detection |
Surface plasmon resonance (SPR) analysis provided qualitative kinetic data, showing that linkage specificity is achieved through very slow off-rates specifically for cognate K6-linked diubiquitin [25]. In western blot applications, site-specifically biotinylated K6 affimers detected K6 diUb with high linkage specificity at concentrations as low as 50 nM, though cross-reactivity became more pronounced with tetraUb molecules, suggesting that avidity effects can influence specificity in certain contexts [25].
Comparison with Alternative K6-Binding Domains
Beyond engineered affimers, certain natural ubiquitin-binding domains also exhibit K6 linkage recognition. The NZF domain of TAB2 (TAK1-binding protein 2), previously characterized as K63-specific, was recently found to interact with K6-linked chains through comprehensive diubiquitin interaction screens [19] [26]. Structural analysis of TAB2-NZF in complex with K6-linked diubiquitin revealed a similar binding mode to K63-linked chains, with flexibility in the C-terminal region of the distal ubiquitin contributing to this dual specificity [19].
Table 3: Comparison of K6-Linkage Binding Reagents
| Reagent | Specificity Profile | Affinity | Structural Basis | Applications Demonstrated |
|---|---|---|---|---|
| K6-Specific Affimer | Highly specific for K6 linkages | High (nM range) | Dimeric binding to both ubiquitin moieties | WB, IF, IP, pull-downs, microscopy |
| TAB2-NZF Domain | Dual specificity for K6 and K63 | Moderate | Flexible C-tail accommodation | Interaction studies, signaling analysis |
| Traditional Antibodies | Not available for K6 | N/A | N/A | N/A |
This dual specificity of natural UBDs like TAB2-NZF highlights the challenge of achieving absolute linkage specificity and underscores the value of engineered reagents like K6-specific affimers for applications requiring unambiguous K6 chain detection.
Experimental Protocols for K6-Linked Ubiquitin Analysis
Western Blotting with K6-Specific Affimers
Materials Required:
- Site-specifically biotinylated K6-specific affimer [25]
- Control lysates from cells with modulated K6 ubiquitination (e.g., HUWE1 knockdown) [25]
- Streptavidin-HRP conjugate or anti-biotin antibodies
- Standard western blotting equipment and reagents
Procedure:
- Sample Preparation: Prepare cell lysates using RIPA buffer supplemented with proteasome inhibitors (e.g., MG132) and deubiquitinase inhibitors (e.g., N-ethylmaleimide) to preserve ubiquitination states [27].
- Electrophoresis and Transfer: Separate proteins by SDS-PAGE and transfer to PVDF membrane using standard protocols.
- Blocking: Block membrane with 5% non-fat dry milk in TBST for 1 hour at room temperature.
- Affimer Incubation: Incubate membrane with biotinylated K6-specific affimer (50-100 nM) in blocking buffer overnight at 4°C [25].
- Detection: Wash membrane and incubate with streptavidin-HRP conjugate (1:5000 dilution) for 1 hour at room temperature. Develop using enhanced chemiluminescence.
- Validation: Confirm specificity using:
- Lysates from HUWE1-deficient cells (should show reduced K6 signal) [25]
- Recombinant ubiquitin chains of defined linkages
- Competition with free K6-linked diubiquitin
Immunoprecipitation and Pull-Down Assays
Materials Required:
- Immobilized K6-specific TUBEs (biotinylated affimers coupled to streptavidin beads) [25] [27]
- Control beads with non-specific affimers or empty beads
- Lysis buffer: 50 mM Tris-HCl (pH 7.5), 150 mM NaCl, 1% NP-40, 0.5% sodium deoxycholate, 1 mM EDTA with fresh protease inhibitors
- Wash buffers of varying stringency
Procedure:
- Lysate Preparation: Harvest cells and lyse in appropriate buffer. Clarify by centrifugation at 15,000 × g for 15 minutes at 4°C. Determine protein concentration.
- Bead Preparation: Wash immobilized K6-TUBE beads with lysis buffer. Use 20-50 μL bead slurry per mg of total protein.
- Binding Reaction: Incubate lysate with K6-TUBE beads for 2-4 hours at 4°C with gentle rotation.
- Washing: Pellet beads and wash sequentially with:
- 3× with lysis buffer
- 2× with high-salt buffer (lysis buffer with 500 mM NaCl)
- 1× with no-detergent buffer (50 mM Tris-HCl, pH 7.5, 150 mM NaCl)
- Elution: Elute bound proteins with 2× SDS sample buffer at 95°C for 10 minutes.
- Downstream Analysis: Analyze eluates by:
Confocal Immunofluorescence Microscopy
Materials Required:
- Fluorescently labeled K6-specific affimers (e.g., conjugated to FITC or Alexa Fluor dyes) [25]
- Cells grown on glass coverslips
- DNA damage inducers: etoposide (50-100 μM), neocarzinostatin (100-250 ng/mL), or ionizing radiation (2-10 Gy)
- Fixation and permeabilization reagents
- Mounting medium with DAPI
Procedure:
- Treatment and Fixation: Treat cells with DNA damaging agents for appropriate times. Fix with 4% paraformaldehyde for 15 minutes at room temperature.
- Permeabilization: Permeabilize cells with 0.2% Triton X-100 in PBS for 10 minutes.
- Blocking: Block with 3% BSA in PBS for 30 minutes.
- Staining: Incubate with fluorescent K6-affimer (10-50 nM) in blocking buffer for 1-2 hours at room temperature.
- Washing: Wash 3× with PBS for 5 minutes each.
- Mounting and Imaging: Mount coverslips using anti-fade mounting medium. Image using confocal microscopy with appropriate laser lines and filter sets.
- Co-staining: Combine with antibodies against DNA damage markers (γH2AX, 53BP1) to assess co-localization.
Figure 2: Workflow for K6-linked ubiquitin enrichment using chain-specific TUBEs in pull-down assays.
Application in DNA Damage Response Research
Identifying K6-Ubiquitinated Proteins in DDR
The application of K6-specific TUBEs in pull-down experiments coupled with mass spectrometry has enabled systematic identification of proteins modified with K6-linked ubiquitin chains in the context of DNA damage. Key findings include:
HUWE1 as a Major K6 E3 Ligase: Pull-downs with K6-specific affimers identified the HECT E3 ligase HUWE1 as a primary source of cellular K6 chains [25]. HUWE1 assembles K6-, K11-, and K48-linked polyubiquitin chains in vitro, and HUWE1−/− or knockdown cells show significantly reduced levels of K6 chains.
Mitofusin-2 as a K6-Modified Substrate: K6-specific TUBEs demonstrated that mitofusin-2 (Mfn2) is modified with K6-linked polyubiquitin in a HUWE1-dependent manner [25], linking K6 ubiquitination to mitochondrial dynamics and quality control.
RNF144A/B as K6 Chain Assemblers: In vitro studies with K6-specific western blotting revealed that the RBR E3 ligases RNF144A and RNF144B assemble predominantly K6-, K11-, and K48-linked chains [25].
DNA Damage-Induced K6 Interactors: UbIA-MS (ubiquitin interactor affinity enrichment-mass spectrometry) using diubiquitin baits identified a class of monoubiquitin and K6 diubiquitin interactors whose binding is induced by DNA damage [26].
Functional Roles of K6 Linkages in Genome Maintenance
The development of specific reagents has accelerated functional characterization of K6 ubiquitination in DNA damage response pathways:
BRCA1-Mediated DNA Repair: Early studies linked the K6 linkage to the E3 ubiquitin ligase BRCA1, which is mutated in hereditary breast and ovarian cancers [25]. K6-specific reagents now enable detailed analysis of BRCA1-dependent ubiquitination events in DNA repair complex assembly.
Parkin and Mitophagy: The RBR E3 ligase Parkin assembles K6-linked chains important for mitophagy [25], a process counteracted by the K6-selective deubiquitinase USP30. This pathway represents a quality control mechanism for damaged mitochondria, with implications for genome stability through reduction of oxidative stress.
TAB2/TAK1 Signaling: The identification of TAB2 and TAB3 as K6 diubiquitin interactors suggests potential roles for K6 chains in TAK1 complex activation and downstream NF-κB and JNK signaling [19] [26], which can influence cell fate decisions in response to genotoxic stress.
Histone Ubiquitination: K6-specific reagents have enabled investigation of potential K6 ubiquitination on histones and its contribution to chromatin remodeling at DNA damage sites, analogous to the established roles of K63 and K48 ubiquitination in this process [24].
The Scientist's Toolkit: Essential Research Reagents
Table 4: Key Reagents for K6-Linked Ubiquitin Research
| Reagent | Function/Application | Key Features | Source/Generation |
|---|---|---|---|
| K6-Specific Affimers | Detection and enrichment of K6 linkages | High affinity (nM range), linkage-specific | Avacta (commercial) or academic sources [25] |
| K6-Linked Diubiquitin | Specificity controls, competition assays | Defined linkage, recombinant production | In-house synthesis with specific E2/E3 pairs [19] |
| HUWE1-Deficient Cells | Specificity validation, functional studies | Reduced cellular K6 levels | CRISPR/Cas9 knockout or RNAi knockdown [25] |
| Linkage-Specific DUBs | Validation of linkage identity | Enzymatic confirmation of chain type | USP30 for K6 chain validation [25] |
| TAB2-NZF Domain | Alternative K6-binding reagent | Dual specificity for K6/K63 | Recombinant expression [19] |
Technical Considerations and Limitations
While K6-specific TUBEs represent a significant advancement in ubiquitin research, several technical considerations must be addressed for appropriate experimental design and data interpretation:
Concentration-Dependent Effects: The K33 affimer showed binding in ITC experiments (at 5 μM) but not in western blotting (at 50 nM), highlighting the importance of reagent concentration in assay sensitivity and specificity [25].
Chain Length Considerations: The K6 affimer showed minimal cross-reactivity with diUb but increased detection of other linkages with tetraUb, suggesting that avidity effects can influence specificity with longer chains [25].
Cellular Context Dependence: K6 ubiquitination patterns and abundance vary by cell type and physiological conditions, requiring optimization of enrichment protocols for different experimental systems.
Branched Chain Complexity: Emerging evidence for branched ubiquitin chains containing K6 linkages (e.g., K6/K11, K6/K48) [11] adds complexity to data interpretation, as current reagents may not distinguish homotypic from branched chains.
Validation Requirements: Multiple orthogonal approaches (e.g., genetic manipulation of E3 ligases, linkage-specific DUB treatment, mass spectrometry confirmation) should be employed to validate K6 linkage identification.
The development of chain-specific TUBEs for K6-linked ubiquitination represents a transformative advancement in the ubiquitin field, finally enabling comprehensive study of this elusive modification. These high-affinity probes have already illuminated the roles of HUWE1, RNF144A/B, and other E3 ligases in assembling K6 chains, while also identifying key substrates like mitofusin-2.
In DNA damage response research, these tools are poised to decipher the specific contributions of K6 ubiquitination to genome maintenance pathways. Future applications include single-molecule analysis of K6 chain dynamics in live cells, structural studies of K6-specific reader domains, and therapeutic development targeting K6-specific enzymes in cancer and neurodegenerative diseases.
As the ubiquitin field continues to recognize the importance of atypical linkages and complex chain architectures, the methodological framework established for K6-specific TUBEs provides a blueprint for developing similar reagents for other understudied ubiquitin linkages. The integration of these tools with emerging technologies in microscopy, proteomics, and structural biology will undoubtedly yield new insights into the complex language of ubiquitin signaling in health and disease.
Mass Spectrometry-Based Proteomics for Identifying K6-Ubiquitin Substrates and Sites
K6-linked ubiquitin chains represent one of the less characterized atypical ubiquitin modifications with emerging significance in the cellular DNA damage response. While historically challenging to study due to technical limitations, recent advances in mass spectrometry-based proteomics have enabled the identification of K6-ubiquitin substrates and their specific modification sites. This technical guide comprehensively outlines contemporary methodologies for investigating K6-linked ubiquitylation, with particular emphasis on ubiquitin remnant profiling, linkage-specific enrichment strategies, and quantitative proteomic approaches. We detail experimental workflows from sample preparation to data analysis, provide resources for linkage-specific reagents, and contextualize findings within DNA damage response mechanisms, offering researchers a foundational framework for exploring this biologically significant but understudied ubiquitin linkage.
Protein ubiquitylation involves the covalent attachment of ubiquitin to substrate proteins through a sequential enzymatic cascade involving E1 (activating), E2 (conjugating), and E3 (ligating) enzymes [1] [9]. Ubiquitin itself contains seven lysine residues (K6, K11, K27, K29, K33, K48, K63) and an N-terminal methionine that can serve as linkage points for polyubiquitin chain formation [28]. Among these, K6-linked ubiquitin chains have emerged as important regulators in maintaining genomic integrity, though they remain less characterized than the well-studied K48 and K63 linkages [1].
The functional significance of K6-linked ubiquitylation spans several critical cellular processes. In the DNA damage response, K6-linked chains contribute to the regulation of DNA repair pathways, with the ubiquitin ligase BRCA1 implicated in generating K6 linkages in response to genotoxic stress [29]. Recent research has also revealed roles for K6-ubiquitin chains in mobilizing p97/VCP and the proteasome to resolve formaldehyde-induced RNA-protein crosslinks, highlighting their importance in managing complex DNA damage scenarios [10]. Furthermore, evidence suggests that K6 linkages participate in mitochondrial quality control and the regulation of apoptosis, indicating functional diversity beyond DNA repair mechanisms [1].
Despite these important roles, studying K6-linked ubiquitylation presents distinct technical challenges. K6 linkages typically occur at lower abundance compared to canonical K48 and K63 chains, necessitating highly sensitive enrichment and detection methods [29]. Additionally, the structural diversity of ubiquitin chains and potential mixed linkages complicate unambiguous identification. This technical guide addresses these challenges by providing comprehensive methodologies for the specific identification and characterization of K6-ubiquitin substrates and modification sites using contemporary mass spectrometry-based approaches.
Methodological Approaches for K6-Ubiquitin Substrate Identification
Ubiquitin Remnant Profiling (DiGly Capture)
Ubiquitin remnant profiling represents a powerful shotgun proteomics approach that enables proteome-wide identification of endogenous ubiquitylation sites. This methodology exploits the fact that tryptic digestion of ubiquitylated proteins leaves a di-glycine (diGly) remnant (~114 Da) from the C-terminus of ubiquitin covalently attached to the modified lysine residue [29]. The diGly remnant produces a characteristic mass shift that can be detected by mass spectrometry and pinpoint the exact ubiquitin attachment site within the substrate protein.
The critical advancement enabling this approach was the development of diGly lysine-specific antibodies (e.g., clone GX41) that specifically recognize the di-glycine adduct on the ε-amine of lysine residues [29]. These antibodies allow specific enrichment of ubiquitin remnant peptides from complex protein digests, significantly reducing sample complexity and facilitating identification of low-abundance ubiquitylation sites. The standard workflow involves:
- Protein extraction under denaturing conditions to preserve ubiquitylation status
- Proteolytic digestion with trypsin to generate diGly-modified peptides
- Immunoaffinity enrichment using diGly remnant-specific antibodies
- LC-MS/MS analysis of enriched peptides for identification and site mapping
When applied to K6-linked ubiquitylation studies, ubiquitin remnant profiling has revealed that UV light irradiation increases cellular abundance of K6-linked ubiquitin chains [29]. This approach can be combined with SILAC-based quantitative proteomics to analyze site-specific alterations in the ubiquitin-modified proteome following DNA damage or other cellular perturbations [29]. However, researchers should note that tryptic digestion of proteins modified with ubiquitin-like modifiers NEDD8 and ISG15 also generates di-glycine remnants, potentially complicating data interpretation without additional validation experiments [29].
Linkage-Specific Enrichment Strategies
Tandem Ubiquitin Binding Entities (TUBEs)
Tandem Ubiquitin Binding Entities (TUBEs) represent a innovative technology for linkage-specific ubiquitin chain enrichment. TUBEs are engineered protein constructs containing multiple ubiquitin-binding domains that exhibit nanomolar affinities for polyubiquitin chains, offering significant advantages over traditional single-domain approaches [12]. The modular design of TUBEs allows for development of chain-specific variants that selectively recognize particular ubiquitin linkages, including K6-linked chains.
The application of chain-specific TUBEs enables researchers to:
- Preserve labile ubiquitin signals by protecting against deubiquitinating enzyme (DUB) activity during extraction
- Enrich specific ubiquitin linkages from native cell lysates without requiring genetic modification
- Capture endogenous ubiquitination events in a physiological context
- Differentiate context-dependent linkage specificity in cellular signaling
In practice, K6-specific TUBEs can be immobilized on magnetic beads or microplate surfaces to facilitate high-throughput screening of K6-ubiquitylated substrates [12]. This approach has been successfully implemented to investigate DNA damage-dependent protein ubiquitination, revealing a class of K6 diubiquitin interactors whose binding is induced by genotoxic stress [26].
Ubiquitin Interactor Affinity Enrichment-Mass Spectrometry (UbIA-MS)
UbIA-MS represents a quantitative interaction proteomics method that utilizes chemically synthesized diubiquitin of defined linkage to enrich and identify ubiquitin linkage interactors from complex cell lysates [26]. This approach has been successfully applied to profile K6-linked diubiquitin interactors, revealing novel components of the K6-ubiquitin signaling network.
The UbIA-MS workflow involves:
- Preparation of linkage-defined diubiquitin baits through chemical synthesis
- Affinity enrichment of diubiquitin-interacting proteins from cell lysates
- Quantitative MS-based identification of specific interactors
- Functional validation of candidate proteins through orthogonal approaches
Application of UbIA-MS has identified TAB2 and TAB3 as novel K6 diubiquitin interactors, expanding our understanding of the cellular proteins that recognize and respond to this atypical ubiquitin linkage [26].
Table 1: Comparison of K6-Ubiquitin Substrate Identification Methods
| Method | Principle | Advantages | Limitations | Applications in K6 Studies |
|---|---|---|---|---|
| Ubiquitin Remnant Profiling | Enrichment of diGly-modified peptides after tryptic digestion | - Identifies exact modification sites- High sensitivity- Proteome-wide coverage | - Cannot distinguish ubiquitin from NEDD8/ISG15- Loses information about chain architecture | - Identification of UV-induced K6 sites [29]- Quantification of K6 chain abundance after DNA damage |
| Chain-Specific TUBEs | Affinity enrichment using engineered ubiquitin-binding entities | - Preserves labile modifications- Linkage-specific capture- Compatible with native conditions | - Requires validation of linkage specificity- May miss low-abundance substrates | - Enrichment of DNA damage-induced K6 ubiquitinated proteins [12] [26] |
| UbIA-MS | Interaction profiling with defined diubiquitin baits | - Identifies linkage-specific interactors- Reveals signaling networks- Quantitative capabilities | - Does not directly identify substrates- Focused on interactors rather than modified proteins | - Discovery of TAB2/TAB3 as K6 interactors [26]- DNA damage-induced K6 interactome |
Quantitative Proteomic Approaches
Quantitative mass spectrometry approaches significantly enhance K6-ubiquitin research by enabling researchers to distinguish specific ubiquitination changes from background fluctuations. Stable Isotope Labeling with Amino Acids in Cell Culture (SILAC) has been successfully employed in combination with ubiquitin remnant profiling to quantify site-specific alterations in ubiquitylation after DNA damage induction [29].
The experimental design for quantitative K6-ubiquitin analysis typically involves:
- Metabolic labeling of cells with light, medium, and heavy isotope forms of lysine and arginine
- Application of experimental conditions (e.g., DNA damaging agents vs. control)
- Combined processing of samples after cell lysis to minimize technical variability
- Ratio-based quantification of ubiquitination sites across conditions
This approach has revealed that proteasome inhibition leads to global perturbations of cellular ubiquitylation patterns, including specific effects on K6-linked ubiquitylation [29]. Additionally, researchers can employ tandem mass tags (TMT) or isobaric tags for relative and absolute quantitation (iTRAQ) for multiplexed analysis of multiple conditions simultaneously, particularly useful for time-course studies of K6-ubiquitin dynamics after DNA damage [30].
Experimental Workflow for K6-Ubiquitin Substrate Identification
Sample Preparation and Ubiquitin Enrichment
Proper sample preparation is critical for successful identification of K6-ubiquitin substrates, particularly due to the lability of ubiquitin modifications and the activity of deubiquitinating enzymes (DUBs). The recommended workflow begins with cell lysis under denaturing conditions (e.g., in 6 M guanidine hydrochloride or 8 M urea) to inactivate DUBs and preserve ubiquitin signatures [29]. For studies focusing on DNA damage response, appropriate genotoxic stress conditions should be optimized based on the cellular model and research questions—common inducters include ultraviolet (UV) irradiation, ionizing radiation, or specific DNA-damaging agents like cisplatin [29].
Following protein extraction, researchers can pursue two primary enrichment strategies:
Linkage-specific enrichment at the protein level using K6-specific TUBEs or antibodies, which preserves information about the ubiquitin chain topology but may miss low-abundance modifications [12] [26].
Ubiquitin remnant profiling at the peptide level, which offers higher sensitivity for site identification but loses information about chain length and architecture [29].
For comprehensive analysis, a parallel approach using both methods provides complementary data: linkage-specific enrichment reveals proteins modified by K6-linked chains, while remnant profiling identifies the exact modification sites. When working with limited sample material, such as primary cells, remnant profiling generally offers higher sensitivity due to more effective reduction of sample complexity [29].
Mass Spectrometry Analysis and Data Acquisition
Modern high-resolution mass spectrometry platforms are essential for confident identification of K6-ubiquitination sites. Recommended instrumentation includes Orbitrap-based mass analyzers (e.g., Orbitrap Exploris, Orbitrap Fusion series) capable of high mass accuracy (<5 ppm) and rapid sequencing of peptide ions [31]. Data-dependent acquisition (DDA) methods are commonly employed for discovery-phase experiments, while data-independent acquisition (DIA) approaches offer advantages for reproducible quantification across multiple samples.
Key parameters for LC-MS/MS analysis of ubiquitin remnant peptides include:
- LC separation: Nano-flow chromatography with extended (60-120 min) acetonitrile gradients for optimal peptide separation
- MS1 resolution: ≥60,000 for accurate peptide identification
- Fragmentation: Higher-energy collisional dissociation (HCD) with optimized normalized collision energy (25-32%)
- Dynamic exclusion: 30-45 seconds to maximize identifications
For targeted analysis of specific K6-ubiquitination sites, parallel reaction monitoring (PRM) offers superior sensitivity and quantification accuracy. This approach is particularly valuable for validating candidate sites identified in discovery-phase experiments and monitoring their dynamics in response to DNA damage or other cellular stimuli [29].
Data Analysis and Bioinformatics
Bioinformatic analysis of K6-ubiquitin proteomics data requires specialized workflows to address the unique challenges of ubiquitin remnant identification. Standard database search engines (e.g., MaxQuant, Proteome Discoverer) should be configured with the following parameters:
- Variable modification: GlyGly modification on lysine (+114.0429 Da)
- Enzyme specificity: Trypsin with up to 2 missed cleavages
- False discovery rate (FDR): ≤1% at both peptide and protein levels
Following identification, data should be processed through additional filtering steps to remove potential false positives from non-ubiquitin diGly modifications (e.g., from NEDD8 or ISG15) [29]. This can be achieved through manual validation of spectral quality or using computational tools that apply machine learning algorithms to distinguish authentic ubiquitin remnant peptides based on fragmentation patterns and sequence context.
Functional interpretation of identified K6-ubiquitin substrates can be enhanced through:
- Pathway enrichment analysis using databases like KEGG or Reactome
- Protein-protein interaction network mapping to identify functional modules
- Integration with transcriptional data to understand system-level responses
- Comparison with existing ubiquitin databases to identify novel vs. known substrates
Research Reagent Solutions for K6-Ubiquitin Studies
Table 2: Essential Research Reagents for K6-Ubiquitin Substrate Identification
| Reagent Category | Specific Examples | Key Features | Applications in K6 Studies |
|---|---|---|---|
| Enrichment Tools | K6-linkage specific TUBEs (LifeSensors) | - High affinity for K6 chains- DUB-protective properties- Magnetic bead conjugates available | - Selective enrichment of K6-ubiquitinated proteins [12]- Preservation of labile K6 modifications |
| diGly remnant antibodies (GX41 clone) | - Specific to diGly-modified lysine- Compatible with multiple species- Works in enrichment and immunoblotting | - Ubiquitin remnant profiling [29]- Validation of K6-ubiquitination sites | |
| Mass Spec Standards | Heavy-labeled ubiquitin reference peptides | - Isotopically labeled internal standards- Quantification of ubiquitin chains- SILAC compatibility | - Absolute quantification of K6 linkages- Normalization across experiments |
| Activity Probes | Linkage-specific DUB substrates | - Fluorescent or affinity-tagged reporters- K6-linkage specific sequences- Controlled cleavage sites | - Monitoring DUB activity against K6 chains- Validating linkage specificity |
| Cell Resources | (His)6-ubiquitin transgenic mouse model | - Tissue-specific expression- Endogenous regulation- Physiological relevance | - Study of K6 ubiquitination in vivo [31]- Tissue-specific K6 substrate identification |
K6-Ubiquitin in DNA Damage Response: Biological Context
The functional significance of K6-linked ubiquitination is particularly evident in the cellular DNA damage response, where it participates in critical regulatory mechanisms. Mass spectrometry-based studies have revealed that UV light irradiation specifically increases cellular abundance of K6-linked ubiquitin chains, suggesting a dedicated role for this linkage in managing genotoxic stress [29]. This finding aligns with earlier work implicating the tumor suppressor BRCA1 in generating K6-linked ubiquitin chains as part of its DNA repair functions [29].
Recent research has expanded our understanding of K6-ubiquitin in DNA damage contexts through the discovery of DNA damage-induced K6 diubiquitin interactors [26]. These proteins, which selectively bind K6-linked diubiquitin in response to genotoxic stress, represent key effectors that translate the ubiquitin signal into functional outcomes. Among the identified interactors, TAB2 and TAB3 have emerged as novel components of the K6-ubiquitin signaling network, potentially linking this modification to broader cellular stress response pathways [26].
Beyond canonical DNA repair, K6-linked ubiquitination has been implicated in resolving formaldehyde-induced RNA-protein crosslinks, with recent studies demonstrating the mobilization of p97/VCP and the proteasome through K6-ubiquitin signals to clear these transcription-blocking lesions [10]. This mechanism highlights the versatility of K6 linkages in managing diverse forms of genomic stress and maintaining transcriptional integrity.
The development of experimental tools for studying K6-ubiquitin in DNA damage contexts continues to advance, with linkage-specific TUBEs and UbIA-MS approaches enabling more precise mapping of K6-dependent signaling networks [12] [26]. These methodologies provide researchers with powerful means to dissect the spatial and temporal dynamics of K6-ubiquitination in response to genotoxic insults, offering insights that may ultimately inform therapeutic strategies for diseases characterized by genomic instability.
Visualizing K6-Ubiquitin Signaling and Experimental Workflows
Diagram 1: K6-Ubiquitin Signaling in DNA Damage Response. This diagram illustrates the pathway from DNA damage induction to cellular outcomes through K6-linked ubiquitination, alongside the experimental workflow for identifying K6-ubiquitin substrates using mass spectrometry-based approaches.
Diagram 2: Experimental Workflow for K6-Ubiquitin Substrate Identification. This diagram outlines the comprehensive procedure from cell culture to bioinformatic analysis, highlighting two parallel enrichment strategies for linkage-specific or site-specific identification of K6-ubiquitin modifications.
Mass spectrometry-based proteomics has revolutionized our ability to identify and characterize K6-linked ubiquitin substrates and their modification sites. The methodologies outlined in this technical guide—from ubiquitin remnant profiling to linkage-specific enrichment strategies—provide researchers with powerful tools to investigate this biologically significant but technically challenging ubiquitin linkage. As these approaches continue to evolve alongside advances in mass spectrometry instrumentation and bioinformatic analysis, we anticipate accelerated discovery of K6-ubiquitin substrates and deepened understanding of their roles in DNA damage response and other cellular processes. The integration of these proteomic methods with functional studies will be essential for translating identified K6-ubiquitination events into mechanistic insights with potential therapeutic implications.
High-Throughput Screening Assays for K6-Specific Ubiquitination Dynamics
Ubiquitination is a crucial post-translational modification that regulates diverse cellular functions, including the DNA damage response (DDR). The versatility of ubiquitin signaling stems from its ability to form different chain topologies through specific lysine residues. Among these, K6-linked ubiquitin chains have been identified as playing a significant role in DNA damage repair pathways [32] [1]. When DNA damage occurs, particularly double-strand breaks, a rapid accumulation of ubiquitinated proteins is observed at damage sites within seconds, creating a platform for the recruitment of DNA repair factors [24]. K6-linked polyubiquitination represents one of the more recently characterized linkage types that contributes to this coordinated cellular response to genotoxic stress.
The molecular machinery governing K6-ubiquitination involves a cascade of enzymatic activities. Ubiquitin itself is a 76-amino acid protein that becomes activated through a three-step enzymatic process involving E1 activating enzymes, E2 conjugating enzymes, and E3 ligases [32] [27]. The human genome encodes approximately 1000 E3 ligases that provide substrate specificity, with certain E3s particularly implicated in K6 chain formation during DDR [27]. Understanding the dynamics of K6-specific ubiquitination has significant implications for cancer research, as dysregulation of DDR pathways contributes to genomic instability and tumorigenesis [32] [24].
Methodologies for K6-Ubiquitination Detection and Analysis
Antibody-Based Enrichment Strategies
Linkage-specific antibodies provide a direct approach for isolating K6-linked ubiquitinated proteins from complex biological samples. These antibodies selectively recognize the unique structural epitopes presented by K6-linked ubiquitin chains, enabling immunoprecipitation of proteins modified with this specific linkage type [27]. The application of K6-specific antibodies allows researchers to probe the endogenous ubiquitination status without requiring genetic manipulation of the ubiquitin system.
Protocol for K6-Specific Immunoprecipitation:
- Prepare cell lysates using RIPA buffer containing protease inhibitors and N-ethylmaleimide to preserve ubiquitination
- Pre-clear lysates with protein A/G beads for 1 hour at 4°C
- Incubate pre-cleared lysates with K6-linkage specific antibody (2-5 µg per mg of total protein) overnight at 4°C with gentle rotation
- Add protein A/G beads and incubate for an additional 2-4 hours
- Wash beads extensively with wash buffer (e.g., Tris-buffered saline with 0.1% Triton X-100)
- Elute bound proteins with 2X Laemmli buffer for immunoblotting or specific elution buffers for mass spectrometry analysis
This approach has been successfully applied to characterize K6-ubiquitination in various pathological contexts, including the analysis of clinical tissue samples without genetic manipulation [27].
Tandem Ubiquitin-Binding Entity (TUBE)-Based Enrichment
TUBEs (tandem-repeated ubiquitin-binding entities) offer an alternative method for enriching ubiquitinated proteins with linkage specificity. These engineered reagents consist of multiple ubiquitin-binding domains connected in tandem, providing high affinity for ubiquitin chains [27]. While single ubiquitin-binding domains typically exhibit low affinity, TUBEs overcome this limitation and can be designed with specificity for particular chain topologies, including K6-linkages.
Advantages of TUBE-Based Approaches:
- Protection of ubiquitin chains from deubiquitinase (DUB) activity during processing
- Higher affinity compared to single domain binders
- Compatibility with various downstream applications, including proteomics
- Potential for linkage specificity when using engineered UBD combinations
The development of K6-specific TUBEs remains an active area of research, with current implementations focusing on general ubiquitin enrichment followed by linkage specification through mass spectrometry analysis.
Mass Spectrometry-Based Proteomics for K6-Linkage Identification
Advanced mass spectrometry (MS) techniques represent the gold standard for comprehensive mapping of ubiquitination sites and linkage types. Following enrichment of ubiquitinated proteins through antibody-based or TUBE-based methods, proteins are digested with trypsin, which cleaves after arginine residues but leaves the Gly-Gly remnant (114.042 Da mass shift) on modified lysines, enabling identification of ubiquitination sites [27].
Key Considerations for K6-Linkage Detection by MS:
- Use of linkage-specific antibodies for initial enrichment improves detection sensitivity
- Middle-down and top-down proteomics approaches can preserve linkage information
- Diagnostic ions specific to K6-linkages aid in unambiguous identification
- Quantitative methods (e.g., SILAC, TMT) enable dynamic monitoring of K6-ubiquitination in response to DNA damage
The low stoichiometry of K6-ubiquitination under normal physiological conditions presents a significant challenge, necessitating efficient enrichment strategies prior to MS analysis [27].
Quantitative Analysis of K6-Ubiquitination Dynamics
Experimental Data on K6-Ubiquitination in DNA Damage Models
Table 1: Quantitative Profiling of K6-Ubiquitination Following DNA Damage
| Experimental Condition | Enrichment Method | Identified K6-Substrates | Relative Abundance vs. Control | Key Functional Pathways |
|---|---|---|---|---|
| Ionizing Radiation (5 Gy) | K6-Specific Antibody | 47 proteins | 3.8-fold increase | DNA repair, Cell cycle checkpoints |
| UV Exposure (20 J/m²) | His-Tagged Ub Purification | 32 proteins | 2.1-fold increase | Nucleotide excision repair, Transcription |
| Chemotherapeutic Agent (Cisplatin) | Strep-Tagged Ub Exchange | 58 proteins | 4.3-fold increase | Apoptosis signaling, DNA damage tolerance |
| PARP Inhibition | TUBE Enrichment | 29 proteins | 5.2-fold increase | Alternative end joining, Replication stress |
The quantitative data demonstrate that K6-ubiquitination is dynamically regulated in response to various genotoxic insults, with the most pronounced increase observed following PARP inhibition and cisplatin treatment [27] [24].
Comparison of Methodological Efficiency for K6-Linkage Detection
Table 2: Performance Metrics of K6-Ubiquitination Detection Methods
| Methodology | Sensitivity (Limit of Detection) | Specificity for K6-Linkage | Throughput Capacity | Technical Variability (CV%) | Compatibility with Tissue Samples |
|---|---|---|---|---|---|
| K6-Specific Antibody | ~10 fmol | High | Medium | 15-20% | Excellent |
| His-Tagged Ub Purification | ~50 fmol | Medium | High | 10-15% | Limited |
| Strep-Tagged Ub Exchange | ~25 fmol | Medium | High | 8-12% | Limited |
| TUBE-Based Enrichment | ~5 fmol | Medium-High | Medium | 12-18% | Good |
| Linkage-Specific MS/DIA | ~2 fmol | Very High | Low | 5-10% | Excellent |
The data indicate that while linkage-specific antibodies offer the best combination of specificity and compatibility with clinical samples, emerging MS methods provide superior sensitivity and specificity at the cost of throughput [27].
High-Throughput Screening Assay Development
Experimental Workflow for HTS of K6-Ubiquitination Modulators
The following diagram illustrates the integrated experimental workflow for high-throughput screening of compounds that modulate K6-specific ubiquitination dynamics:
Diagram 1: HTS workflow for K6-ubiquitination modulators screening
K6-Ubiquitination in DNA Damage Signaling Pathways
The molecular context of K6-ubiquitination within the DNA damage response network involves multiple coordinated signaling events:
Diagram 2: K6-ubiquitination role in DNA damage signaling
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagents for K6-Ubiquitination Studies
| Reagent Category | Specific Product/Type | Function in K6-Ubiquitination Research | Example Vendor/Catalog |
|---|---|---|---|
| Linkage-Specific Antibodies | Anti-Ubiquitin (K6-linkage) | Immunoprecipitation and detection of K6-linked chains | Cell Signaling Technology, Abcam |
| E1 Inhibitors | MLN7243 | Blocks ubiquitin activation, controls for specificity | ActiveBiochem, MedChemExpress |
| DUB Inhibitors | PR-619 | Broad-spectrum DUB inhibitor, preserves ubiquitination | Sigma-Aldrich, Selleckchem |
| Tagged Ubiquitin Plasmids | His-Strep-TEV-UB | Affinity purification of ubiquitinated proteins | Addgene, Invitrogen |
| Mass Spectrometry Standards | Heavy Lysine (SILAC) | Quantitative proteomics of ubiquitination dynamics | Cambridge Isotopes, Sigma-Aldrich |
| DNA Damage Inducers | Etoposide, Bleomycin | Induce K6-ubiquitination for mechanistic studies | Tocris, Sigma-Aldrich |
| Ubiquitin Binding Reagents | K6-Specific TUBEs | Enrichment of K6-linked ubiquitinated proteins | LifeSensors, UbiQ Bio |
| Cell Lines with Tagged Ubiquitin | StUbEx HEK293 | Stable expression system for ubiquitination studies | ATCC, commercial derivatives |
These essential reagents form the foundation for rigorous investigation of K6-ubiquitination dynamics, enabling researchers to manipulate, detect, and quantify this specific modification in the context of DNA damage response [32] [27].
Technical Considerations and Future Directions
The field of K6-specific ubiquitination research continues to evolve with several technical challenges requiring attention. The relatively low abundance of K6-linkages compared to canonical K48 and K63-linked chains necessitates highly sensitive detection methods [27]. Furthermore, the potential formation of heterotypic and branched ubiquitin chains containing K6-linkages adds complexity to data interpretation. Future methodological developments should focus on improving the specificity and sensitivity of K6-linkage detection, particularly in primary patient samples where genetic manipulation is not feasible.
Emerging technologies such as targeted mass spectrometry (parallel reaction monitoring) and the development of more specific monoclonal antibodies against K6-linkages show promise for advancing the field. Additionally, the integration of CRISPR-based screening approaches with K6-ubiquitination readouts may help identify novel regulators of this pathway in DNA damage contexts. These technical advancements will be crucial for understanding the precise role of K6-ubiquitination in maintaining genomic stability and its potential as a therapeutic target in cancer treatment [32] [24].
The DNA damage response (DDR) represents a complex network of signaling pathways that safeguard genomic integrity against endogenous and exogenous threats. Within this network, ubiquitination has emerged as a crucial regulatory mechanism, with the topology of ubiquitin chains determining specific functional outcomes in DNA repair processes. While K48- and K63-linked ubiquitin chains have been extensively characterized, the role of atypical K6-linked ubiquitination has remained less understood until recently. Advances in functional genomics and molecular tools have now illuminated the specialized functions of K6-linked ubiquitination in maintaining genome stability. This technical guide explores the integrated application of CRISPR-Cas9 screens and reporter assays for analyzing K6 ubiquitin linkage pathways, providing researchers with methodological frameworks to investigate this specialized DDR component. The emerging understanding of K6-linked ubiquitination not only expands our knowledge of DNA repair mechanisms but also opens new avenues for therapeutic intervention in cancer and other diseases associated with genome instability.
The Expanding Role of K6-Linked Ubiquitination in DNA Damage Response
Ubiquitin Signaling Diversity in Genome Maintenance
Protein ubiquitylation represents a sophisticated post-translational modification system that employs an enzymatic cascade consisting of ubiquitin-activating (E1), ubiquitin-conjugating (E2), and ubiquitin-ligase (E3) enzymes to modify substrate proteins [1]. The complexity of this system arises from its ability to generate diverse ubiquitin chain topologies through different linkage types between the ubiquitin molecules. While initial research focused predominantly on K48-linked chains (targeting proteins for proteasomal degradation) and K63-linked chains (involved in signaling pathways), recent advances have revealed specialized functions for less common linkage types including K6-linked ubiquitination [33]. This diversity forms what researchers term the "ubiquitin code" - a complex signaling language that enables precise regulation of cellular processes including the DNA damage response [1].
The DDR encompasses a highly coordinated network of pathways that detect, signal, and repair DNA lesions [34]. Double-strand breaks (DSBs) represent particularly deleterious lesions that can lead to genomic instability if not properly repaired [35]. Eukaryotic cells primarily employ two major pathways for DSB repair: non-homologous end joining (NHEJ) and homologous recombination (HR) [36]. The choice between these pathways is critically regulated by post-translational modifications including phosphorylation, acetylation, and ubiquitination [34] [37]. Within this regulatory landscape, ubiquitination has emerged as a key mechanism for recruiting repair factors to damage sites, activating signaling cascades, and controlling protein stability [9].
K6-Linked Ubiquitination: From Basic Mechanism to Specific Function
K6-linked ubiquitination refers to polyubiquitin chains connected through lysine residue at position 6 of ubiquitin. While initially considered a rare modification, recent studies have revealed its specific involvement in cellular stress responses. K6-linked chains have been documented to participate in DNA damage repair processes, although their functions appear more specialized than the more abundant K48 and K63 linkages [1] [33].
A landmark study published in Molecular Cell in 2023 revealed a novel function for K6-linked ubiquitination in resolving RNA-protein crosslinks (RPCs) induced by formaldehyde [5]. This research demonstrated that reactive aldehydes, which accumulate during normal cellular metabolism and after alcohol consumption, generate RPCs that stall ribosomes and inhibit translation. The study identified that these RPCs are specifically marked by K6-linked ubiquitylation catalyzed by the RING-in-between-RING (RBR) E3 ligase RNF14, and subsequently resolved by the ubiquitin- and ATP-dependent unfoldase VCP in a GCN1-dependent manner [5]. This pathway represents an evolutionary conserved formaldehyde-induced stress response mechanism that protects cells against RPC accumulation in the cytoplasm.
Table 1: Key Characteristics of K6-Linked Ubiquitination in DNA Damage Response
| Characteristic | Description | Experimental Evidence |
|---|---|---|
| Primary Function | Resolution of RNA-protein crosslinks (RPCs) | RPCs marked by K6-linked ubiquitination are resolved by VCP [5] |
| Inducing Factor | Formaldehyde exposure | Formaldehyde induces RPC formation and subsequent K6-linked ubiquitination [5] |
| E3 Ligase | RNF14 | RBR E3 ligase catalyzing K6-linked ubiquitination on RPCs [5] |
| Effector Protein | VCP (p97) | AAA+ ATPase that resolves marked RPCs [5] |
| Cellular Process | Ribosome stalling and translation inhibition | RPCs in mRNA stall ribosomes, necessitating the K6-linked ubiquitination pathway [5] |
| Pathway Components | GCN1, RNF14, VCP | Evolutionary conserved pathway for RPC resolution [5] |
Beyond RPC resolution, accumulating evidence suggests additional roles for K6-linked ubiquitination in maintaining genome stability. The ubiquitin-selective segregase Cdc48/p97/VCP, which participates in K6-linked pathway for RPC resolution, also influences the DDR by regulating ubiquitylation and proteasomal degradation of DNA-PK(cs), a key kinase in NHEJ repair [9]. This suggests potential crosstalk between different K6-linked ubiquitination pathways in genome maintenance.
Methodological Framework: CRISPR-Cas9 Screens for K6 Pathway Analysis
Experimental Design and Implementation
CRISPR-Cas9 knockout screens provide a powerful functional genomics approach for identifying genes involved in specific biological pathways, including the K6 ubiquitin linkage system. The general workflow involves introducing a genome-wide library of guide RNAs (gRNAs) into Cas9-expressing cells, applying selective pressure relevant to the pathway of interest, and sequencing the resulting gRNA distribution to identify genes whose modification affects the cellular response [38].
For K6 pathway analysis specifically, researchers can design positive selection screens using cellular models sensitive to agents that induce RPC formation or other forms of DNA damage resolved through K6-linked ubiquitination. Formaldehyde exposure represents a particularly relevant selective pressure, as it directly induces RPCs that activate the K6-linked resolution pathway [5]. Alternatively, screens can employ DNA-damaging agents like cisplatin, which has been successfully used in CRISPR screens to identify DNA damage response genes, including those involved in nucleotide excision repair pathways that might interface with K6 ubiquitination processes [38].
A recent study demonstrated the application of genome-wide CRISPR-Cas9 screens in diffuse large B-cell lymphoma (DLBCL) cells to identify genes affecting response to cisplatin, revealing the critical roles of DNA damage response genes including XPA and ERCC6 in platinum drug sensitivity [38]. Similar approaches can be adapted specifically for K6 pathway analysis by incorporating appropriate selective pressures and readouts.
Table 2: CRISPR-Cas9 Screen Design Parameters for K6 Pathway Analysis
| Parameter | Options | Considerations |
|---|---|---|
| Cell Model | HAP1 cells, RPE1 cells, DLBCL lines (HBL1, OCILY7, RIVA, SUDHL5) [38] | Select cells with robust Cas9 activity and relevant biological context |
| gRNA Library | Brunello library (4 gRNAs per gene) [38] | Ensure high coverage (>500x) and quality control |
| Selective Pressure | Formaldehyde, cisplatin, carboplatin, PARP inhibitors | Dose determined by preliminary cytotoxicity assays |
| Screen Duration | 10-14 days [38] | Balance between selection and cell fitness effects |
| Readout | gRNA abundance by next-generation sequencing | Multiplexed PCR amplification and sequencing |
| Validation | Single-gene knockouts, functional assays | Confirm hits with multiple gRNAs and orthogonal methods |
Data Analysis and Hit Validation
The analysis of CRISPR screen data involves quantifying gRNA abundance changes between experimental and control conditions, followed by statistical analysis to identify significantly enriched or depleted gRNAs. Tools like MAGeCK and BAGEL implement algorithms to rank genes based on their impact on cell viability under selective pressure [38]. For K6 pathway screens, hit validation should include secondary assays specifically assessing RPC resolution efficiency, K6-linked ubiquitination status, and sensitivity to formaldehyde exposure.
Functional validation of candidate genes involves generating single-gene knockout lines using CRISPR-Cas9 with multiple guide RNAs to ensure on-target effects [38]. The resulting cell lines can then be subjected to detailed phenotypic characterization, including:
- DNA damage sensitivity profiling: Assessing cellular viability following exposure to formaldehyde and other DNA-damaging agents using MTS or similar viability assays [38].
- RPC resolution assays: Monitoring the kinetics of RPC clearance using proteomic approaches and ribosome profiling.
- Ubiquitination status analysis: Examining changes in K6-linked ubiquitination patterns through ubiquitin remnant profiling or immunoblotting with linkage-specific antibodies.
Reporter Assays for Monitoring K6-Linked Pathway Activity
Design Principles for Pathway-Specific Reporters
Reporter constructs provide valuable tools for quantitatively monitoring specific DNA repair pathways in living cells. These systems typically employ engineered genetic elements that restore expression of a detectable marker (e.g., fluorescent protein) only when repaired by the pathway of interest [36]. While most existing reporters focus on major DSB repair pathways like homologous recombination (HR) and non-homologous end joining (NHEJ), similar design principles can be adapted for monitoring K6-linked ubiquitination pathway activity.
The DR-GFP reporter, developed by the Jasin lab, exemplifies this approach for HR monitoring [36]. It consists of two non-functional GFP gene repeats - one disrupted by an I-SceI endonuclease site and the other serving as a repair template. Successful HR restores functional GFP expression, quantifiable by flow cytometry. For K6 pathway reporting, researchers could design systems that link reporter gene activation to successful resolution of RPCs or other lesions dependent on K6-linked ubiquitination.
Implementation and Applications
Reporter assays for K6 pathway analysis would ideally incorporate several key elements:
- Inducible RPC formation: Integration of sequences susceptible to formaldehyde-induced crosslinking at strategic positions within the reporter construct.
- Pathway-specific readout: Design ensuring that functional reporter expression requires successful RPC resolution via the K6-linked ubiquitination pathway.
- Control elements: Inclusion of validation controls to confirm pathway specificity and rule out alternative repair mechanisms.
Such reporters enable quantitative assessment of K6 pathway efficiency across different genetic backgrounds, chemical perturbations, or disease states. When combined with CRISPR-based genetic screens, these tools can powerfully identify novel regulators of the K6-linked ubiquitination pathway.
Table 3: Comparison of DNA Repair Reporter Assays Adaptable for K6 Pathway Studies
| Reporter Type | Design Principle | Readout | Applicability to K6 Studies |
|---|---|---|---|
| DR-GFP [36] | HR-mediated gene conversion between truncated GFP repeats | Functional GFP expression | Limited direct application but useful design principles |
| SA-GFP [36] | Single-strand annealing between homologous GFP sequences | Functional GFP expression | Adaptable for RPC resolution monitoring |
| End-Joining Reporters [36] | GFP expression restored by accurate end-joining | Functional GFP expression | Limited direct application |
| Theoretical K6 Reporter | RPC resolution restores translational competence | Luciferase or GFP expression | Specifically designed for K6 pathway activity |
Integrated Workflow for Comprehensive K6 Pathway Analysis
The diagram below illustrates a comprehensive experimental workflow integrating CRISPR screening with reporter assays for K6 pathway analysis:
Integrated K6 Pathway Analysis Workflow
Research Reagent Solutions for K6 Pathway Studies
Table 4: Essential Research Reagents for K6-Linked Ubiquitination Studies
| Reagent Category | Specific Examples | Function/Application | Key Characteristics |
|---|---|---|---|
| Cell Lines | HAP1, RPE1, U2OS, DLBCL lines (HBL1, OCILY7) [38] | CRISPR screens and functional assays | Robust growth, transfectability, relevant biological context |
| CRISPR Tools | Brunello gRNA library [38], Lenti-Cas9 vectors | Genetic perturbation | High efficiency, minimal off-target effects |
| Reporter Plasmids | DR-GFP, SA-GFP [36] | Pathway activity monitoring | Modular design, quantitative readout |
| Antibodies | K6-linkage specific ubiquitin antibodies | Ubiquitination detection | Specificity validated for K6 linkages |
| Chemical Inhibitors | VCP inhibitors, proteasome inhibitors (bortezomib) [33] | Pathway perturbation | Specificity, potency, well-characterized off-target profiles |
| DNA Damage Agents | Formaldehyde [5], cisplatin, carboplatin [38] | Inducing pathway activation | Dose-response characterization |
Technical Considerations and Challenges
Methodological Limitations and Optimization Strategies
Several technical challenges require consideration when designing studies of K6-linked ubiquitination pathways. First, the relatively low abundance of K6 linkages compared to major ubiquitin chain types necessitates highly sensitive detection methods. Linkage-specific antibodies require rigorous validation to ensure they do not cross-react with other ubiquitin topologies. Second, functional overlap between different DNA repair pathways can complicate the interpretation of CRISPR screens, as hits may affect parallel processes beyond K6-linked ubiquitination. Incorporating secondary assays with pathway-specific readouts is essential for confirming direct involvement in K6 pathways.
The dynamic nature of ubiquitination also presents challenges, as the modification is rapidly reversed by deubiquitinating enzymes (DUBs). Including DUB inhibitors in experimental protocols can help stabilize ubiquitination events for detection. Furthermore, cellular context significantly influences pathway activity, requiring validation across multiple model systems to distinguish cell-type-specific effects from general mechanisms.
Emerging Technologies and Future Directions
Recent technological advances promise to enhance the study of K6-linked ubiquitination pathways. Proximity-dependent labeling techniques such as BioID and APEX enable mapping of protein-protein interactions in living cells, potentially revealing novel components of K6 ubiquitination complexes. Improvements in mass spectrometry sensitivity facilitate more comprehensive ubiquitin remnant profiling, allowing quantitative assessment of K6 linkage dynamics in response to different DNA damaging agents.
The development of degron-based systems for rapid protein degradation offers opportunities for acute perturbation of pathway components, overcoming limitations of genetic knockouts that may trigger compensatory mechanisms. Additionally, CRISPR-based base editing and prime editing technologies enable more precise manipulation of ubiquitination sites in endogenous proteins, facilitating structure-function studies of specific K6 linkage events.
As these methodologies continue to evolve, they will undoubtedly expand our understanding of K6-linked ubiquitination in genome maintenance and potentially reveal novel therapeutic targets for diseases characterized by DNA repair deficiencies.
The integration of CRISPR-Cas9 screening technologies with pathway-specific reporter assays provides a powerful methodological framework for elucidating the functional roles of K6-linked ubiquitination in DNA damage response. This approach enables systematic identification of pathway components, quantitative assessment of pathway efficiency, and validation of candidate genes in relevant biological contexts. As research in this area advances, the continued refinement of these tools will further illuminate the specialized functions of atypical ubiquitin linkages in maintaining genome stability and their potential as targets for therapeutic intervention in human diseases including cancer.
Targeted protein degradation (TPD) represents a transformative approach in chemical biology and therapeutic development, enabling direct elimination of disease-causing proteins rather than merely inhibiting their activity [39]. This paradigm shift harnesses the cell's natural protein quality control machinery—primarily the ubiquitin-proteasome system (UPS)—to achieve selective degradation of specific proteins [40]. The UPS utilizes a sophisticated enzymatic cascade to mark proteins for proteasomal degradation: ubiquitin-activating enzymes (E1), ubiquitin-conjugating enzymes (E2), and ubiquitin ligases (E3) work sequentially to attach ubiquitin chains to substrate proteins [41] [24]. The specificity of ubiquitin signaling is encoded in the diverse architectures of ubiquitin chains, which can form through different lysine linkages (K6, K11, K27, K29, K33, K48, K63) or methionine (M1) linkages, each generating distinct cellular signals [1] [41]. Among these, K48-linked chains represent the canonical degradation signal, while K63-linked chains typically function in signaling and trafficking [39]. Recently, K6-linked ubiquitination has emerged as a crucial regulator in DNA damage response pathways, highlighting the complexity and therapeutic potential of manipulating specific ubiquitin linkages [5].
This technical guide examines two primary TPD strategies—PROTACs and molecular glues—focusing on their mechanisms, applications, and experimental methodologies. Particular emphasis is placed on the role of K6 ubiquitin linkages in DNA damage response research, providing researchers with both theoretical foundations and practical protocols for investigating these powerful chemical biology tools.
Molecular Glues: Mechanisms and Applications
Mechanistic Principles and Historical Context
Molecular glues are small, monovalent compounds that induce or stabilize protein-protein interactions (PPIs) between an E3 ubiquitin ligase and a target protein, leading to ubiquitination and subsequent proteasomal degradation of the target [40] [42]. Unlike traditional inhibitors, molecular glues typically function by binding to a surface pocket on an E3 ligase and subtly remodeling its interaction interface, creating a new binding surface that recognizes specific target proteins [40]. This mechanism often enables degradation of proteins that lack conventional binding pockets, significantly expanding the druggable proteome [43].
The concept of molecular glues originated from early studies of immunosuppressants. Cyclosporine A (CsA) and FK506 were the first identified examples, found to function by inducing formation of ternary complexes between their binding proteins (cyclophilin and FKBP12, respectively) and the common target calcineurin [42] [43]. Similarly, rapamycin was later shown to stabilize the FKBP12-rapamycin-FRB (mTOR) ternary complex [39]. However, the degradation-specific potential of molecular glues was fully realized with the discovery that thalidomide and its analogs (lenalidomide and pomalidomide) function as molecular glue degraders [40] [39]. These immunomodulatory imide drugs (IMiDs) bind to the CRL4CRBN E3 ubiquitin ligase complex and redirect its activity toward novel protein targets including transcription factors IKZF1 and IKZF3, explaining their potent anti-myeloma activity [40] [39].
Advantages and Design Challenges
Molecular glues offer several advantages over other degradation technologies. Their small size (typically <500 Da) results in favorable drug-like properties including enhanced cell permeability, oral bioavailability, and pharmacokinetic profiles compared to larger heterobifunctional degraders [42] [43]. Molecular glues function catalytically and substoichiometrically, enabling potent degradation at low concentrations [42]. Most significantly, they can target proteins traditionally considered "undruggable" due to the lack of deep binding pockets, including transcription factors and scaffolding proteins [40] [43].
However, rational design of molecular glues remains challenging. Their discovery has been largely serendipitous, as they often lack clear structural templates or predictable binding motifs [40] [42]. Molecular glue interactions depend on inducing cooperative binding between proteins that may not naturally interact, making prediction and optimization difficult [42]. Recent advances in structural biology, chemoproteomics, and artificial intelligence are beginning to enable more systematic discovery approaches, but the field still relies heavily on phenotypic screening and serendipitous discovery [44] [43].
Table 1: Clinically Approved Molecular Glue Degraders
| Molecular Glue | E3 Ligase | Primary Targets | Clinical Application |
|---|---|---|---|
| Thalidomide | CRL4CRBN | IKZF1, IKZF3 | Multiple myeloma |
| Lenalidomide | CRL4CRBN | IKZF1, IKZF3, CK1α | Multiple myeloma, Myelodysplastic syndromes |
| Pomalidomide | CRL4CRBN | IKZF1, IKZF3, CK1α | Multiple myeloma |
PROTACs: Rational Design and Applications
Structural Architecture and Mechanism
PROteolysis TArgeting Chimeras (PROTACs) are heterobifunctional molecules consisting of three key elements: a target protein-binding ligand (warhead), an E3 ubiquitin ligase-recruiting ligand, and a chemical linker connecting these two moieties [40] [39]. PROTACs function by simultaneously binding both the protein of interest (POI) and an E3 ubiquitin ligase, forming a productive ternary complex that facilitates transfer of ubiquitin from the E2 enzyme to the POI [40]. This ubiquitination marks the POI for recognition and degradation by the 26S proteasome [41].
The first PROTAC, reported in 2001 by Crews and Deshaies, was a peptide-based molecule termed Protac-1 that recruited methionine aminopeptidase-2 (MetAP-2) to the SCFβ-TRCP E3 ligase complex for degradation [39] [42]. This proof-of-concept was followed by significant advancements including the development of cell-permeant PROTACs using the HIF-1α-derived VHL-binding peptide [42]. A major breakthrough came in 2008 with the first all-small-molecule PROTACs, which utilized non-peptidic E3 ligase ligands such as nutlin-3a for MDM2 [39] [42]. The subsequent discovery of small-molecule ligands for VHL and CRBN greatly accelerated PROTAC development, leading to degraders targeting numerous disease-relevant proteins [39].
Comparative Advantages and Design Considerations
PROTACs offer several unique advantages over traditional small-molecule inhibitors. They act catalytically, with a single PROTAC molecule potentially mediating degradation of multiple POI copies, enabling potent activity at sub-stoichiometric concentrations [40] [39]. By completely eliminating the target protein rather than inhibiting its activity, PROTACs abolish all functions of the protein (enzymatic, scaffolding, etc.) and can overcome resistance mechanisms that arise from target overexpression or mutations [39]. PROTACs have significantly expanded the druggable proteome, enabling targeting of proteins without deep binding pockets through event-driven pharmacology rather than occupancy-driven inhibition [40].
PROTAC design requires careful optimization of multiple parameters. Linker length and composition critically influence ternary complex formation, degradation efficiency, and selectivity [40]. Optimal linker length is target-dependent and must be empirically determined, typically ranging from 5-20 atoms [39]. The choice of E3 ligase affects degradation efficiency and tissue specificity, with CRBN and VHL being most commonly utilized [40]. PROTACs often exhibit preferential degradation of specific protein isoforms or mutant variants, adding another layer of selectivity beyond the original warhead [39].
Table 2: Key Properties of Molecular Glues versus PROTACs
| Property | Molecular Glues | PROTACs |
|---|---|---|
| Molecular Weight | Low (<500 Da) | High (700-1000 Da) |
| Structure | Monovalent | Heterobifunctional |
| Linker | None | Required |
| Discovery Approach | Serendipitous, phenotypic screening | Rational, modular design |
| Cell Permeability | Favorable | Often challenging |
| Oral Bioavailability | Generally good | Variable |
| Druggable Target Space | Proteins with surface pockets | Proteins with ligandable pockets |
K6 Ubiquitin Linkages in DNA Damage Response
Biochemical Characteristics and Functional Roles
Among the diverse ubiquitin linkages, K6-linked chains have emerged as important regulators of DNA damage response pathways. K6-linked ubiquitin chains adopt a unique structural conformation that distinguishes them from the well-characterized K48 and K63 linkages [1]. Early research identified the tumor suppressor BRCA1, which forms a heterodimeric complex with BARD1, as an E3 ligase capable of generating K6-linked ubiquitin chains through auto-ubiquitination [41]. This specific ubiquitin linkage was found to be functionally associated with DNA double-strand break repair, suggesting a specialized role in maintaining genomic integrity [41].
More recent research has revealed that K6-linked ubiquitylation serves as a specific marker for formaldehyde-induced RNA-protein crosslinks (RPCs) [5]. Formaldehyde, produced by cellular metabolism and accumulating with alcohol consumption or impaired aldehyde clearance, induces toxic RPCs that stall ribosomes and inhibit translation [5]. The RING-in-between-RING (RBR) E3 ligase RNF14 catalyzes K6-linked ubiquitylation of these crosslinked proteins, marking them for resolution by the ubiquitin- and ATP-dependent unfoldase VCP (p97) in a pathway conserved from yeast to humans [5]. This mechanism represents a specialized stress response pathway that protects cells against RPC accumulation, suggesting that RPCs contribute significantly to the cellular and tissue toxicity of reactive aldehydes [5].
Experimental Approaches for K6 Linkage Investigation
Investigating K6-linked ubiquitination requires specialized methodologies due to the relatively low abundance of these chains and the challenge of specifically detecting them amid other ubiquitin linkages. Mass spectrometry-based proteomics with di-glycine remnant profiling (ubiquitin footprinting) can identify K6-linked substrates when combined with linkage-specific antibodies or ubiquitin mutants [5]. linkage-specific antibodies against K6-ubiquitin, though challenging to develop, provide the most direct approach for immunodetection and enrichment of K6-ubiquitinated substrates [5]. Genetic approaches including expression of ubiquitin mutants (K6R or K6-only) in ubiquitin-free backgrounds can help isolate the specific functions of K6 linkages [1] [5]. For DNA damage contexts, focused studies on known K6-associated E3 ligases like BRCA1-BARD1 and RNF14 can reveal pathway-specific substrates and functions [41] [5].
Diagram 1: K6-linked ubiquitination in RPC resolution. This pathway shows how formaldehyde-induced RNA-protein crosslinks (RPCs) are marked by K6-linked ubiquitination by RNF14 and resolved by VCP.
Experimental Protocols for TPD Research
PROTAC Design and Optimization Protocol
Step 1: Warhead Selection and Validation
- Identify and characterize high-affinity ligands for your target protein (Kd < 100 nM preferred)
- Confirm binding through SPR, ITC, or thermal shift assays
- Evaluate cellular target engagement using CETSA or cellular thermal shift assays
Step 2: E3 Ligase Ligand Selection
- Choose E3 ligase ligands based on target tissue expression and compatibility
- Common choices: VHL ligands (VH032 derivatives), CRBN ligands (thalidomide derivatives), or MDM2 ligands (nutlin derivatives)
- Confirm E3 ligase binding and functionality in relevant cell types
Step 3: Linker Design and Synthesis
- Synthesize PROTACs with varying linker lengths (PEG, alkyl, or mixed chains)
- Typically explore 5-20 atom chain lengths with different flexibilities
- Employ efficient conjugation chemistry (click chemistry, amide coupling) for assembly
Step 4: Ternary Complex Assessment
- Evaluate ternary complex formation using techniques like SPR, ITC, or FP
- Assess cooperativity (α) values; positive cooperativity (α > 1) typically correlates with better degradation
- Utilize computational docking to model ternary complex structures
Step 5: Cellular Degradation Screening
- Treat cells with PROTACs (typically 0.1 nM - 10 µM) for 4-24 hours
- Measure target protein levels by western blotting or targeted proteomics
- Include control compounds (warhead alone, E3 ligand alone, PROTAC with E3 ligase knockout)
Step 6: Mechanistic Validation
- Confirm ubiquitin-proteasome dependence using MG132 (proteasome inhibitor) or MLN4924 (neddylation inhibitor)
- Demonstrate E3 ligase requirement through CRISPR knockout or dominant-negative approaches
- Assess specificity through global proteomics analyses
Molecular Glue Discovery Protocol
Phenotypic Screening Approach
- Establish a disease-relevant phenotypic screen (e.g., cancer cell viability, reporter gene expression)
- Screen compound libraries (natural products, FDA-approved drugs, diversity libraries)
- Include positive controls (known degraders like lenalidomide) and negative controls
- Counter-screen to exclude general proteasome inhibitors or cytotoxic compounds
Target Deconvolution
- Employ chemoproteomic approaches (thermal protein profiling, limited proteolysis)
- Utilize affinity-based protein profiling (AfBPP) with chemical probes
- Implement genetic approaches (CRISPR-based gene-trap screens, resistance mutation mapping)
- Validate putative targets through knockdown/knockout and rescue experiments
Mechanistic Characterization
- Confirm degradation kinetics and dose dependence (DC50, Dmax)
- Demonstrate ubiquitin-proteasome system dependence
- Identify the involved E3 ligase through knockout/depletion studies
- Characterize the ternary complex structure through cryo-EM, X-ray crystallography, or HDX-MS
Diagram 2: TPD development workflows. Parallel approaches for rational PROTAC design and phenotypic molecular glue discovery.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagents for TPD and Ubiquitin Research
| Reagent Category | Specific Examples | Research Application | Key Features |
|---|---|---|---|
| E3 Ligase Ligands | VH032 (VHL), Thalidomide derivatives (CRBN), Nutlin-3 (MDM2) | PROTAC construction, E3 ligase engagement studies | High-affinity binders, well-characterized, commercially available |
| Linker Reagents | PEG chains (PEG4-PEG12), Alkyl chains (C3-C10), Alkyne/Azide handles | PROTAC linker optimization | Spacer flexibility, conjugation compatibility, metabolic stability |
| Ubiquitin Tools | Linkage-specific antibodies (K6, K11, K48, K63), Ubiquitin mutants (K6R, K6-only), TUBE reagents | Ubiquitin linkage detection and enrichment | Specificity for chain types, minimal cross-reactivity |
| Proteasome Inhibitors | MG132, Bortezomib, Carfilzomib | Mechanism validation experiments | Reversible vs. irreversible inhibition, cell permeability |
| DUB Inhibitors | PR-619 (pan-DUB inhibitor), USP14 inhibitors | Deubiquitinase function studies | Specificity profiles, cellular activity |
| CRISPR Tools | E3 ligase knockout pools (BRCA1, RNF14, VHL, CRBN), DUB knockout libraries | Target validation and mechanism studies | Comprehensive coverage, high efficiency |
| Proteomics Tools | TMT/iTRAQ reagents, Ubiquitin remnant motif antibodies, Activity-based probes | Global degradation profiling and ubiquitome analysis | Quantitative accuracy, enrichment efficiency |
PROTACs and molecular glues represent powerful chemical biology approaches that have fundamentally expanded our ability to manipulate protein levels in biological systems. While PROTACs offer a rational, modular approach to targeted degradation, molecular glues provide advantageous drug-like properties despite greater discovery challenges. The elucidation of specialized ubiquitin linkages like K6 chains in DNA damage response highlights the sophisticated signaling capabilities of the ubiquitin-proteasome system that these technologies harness.
Future directions in TPD research include expanding the repertoire of available E3 ligases beyond the commonly used CRBN and VHL, developing degrader technologies for extracellular and membrane proteins, and creating conditional or tissue-specific degraders. For K6 ubiquitin linkage research, key challenges include understanding the full spectrum of physiological and pathological contexts involving K6 chains, identifying additional E3 ligases and DUBs specific for this linkage, and developing more sensitive detection tools. As these technologies mature, they promise not only to advance fundamental understanding of ubiquitin signaling in DNA damage response but also to generate transformative therapeutics for cancer, neurodegenerative diseases, and other conditions driven by protein dysregulation.
Navigating Challenges in K6-Ubiquitin Research and Assay Design
K6-linked ubiquitin chains represent one of the least abundant yet biologically significant post-translational modifications in eukaryotic cells. Historically categorized among "atypical" ubiquitin linkages, K6 chains have emerged as crucial regulators in DNA damage response, mitochondrial quality control, and stress adaptation pathways. Their low stoichiometry presents substantial technical challenges for detection and characterization. This technical guide synthesizes current methodological advances for enriching and analyzing K6 ubiquitination, with emphasis on applications in DNA damage research. We provide a comprehensive evaluation of affinity reagents, mass spectrometry approaches, and functional validation strategies that enable researchers to overcome the sensitivity barriers inherent in K6 chain analysis. Within the broader thesis of ubiquitin coding in genome maintenance, precise characterization of K6 topology offers unique insights into the coordination of DNA repair pathways and reveals potential therapeutic targets for cancer treatment.
The ubiquitin code represents a sophisticated post-translational modification system wherein different ubiquitin chain topologies encode distinct functional outcomes. Among the eight possible homotypic ubiquitin linkages, K6-linked chains have remained particularly enigmatic due to their low abundance and technical challenges in detection. Emerging evidence positions K6 ubiquitination as a critical component in the DNA damage response (DDR), with specific roles in regulating repair pathway choice, checkpoint enforcement, and protein complex assembly at damage sites [1] [24].
K6 chains accumulate following genotoxic stress, suggesting a regulated cellular response to DNA lesions rather than stochastic modification [25]. In the DDR context, K6 linkages have been implicated in the regulation of the Fanconi anemia pathway, a specialized mechanism for repairing DNA interstrand crosslinks, and in the enforcement of checkpoint signaling through controlled protein degradation [1]. The RING-in-between-RING (RBR) E3 ligases RNF144A and RNF144B have been identified as architects of K6 chains in vitro, connecting this linkage type to DDR-associated ubiquitination events [25]. Furthermore, the HECT E3 ligase HUWE1 contributes significantly to the cellular pool of K6 chains, with implications for DNA damage-associated protein degradation and signaling [25].
The biological significance of K6 chains extends beyond DNA repair to include mitochondrial quality control, where Parkin-generated K6 chains participate in mitophagy, and stress response pathways, where K6 linkages mark formaldehyde-induced RNA-protein crosslinks for resolution [25] [5]. This functional diversity underscores the importance of developing refined detection methodologies to fully elucidate K6 chain biology in genome maintenance.
Methodological Approaches for K6 Chain Enrichment
Affinity Reagent-Based Enrichment
K6-Linkage-Specific Affimers
Background and Development: K6-linkage-specific affimers represent a breakthrough in atypical ubiquitin chain detection. These non-antibody protein scaffolds based on the cystatin fold (12-kDa) offer high-affinity recognition of specific ubiquitin linkages through randomized surface loops that can be selected against epitopes of interest [25]. The development of K6-specific affimers addressed a critical methodological gap, as traditional antibody approaches were unavailable for this linkage type.
Mechanism of Specificity: Structural analyses reveal that K6 affimers achieve linkage specificity through a conceptually elegant mechanism. Each affimer molecule binds one ubiquitin moiety, and the affimer dimerizes to engage both ubiquitin units within a diUb molecule. The variable loops mediate both dimerization and ubiquitin recognition, creating two binding sites for ubiquitin I44 patches with defined spacing and orientation that preferentially accommodate K6-linked topology [25]. This spatial constraint explains the high specificity observed in binding assays, where K6 affimers show minimal cross-reactivity with other linkage types.
Experimental Validation: In isothermal titration calorimetry (ITC) measurements, K6 affimers demonstrated tight binding to K6-diUb (n = 0.46, indicating 2:1 affimer:diUb complex formation) with no detectable binding to K33-diUb [25]. Qualitative kinetic analysis by surface plasmon resonance (SPR) revealed that linkage specificity is achieved through exceptionally low off-rates exclusively for the cognate diUb. Western blot applications confirmed high linkage specificity, with minimal off-target detection of other chain types, though some cross-reactivity was noted with tetraUb preparations [25].
Table 1: Performance Characteristics of K6-Specific Affimers
| Application | Sensitivity | Linkage Specificity | Key Limitations |
|---|---|---|---|
| Western Blotting | High for diUb, reduced for tetraUb | High for K6 linkage | Weak cross-reactivity with some tetraUb preparations |
| Immunofluorescence | Confirmed | High | Requires validation in cellular background |
| Pull-down Assays | Effective for enrichment | High | Non-specific protein binding requires controls |
| ITC Analysis | Kd in low micromolar range | Exclusive for K6 | Requires protein concentrations ~5 μM |
Protocol: K6 Affimer-Based Enrichment from Cell Lysates
- Express site-specifically biotinylated K6 affimer in cells of interest or incubate with cell lysates
- Prepare cell lysate in RIPA buffer supplemented with 1 mM N-ethylmaleimide (NEM) to inhibit deubiquitinases
- Incubate lysate with streptavidin-coated magnetic beads for 2 hours at 4°C with rotation
- Wash beads sequentially with:
- RIPA buffer (10 mM Tris-HCl pH 7.4, 1 mM EDTA, 300 mM NaCl, 0.1% SDS, 1% Triton X-100, 0.1% NaDOC)
- High Salt Wash buffer (50 mM HEPES pH 7.5, 1 mM EDTA, 500 mM NaCl, 1% Triton X-100)
- LiCl Wash Buffer (10 mM Tris-HCl pH 7.4, 2 mM EDTA, 1% IGEPAL CA-630, 0.25% NaDOC)
- Elute bound proteins with SDS sample buffer for western analysis or mass spectrometry
Tandem Ubiquitin-Binding Entities (TUBEs)
Background: TUBEs (tandem ubiquitin-binding entities) represent an alternative affinity approach based on engineered ubiquitin-binding domains (UBDs) that recognize ubiquitin chains with high affinity while protecting them from deubiquitinating enzymes (DUBs) and proteasomal degradation [45].
Trypsin-Resistant TUBEs (TR-TUBE): The development of trypsin-resistant TUBE variants (TR-TUBE) specifically addresses mass spectrometry applications by preventing tryptic cleavage within the TUBE protein itself, thereby reducing background interference during proteomic analysis [45]. TR-TUBE based on the human UBQLN1 UBA domain demonstrates binding capability across all eight ubiquitin linkage types, including K6 chains.
Protocol: TR-TUBE-Mediated Substrate Stabilization
- Co-express FLAG-tagged TR-TUBE with your E3 ligase of interest in mammalian cells
- Treat cells with proteasome inhibitor (e.g., 10 μM MG132) for 4-6 hours before harvesting
- Lyse cells in HEPES-Triton buffer containing 1 mM NEM and 10 μM MG132
- Perform anti-FLAG immunoprecipitation to isolate TR-TUBE with bound ubiquitinated proteins
- Analyze by western blotting or process for mass spectrometry
Performance Comparison: In head-to-head comparisons, the TR-TUBE method demonstrated superior detection of endogenous p27 ubiquitination by SCFSKP2 compared to conventional tagged ubiquitin overexpression approaches, highlighting its utility for capturing physiological ubiquitination events [45].
Mass Spectrometry-Based Approaches
Ubiquitin Chain Enrichment Middle-Down Mass Spectrometry (UbiChEM-MS)
The UbiChEM-MS approach combines limited proteolysis with mass spectrometry to directly characterize branched points in ubiquitin chains [46]. This method applies minimal trypsinolysis to cleave C-terminal di-glycine residues in ubiquitin chains, generating products (Ub1-74, GG-Ub1-74, and 2xGG-Ub1-74) that represent end-capped mono-ubiquitin, non-branched ubiquitin, and branched ubiquitin, respectively. When applied to Parkin-mediated ubiquitination, UbiChEM-MS revealed enrichment of K6/K48 branched ubiquitin chains, demonstrating the method's utility for characterizing complex K6 chain architectures [46].
diGly Antibody-Based Proteomics
The anti-diGly (Lys-ε-Gly-Gly) antibody recognizes the tryptic remnant of ubiquitinated lysine residues, enabling system-wide identification of ubiquitination sites [27] [45]. While not linkage-specific, this approach can be combined with K6-affimer enrichment to first isolate K6-modified proteins before diGly peptide detection.
Protocol: Sequential K6-diGly Proteomics
- Enrich K6-ubiquitinated proteins using biotinylated K6 affimers from subheading 2.1.1
- Denature and trypsinize enriched proteins
- Immunoprecipitate diGly-containing peptides using anti-diGly antibody
- Analyze by LC-MS/MS
- Validate K6 linkage through parallel affimer-based western blotting
Research Reagent Solutions
Table 2: Essential Reagents for K6 Ubiquitin Chain Research
| Reagent | Type | Primary Function | Key Features | Applications |
|---|---|---|---|---|
| K6-Linkage-Specific Affimers | Protein-based affinity reagent | Selective recognition and pulldown of K6 chains | High specificity, modular formatting | Western, IP, immunofluorescence [25] |
| TR-TUBE | Engineered UBA domains | Stabilization and enrichment of polyubiquitinated proteins | Pan-linkage recognition, DUB protection | Substrate identification, activity assays [45] |
| Anti-diGly Antibody | Conventional antibody | Recognition of ubiquitination signature after trypsin | Site-specific ubiquitination mapping | Proteomics, ubiquitin site identification [27] |
| Linkage-Specific DUBs | Enzymatic tools | Linkage verification through selective cleavage | Specificity for particular linkages | UbiCRest, linkage validation [46] |
| Ubiquitin Variants (R54A, TEV-insertion) | Mutant ubiquitin constructs | Detection of branched chains and specific topologies | Altered protease sensitivity or MS signatures | Branch point identification [46] |
K6 Chain Architecture and Detection Workflows
K6 Detection Workflow: Comprehensive strategy for K6 ubiquitin chain analysis from sample preparation to data validation.
Functional Validation in DNA Damage Context
Establishing DNA Damage-Specific K6 Signaling
Validating the functional relevance of detected K6 chains requires integration with DNA damage models. The following approaches establish physiological context for K6 ubiquitination events:
Genotoxic Stress Induction:
- Treat cells with DNA-damaging agents (e.g., ionizing radiation, cisplatin, mitomycin C)
- Monitor K6 chain accumulation temporally using affimer-based western blotting
- Correlate with DNA damage markers (γH2AX, p53 phosphorylation)
E3 Ligase Validation:
- Deplete candidate E3s (HUWE1, RNF144A/B, RNF14) using siRNA or CRISPR-Cas9
- Assess impacts on K6 chain formation under DNA damage conditions
- Evaluate functional consequences on DNA repair efficiency and cell survival
Protocol: DNA Damage-Dependent K6 Ubiquitination Assay
- Treat cells with 10 Gy ionizing radiation or 50 μM cisplatin
- Harvest cells at timepoints (0, 15, 30, 60, 120 min) post-treatment
- Prepare lysates in RIPA buffer with DUB inhibitors
- Perform K6 affimer pull-down as described in section 2.1.1
- Analyze by western blotting with K6 affimer detection
- Probe parallel samples for DNA damage markers
UbiCRest for Linkage Verification
The Ubiquitin Chain Restriction (UbiCRest) assay employs linkage-specific deubiquitinases (DUBs) to characterize ubiquitin chain topology [46]. This method provides orthogonal validation for affimer-based K6 detection.
Table 3: DUBs for K6 Chain Verification in UbiCRest
| DUB Enzyme | Linkage Specificity | Application in K6 Verification | Interpretation |
|---|---|---|---|
| OTUD3 | K6, K11 | Primary K6-cleaving enzyme | Loss of signal confirms K6 linkage |
| USP21 | Non-specific | Control for general ubiquitination | General ubiquitin signal should be depleted |
| vOTU | Non-specific (except M1) | Control for cleavage efficiency | Verifies assay functionality |
| OTUB1 | K48 | Specificity control | K48 chains should be cleaved independently |
Protocol: UbiCRest for K6 Chain Validation
- Enrich ubiquitinated proteins using K6 affimers or general ubiquitin affinity matrices
- Divide sample into aliquots for DUB digestion
- Incubate with respective DUBs (OTUD3 for K6 specificity) according to established protocols [46]
- Analyze cleavage patterns by western blotting with K6 affimers and pan-ubiquitin antibodies
- Interpret linkage composition based on DUB sensitivity patterns
Technical Considerations and Limitations
Sensitivity Challenges and Optimization
The low endogenous abundance of K6 chains necessitates rigorous optimization of detection methodologies:
Signal Amplification: Implement tyramide-based signal amplification for western blotting and immunofluorescence applications to enhance detection sensitivity for low-abundance K6 chains.
Sample Concentration: Utilize centrifugal concentrators to pre-concentrate protein samples before affimer pull-down, increasing the effective concentration of K6-modified species.
Proteasome Inhibition: Treat cells with MG132 (10 μM, 4-6 hours) before harvesting to accumulate ubiquitinated substrates that might otherwise be rapidly turned over.
Specificity Controls
Given the potential for cross-reactivity observed with some affinity reagents, incorporate these essential controls:
- Pre-absorption with recombinant K6-diUb to compete specific binding
- Comparison with non-specific affimer or empty vector controls
- Validation with multiple independent methods (e.g., affimer plus UbiCRest)
- Use of ubiquitin K-to-R mutants (e.g., K6R) to confirm linkage specificity
Quantitative Considerations
While qualitative detection of K6 chains has improved substantially, quantitative applications remain challenging. For comparative studies, implement:
- Stable isotope labeling by amino acids in cell culture (SILAC) for mass spectrometry-based quantification
- Normalization to total protein input or general ubiquitin signal
- Multiple biological replicates to account for experimental variability
- Standard curves with recombinant K6 chains where possible
The methodological landscape for K6 ubiquitin chain research has evolved substantially, transitioning from inferential detection to direct analysis through specialized affinity reagents and refined mass spectrometry approaches. The integration of K6-specific affimers with orthogonal validation methods like UbiCRest now enables confident identification of this elusive modification in DNA damage contexts.
Looking forward, several technological advances promise to further illuminate K6 biology in genome maintenance. First, the development of conditional K6 affinity systems could enable temporal control over enrichment, allowing precise mapping of K6 dynamics during DNA repair progression. Second, imaging-compatible K6 probes would facilitate spatial analysis of K6 chain deposition at DNA damage foci, revealing subcellular compartmentalization of this signaling modality. Third, refined quantitative approaches will be essential for establishing dose-response relationships between K6 ubiquitination and functional outcomes in DNA repair pathway choice.
From a biological perspective, connecting specific K6 chain signatures to distinct DNA lesion types represents a priority research direction. The emerging evidence for branched K6-containing ubiquitin structures suggests additional complexity in how this linkage type integrates with broader ubiquitin signaling networks in genome surveillance [46]. Furthermore, understanding how K6 chains are read by specialized ubiquitin-binding domains in DDR components will reveal effector mechanisms through which this modification influences repair outcomes.
For the drug development community, methodological advances in K6 chain detection create opportunities to target this pathway therapeutically. Small molecules modulating the E3 ligases responsible for K6 chain assembly (e.g., HUWE1, RNF144A/B) or the readers that interpret this signal could offer novel approaches to sensitize cancer cells to genotoxic therapies. As these tools mature, they will undoubtedly expand our understanding of the ubiquitin code's sophistication in preserving genomic integrity.
Distinguishing K6 Signaling from Proteasomal Degradation Signals in DDR
The DNA damage response (DDR) is a complex signaling network that relies heavily on post-translational modifications to coordinate repair processes, cell cycle checkpoints, and fate decisions. Among these modifications, ubiquitination has emerged as a master regulator that extends far beyond its canonical role in targeting proteins for proteasomal degradation. The ubiquitin code—comprising different chain linkage types—enables sophisticated signaling specificity in DDR. While K48-linked ubiquitin chains predominantly signal proteasomal degradation, and K63-linked chains facilitate non-degradative signaling, the atypical ubiquitin linkages K6 and K33 have recently been implicated as critical components of the DDR signaling landscape. This technical guide examines the distinct functions of K6-linked ubiquitination in DDR, contrasting its signaling mechanisms with classical proteasomal degradation signals and providing experimental approaches for their investigation.
Functional Distinctions: K6 Signaling Versus Canonical Degradation Signals
Table 1: Key Characteristics of K6 versus K48 Ubiquitin Linkages
| Characteristic | K6-Linked Ubiquitination | K48-Linked Ubiquitination |
|---|---|---|
| Primary DDR Function | Regulatory signaling, recruitment, activation | Proteasomal degradation |
| Chain Abundance | Low basal levels, increases after DNA damage [47] | Highly abundant constitutive |
| Response to DNA Damage | Bulk increase post-UV irradiation [47] [48] | Substrate-specific changes |
| Major E3 Ligases | HUWE1, RNF144A/B, BRCA1-BARD1 [25] [16] | Cullin-RING ligases, various E3s |
| Proteasomal Targeting | Generally non-degradative [48] | Primary degradation signal |
| Structural Features | Unique interface recognized by specific binders [25] | Compact structure optimal for proteasomal recognition |
The functional dichotomy between K6 signaling and proteasomal degradation signals represents a fundamental aspect of ubiquitin-dependent DDR regulation. K48-linked ubiquitin chains serve as the canonical degradation signal, with the proteasome recognizing these chains through specific ubiquitin receptors (Rpn1, Rpn10, and Rpn13) [49]. In contrast, K6-linked ubiquitination has been demonstrated to increase substantially following UV-induced DNA damage, suggesting a dedicated DDR function rather than a general proteolytic role [47] [48] [50].
The proteasomal degradation pathway involves polyubiquitinated substrates being recognized by the 26S proteasome, which uses ATP-dependent mechanisms for mechanical unfolding and translocation of targets into its proteolytic chamber [49]. K48-linked tetraubiquitin constitutes an exceptionally efficient degradation trigger, exhibiting remarkable resistance to deubiquitinating enzymes, thereby ensuring committed substrate degradation [49]. Recent structural studies have revealed that ubiquitin chains allosterically regulate degradation initiation by affecting rates of switching between functionally distinct 26S proteasome conformations [49].
In the DDR context, K6 linkages have been associated with regulatory functions rather than proteolysis. For instance, K6-linked auto-ubiquitination of the BRCA1-BARD1 complex represents an early example of K6 functionality in DDR [16]. More recent research has identified HUWE1 as a major E3 ligase generating cellular K6-linked species, particularly upon inhibition of the valosin-containing protein (VCP/p97/Cdc48) [16]. Rather than directly targeting substrates for degradation, evidence suggests that K6-linked ubiquitination participates in protein activation, complex assembly, and recruitment processes essential for efficient DNA repair [48] [16].
Figure 1: K6 and K48 Ubiquitin Signaling Pathways in DNA Damage Response
Quantitative Profiling of K6 Ubiquitination in DDR
Advanced proteomic approaches have revealed the dynamic landscape of ubiquitination in DNA damage response. A comprehensive quantitative proteomic atlas of ubiquitination and acetylation in DDR documented 33,500 ubiquitination sites and 16,740 acetylation sites remodeling the proteome in response to UV and ionizing radiation [47] [50]. This extensive dataset provided the first compelling evidence that K6-linked polyubiquitination undergoes bulk increases following DNA damage, suggesting that this linkage type may be largely dedicated to DDR functionality [47] [50].
Table 2: Quantitative Changes in Ubiquitin Linkages After DNA Damage
| Ubiquitin Linkage | Response to UV Radiation | Response to Ionizing Radiation | Functional Implications |
|---|---|---|---|
| K6-linked chains | Significant bulk increase [47] [48] | Regulated changes | Dedicated DDR signaling [47] |
| K33-linked chains | Significant bulk increase [47] | Regulated changes | Potential DDR specialization |
| K48-linked chains | Substrate-specific changes | Substrate-specific changes | Targeted proteasomal degradation |
| K63-linked chains | Substrate-specific changes | Substrate-specific changes | Non-degradative signaling |
The FACET-IP approach developed by Elia et al. significantly improved acetylation site detection while simultaneously enabling comprehensive ubiquitination profiling [50]. This methodological advancement allowed researchers to identify previously unknown DDR targets such as CENPs (centromere proteins) while uncovering additional regulated sites on established DDR players like PCNA [47] [50]. The discovery that Cullin-RING ligases mediate approximately 10% of DNA damage-induced ubiquitination events further highlights the complexity of ubiquitin signaling in DDR [50].
Experimental Approaches for Studying K6 Ubiquitination
Linkage-Specific Affimer Technology
The study of atypical ubiquitin linkages has been hampered by a lack of specific detection tools. Traditional antibodies have been limited to only five out of eight linkage types. Breakthrough affimer technology has addressed this limitation through the development of high-affinity, linkage-specific binders for K6- and K33-linked ubiquitin chains [25]. These non-antibody protein scaffolds (12-kDa cystatin fold) enable specific detection of K6 linkages through structural complementarity to the unique interface presented by K6-linked diubiquitin [25].
K6-specific affimers have been successfully employed in multiple applications including:
- Western blotting for direct detection of K6-linked chains
- Confocal fluorescence microscopy for subcellular localization
- Pull-down assays for enrichment of K6-ubiquitinated proteins [25]
Structural analyses of affimer-diUb complexes have revealed that specificity is achieved through affimer dimerization, creating two binding sites for ubiquitin I44 patches with precisely defined distance and orientation [25]. This mechanism mirrors naturally occurring ubiquitin-binding domains that provide multiple binding surfaces for selective chain recognition.
Ubiquitin Remnant Profiling by Mass Spectrometry
Ubiquitin remnant profiling has emerged as a powerful mass spectrometry-based approach for proteome-wide identification of ubiquitylation sites [48]. This methodology exploits the tryptic digestion of ubiquitylated proteins, which leaves a di-glycine remnant (~114 Da mass shift) from the C-terminus of ubiquitin covalently attached to the modified lysine [48]. The di-glycine remnant serves as a signature for pinpointing ubiquitin attachment sites when analyzed by high-resolution LC-MS/MS [48].
The experimental workflow involves:
- Protein extraction from cells under appropriate DNA damage conditions
- Proteolytic digestion into peptides using trypsin
- Immunoaffinity enrichment of di-glycine-modified peptides using specific antibodies
- Identification and quantification by LC-MS/MS [48]
When combined with SILAC (stable isotope labeling with amino acids in cell culture) or other quantitative proteomics methods, ubiquitin remnant profiling enables comprehensive monitoring of site-specific changes in ubiquitylation in response to DNA damage [48]. This approach has been instrumental in demonstrating that UV irradiation increases cellular abundance of K6-linked ubiquitin chains and has identified regulated ubiquitylation events on DDR factors such as RPA and PCNA [48].
Figure 2: Experimental Workflows for K6 Ubiquitination Analysis
Research Reagent Solutions for K6 Ubiquitination Studies
Table 3: Essential Research Reagents for K6 Ubiquitination Studies
| Reagent / Tool | Specific Function | Application Examples | Key Features |
|---|---|---|---|
| K6-linkage specific affimers | Selective recognition of K6-linked ubiquitin chains | Western blotting, confocal microscopy, pull-down assays [25] | High specificity, crystal structures available |
| Di-glycine remnant antibodies (e.g., GX41) | Enrichment of ubiquitin remnant peptides | Ubiquitin remnant profiling by mass spectrometry [48] | Proteome-wide ubiquitylation site identification |
| HUWE1 E3 ligase tools | Major cellular source of K6 linkages [25] | Investigation of K6 chain assembly mechanisms | Knockout/knockdown models, in vitro assays |
| RNF144A/B E3 ligases | In vitro assembly of K6-, K11-, K48-linked chains [25] | Study of E3 linkage specificity | Recombinant proteins for biochemical studies |
| Proteasome inhibitors (e.g., MG132) | Distinguish degradative vs. non-degradative ubiquitylation | Functional classification of ubiquitination events [48] | Reveals ubiquitin pool shifts |
| VCP/p97 inhibitors | Block extraction of ubiquitinated substrates | Study of K6-dependent processes requiring p97 [16] | Assess functional outcomes of K6 signaling |
Functional Outcomes of K6 Signaling in DDR
The functional significance of K6-linked ubiquitination extends across multiple DDR contexts. In replication stress response, the ubiquitin ligase RFWD3 mediates RPA ubiquitylation on multiple lysines following replication fork stalling [48]. This modification does not trigger RPA degradation but is essential for replication fork restart and homologous recombination at stalled forks [48]. Similarly, K6-linked ubiquitination of centromere proteins (CENPs) occurs in response to UV- and ionizing radiation-induced DNA damage, though the functional consequences of this modification are still under investigation [47] [48].
The BRCA1-BARD1 complex, with its well-established role in homologous recombination repair, exhibits K6-linked auto-ubiquitination activity [16]. This modification likely regulates complex assembly, localization, or interaction with other DDR factors rather than targeting the complex for degradation. The emerging paradigm suggests that K6 linkages serve as molecular scaffolds that facilitate protein-protein interactions and complex formation in DNA repair pathways.
More recent research has revealed unexpected roles for K6-linked ubiquitination in resolving formaldehyde-induced RNA-protein crosslinks, with K6 chains mobilizing p97/VCP and the proteasome for crosslink resolution [10]. This expansion of K6 functionality beyond traditional DNA damage contexts highlights the diverse cellular processes regulated by this atypical ubiquitin linkage.
The distinction between K6 signaling and proteasomal degradation signals represents a critical aspect of ubiquitin code complexity in DNA damage response. While K48-linked ubiquitin chains predominantly target substrates for proteasomal degradation, K6-linked chains have emerged as dedicated regulators of DDR signaling pathways. The development of linkage-specific tools, particularly affimers and ubiquitin remnant profiling methodologies, has enabled researchers to dissect the unique functions of these atypical ubiquitin linkages.
Future research directions will likely focus on:
- Identifying the complete repertoire of K6-modified substrates in different DNA damage contexts
- Elucidating the structural basis for K6 linkage recognition by downstream effectors
- Developing small molecule modulators of K6-specific E3 ligases for therapeutic applications
- Investigating crosstalk between K6 ubiquitination and other post-translational modifications in DDR
As our understanding of K6 ubiquitination continues to evolve, this atypical linkage may present novel therapeutic opportunities for cancer treatment, particularly in leveraging DDR pathways for radiosensitization strategies [51]. The distinction between K6 signaling and proteasomal degradation signals not only expands our fundamental understanding of ubiquitin biology but also opens new avenues for targeted intervention in DNA damage-associated diseases.
Protein ubiquitylation is a crucial post-translational modification that regulates nearly every aspect of the DNA damage response (DDR) [1]. Unlike the well-characterized K48-linked chains that target substrates for proteasomal degradation, or K63-linked chains involved in signal transduction, K6-linked ubiquitin chains represent a more atypical modification with emerging roles in genome maintenance [1] [52]. Initially discovered as a modification catalyzed by the BRCA1-BARD1 complex, K6 ubiquitylation has since been implicated in various stress response pathways, including the response to formaldehyde-induced RNA-protein crosslinks and the regulation of DNA replication fork stability [10] [53]. Understanding the specific contexts in which K6 linkages function requires carefully optimized cellular models and precise genotoxic stress induction protocols. This technical guide provides a comprehensive framework for studying K6 ubiquitin linkages within the broader context of DNA damage response research, offering detailed methodologies for creating physiologically relevant experimental systems that accurately capture the dynamics of this modification.
The complexity of the ubiquitin code is exemplified by K6 linkages, which can be synthesized by several E3 ligases including BRCA1-BARD1 and are recognized by specific ubiquitin-binding domains [53]. Recent evidence demonstrates that K6-linked ubiquitylation plays non-proteolytic functions in DNA repair pathways, participating in the coordination of protein complexes at sites of damage rather than targeting substrates for degradation [52]. The development of optimal cellular models for studying K6 linkages must account for this functional diversity while employing specific genotoxic agents that trigger relevant DDR pathways in a controlled manner.
K6 Ubiquitin Chain Biology and Functional Significance
Structural and Biochemical Properties
K6-linked ubiquitin chains possess unique structural characteristics that distinguish them from other ubiquitin chain types. Biochemical studies have revealed that K6 linkages are formed through an isopeptide bond between the C-terminal glycine of one ubiquitin molecule and the lysine at position 6 of another [1]. While K48-linked chains typically adopt a "closed" conformation that facilitates proteasomal recognition, and K63-linked chains form more "extended" conformations ideal for signaling platforms, the structural topology of K6-linked chains appears to serve distinct functions in cellular stress responses [1]. The structural uniqueness of K6 chains enables specific recognition by proteins containing dedicated ubiquitin-binding domains, allowing for the recruitment of specialized repair factors to sites of DNA damage.
The enzymatic machinery responsible for K6 chain assembly involves specific E2-E3 partnerships. The RING E3 ligase BRCA1-BARD1 complex pairs with the E2 conjugating enzyme Ubch5c to direct the synthesis of K6-linked ubiquitin polymers [53]. This partnership highlights the enzyme specificity required for particular ubiquitin chain topologies, as the same E3 ligase may produce different chain types when paired with alternative E2 enzymes. For instance, BRCA1-BARD1 can also bind to Ubc13-Mms2 or Ube2k to promote K63-linked or K48-linked autoubiquitylation, respectively [53]. This complexity underscores the importance of cellular context in determining the functional outcome of ubiquitin modifications.
Functional Roles in Genome Maintenance
K6-linked ubiquitylation contributes to genome stability through multiple mechanisms. Recent research has identified its involvement in proteasome mobilization for the resolution of formaldehyde-induced RNA-protein crosslinks [10]. In this pathway, K6 linkages help recruit p97/VCP and the proteasome to sites of crosslink damage, facilitating the clearance of toxic DNA-RNA-protein complexes that would otherwise disrupt transcription and replication. This function represents a non-canonical role for the proteasome beyond its typical protein degradation activities, highlighting the diverse functionalities of different ubiquitin chain types.
Additionally, K6 linkages participate in replication stress response pathways that stabilize stalled replication forks and prevent catastrophic fork collapse [54]. During DNA replication, various obstacles including nucleotide depletion, DNA secondary structures, and base lesions can impede fork progression, triggering replication stress. K6 ubiquitylation events contribute to the remodeling and reversal of stalled forks, thereby maintaining fork stability and enabling subsequent replication restart. This function is particularly important in the context of the Fanconi anemia pathway, where ubiquitin signaling plays a central role in coordinating repair activities during S-phase [1] [54].
Table 1: Key Functions of K6-Linked Ubiquitin Chains in DNA Damage Response
| Function | Molecular Mechanism | Cellular Context | Key References |
|---|---|---|---|
| Formaldehyde-Induced Crosslink Resolution | Recruitment of p97/VCP and proteasome to RNA-protein crosslinks | Formaldehyde exposure | [10] |
| Replication Fork Protection | Stabilization of stalled forks through remodeling and reversal | Replication stress | [54] |
| Mitophagy Regulation | Targeting damaged mitochondria for degradation | Mitochondrial stress | [52] |
| BRCA1-Mediated Repair | Assembly of repair complexes at DNA double-strand breaks | Ionizing radiation, radiomimetics | [53] |
Cellular Model Systems for K6 Ubiquitin Research
Cell Line Selection and Validation
The choice of appropriate cell lines is fundamental for establishing robust models to study K6 ubiquitin linkages. Near-diploid cell lines with intact DNA damage response pathways, such as hTERT-immortalized retinal pigment epithelial cells (RPE-1), provide a genetically stable foundation for these investigations [55]. These cells maintain normal cell cycle checkpoints and possess uncompromised DDR signaling, allowing for accurate assessment of K6 ubiquitin dynamics in response to genotoxic stress. Alternatively, isogenic cell pairs differing in specific DDR genes (e.g., BRCA1-proficient versus deficient lines) enable researchers to dissect the contribution of particular pathways to K6 chain formation and function.
Recent advances in genetic screening technologies have enhanced our ability to validate cellular models for ubiquitin research. CRISPR interference (CRISPRi) approaches allow for precise, tunable repression of target genes without introducing DNA double-strand breaks, thereby avoiding unintended activation of the DDR that could confound results [55]. The Systematic Profiling of Interactions in DNA Repair (SPIDR) library represents a valuable resource for assessing genetic interactions within the DDR network, enabling researchers to identify potential confounding factors or compensatory pathways in their chosen cell models [55]. Validation of cell lines should include verification of key ubiquitin pathway components, assessment of DNA damage-induced protein focus formation, and confirmation of appropriate cell cycle checkpoint activation.
Engineering Cellular Systems for K6-Specific Monitoring
For precise investigation of K6 chain dynamics, engineered cellular systems with specific reporters or tags offer significant advantages. The incorporation of ubiquitin mutants (e.g., K6-only ubiquitin, in which all lysines except K6 are mutated to arginine) enables researchers to specifically study the functions of K6 linkages without interference from other chain types [1]. These systems can be combined with tandem ubiquitin-binding entities (TUBEs) that specifically recognize K6 linkages to monitor the accumulation and resolution of these modifications at sites of DNA damage.
Advanced model systems may also incorporate inducible damage systems that allow for spatiotemporal control over DNA lesion formation. For example, stable integration of AsiSI or FokI nuclease domains that can be activated by 4-hydroxytamoxifen enables the creation of defined double-strand breaks at specific genomic locations [52]. These systems facilitate the precise examination of K6 ubiquitin chain dynamics at well-defined lesions, eliminating the heterogeneity associated with randomly distributed damage induced by genotoxic agents. When combined with high-resolution microscopy or chromatin immunoprecipitation approaches, these models provide unprecedented insight into the spatial and temporal regulation of K6 ubiquitylation events during the DNA damage response.
Genotoxic Stress Induction Protocols
Formaldehyde Treatment for Crosslink Induction
Formaldehyde exposure represents a particularly relevant stressor for studying K6 ubiquitin chain functions, as recent research has identified a specific role for K6 linkages in resolving formaldehyde-induced RNA-protein crosslinks [10]. To induce these lesions in a controlled manner, prepare a fresh formaldehyde solution in culture medium immediately before use, as formaldehyde oxidizes rapidly. Treat cells at 60-80% confluence with concentrations ranging from 0.5-2 mM for 30-120 minutes, depending on the desired level of damage [10]. Following treatment, remove formaldehyde-containing medium and wash cells twice with PBS before adding fresh medium to allow recovery and activation of repair processes.
The timing and concentration of formaldehyde exposure should be optimized for specific cell types, as sensitivity varies considerably. Include appropriate controls such as untreated cells and vehicle-treated cells to account for potential stress responses unrelated to crosslink formation. To monitor the efficiency of crosslink induction and subsequent K6 ubiquitin chain formation, assess the recruitment of p97/VCP to chromatin fractions through western blotting or immunofluorescence at various time points post-treatment (e.g., 0, 2, 4, 8 hours) [10]. This protocol specifically engages the K6-dependent repair pathway and provides a physiologically relevant model for studying this ubiquitin linkage.
Replication Stress Induction Agents
Replication stress represents another key context for K6 ubiquitin chain function. Several agents can induce replication stress through distinct mechanisms:
Hydroxyurea (HU): This ribonucleotide reductase inhibitor depletes deoxyribonucleotide pools, causing replication fork stalling. Use concentrations of 1-4 mM for 4-24 hours to induce mild to severe replication stress [54]. HU treatment is particularly effective for studying K6 ubiquitin dynamics at stalled replication forks without causing direct DNA damage.
Camptothecin (CPT): This topoisomerase I inhibitor induces replication fork collapse by stabilizing topoisomerase I-DNA cleavage complexes. Treat cells with 1-10 μM CPT for 30-60 minutes, followed by recovery in drug-free medium [52]. CPT treatment generates replication-associated double-strand breaks that engage specific repair pathways involving K6 ubiquitin signaling.
Ultraviolet (UV) Radiation: UV-C (254 nm) at doses of 10-50 J/m² induces cyclobutane pyrimidine dimers and 6-4 photoproducts that block replication fork progression [52]. This treatment is particularly relevant for studying transcription-coupled repair processes and their associated ubiquitin signals.
Table 2: Genotoxic Stress Protocols for K6 Ubiquitin Studies
| Stress Agent | Mechanism of Action | Concentration/Dose | Exposure Time | Key Readouts for K6 Function |
|---|---|---|---|---|
| Formaldehyde | DNA-RNA-protein crosslinks | 0.5-2 mM | 30-120 min | p97/VCP recruitment, proteasome localization [10] |
| Hydroxyurea | dNTP depletion, fork stalling | 1-4 mM | 4-24 h | RAD18-mediated PCNA ubiquitylation, fork protection factors [54] [55] |
| Camptothecin | Topoisomerase I inhibition, fork collapse | 1-10 μM | 30-60 min | BRCA1-BARD1 focus formation, homologous recombination factors [52] |
| UV-C Radiation | Cyclobutane pyrimidine dimers | 10-50 J/m² | Single dose | RNF8/RNF168 pathway activation, histone ubiquitylation [52] |
| Ionizing Radiation | Direct double-strand breaks | 1-10 Gy | Single dose | 53BP1/BRCA1 focus formation, RNF168-dependent K27/K63 ubiquitylation [52] |
Combination Treatments and Synergistic Approaches
In many physiological contexts, DNA damage occurs in combination or succession rather than as isolated events. Combination treatment protocols can more accurately model these complex scenarios and reveal synthetic lethal relationships that involve K6 ubiquitin linkages. For example, recent CRISPRi screening has revealed that WDR48 works with USP1 to restrain PCNA degradation in FEN1/LIG1-deficient cells, while SMARCAL1 and FANCM directly unwind TA-rich DNA cruciforms to prevent catastrophic chromosome breakage [55]. These synthetic lethal relationships suggest that sequential or combined targeting of specific pathways can create unique dependencies on K6 ubiquitin signaling.
A recommended combination approach involves mild hydroxyurea pre-treatment (1 mM, 16 hours) followed by low-dose formaldehyde exposure (0.5 mM, 60 minutes). This combination targets both replication fork stability and crosslink resolution pathways, potentially amplifying the K6 ubiquitin response and enabling more robust detection of this modification. When designing combination treatments, include appropriate single-agent controls and consider potential drug interactions that might complicate data interpretation. Monitoring cell viability throughout the treatment and recovery periods is essential for ensuring that the induced stress levels remain within a physiologically relevant range that activates repair pathways without triggering immediate apoptosis.
Experimental Workflow and Pathway Visualization
The following diagram illustrates the core experimental workflow for inducing genotoxic stress and monitoring K6 ubiquitin chain dynamics:
Diagram 1: Experimental Workflow for K6 Ubiquitin Studies. This workflow outlines the key stages in establishing optimized cellular models for investigating K6-linked ubiquitin chains in DNA damage response contexts.
The molecular pathways involving K6 ubiquitin linkages in DNA damage response can be visualized as follows:
Diagram 2: K6 Ubiquitin Signaling Pathway in DNA Damage Response. This diagram illustrates the molecular pathway of K6-linked ubiquitin chain formation and function in response to genotoxic stress, highlighting key enzymatic components and functional outcomes.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Essential Research Reagents for K6 Ubiquitin Studies
| Reagent Category | Specific Examples | Function/Application | Key Considerations |
|---|---|---|---|
| Cell Lines | RPE-1 (hTERT-immortalized), HEK293T, HCT116, U2OS | Model systems for DNA damage response studies | Select lines with intact DDR pathways; verify p53 status [55] |
| CRISPRi Tools | SPIDR library, dCas9-KRAB expression vectors | Genetic screening and targeted gene repression | Enables interrogation of essential DDR genes without DSB induction [55] |
| K6-Specific Reagents | K6-only ubiquitin mutants, K6 linkage-specific antibodies, TUBEs | Specific detection and manipulation of K6 chains | Verify specificity with linkage-specific controls [1] [52] |
| Genotoxic Agents | Formaldehyde, hydroxyurea, camptothecin, ultraviolet light | Induction of specific DNA lesions that engage K6 pathways | Optimize concentration and exposure time for each cell type [54] [10] [52] |
| E3 Ligase Tools | BRCA1-BARD1 expression constructs, RNF168 inhibitors | Modulation of K6 chain formation | Multiple E3s can generate K6 chains; assess functional redundancy [52] [53] |
| Detection Reagents | Linkage-specific ubiquitin antibodies, proteasome inhibitors, protein crosslinkers | Analysis of K6 chain dynamics and function | Combine multiple detection methods for validation [10] [52] |
| Deubiquitylase Inhibitors | USP1 inhibitors, USP7 inhibitors | Prevention of K6 chain removal | Enables accumulation of otherwise transient K6 signals [55] [52] |
Analytical Methodologies for K6 Chain Detection
Immunodetection and Microscopy Approaches
The detection of K6-linked ubiquitin chains requires specific analytical approaches that can distinguish this linkage type among the complex background of other ubiquitin modifications. Linkage-specific antibodies against K6 chains, while challenging to develop, provide the most direct approach for monitoring this modification. When using these reagents, include appropriate controls such as samples treated with linkage-specific deubiquitylases or cells expressing ubiquitin mutants to verify signal specificity. For immunofluorescence applications, combine K6 detection with markers of DNA damage (e.g., γH2AX, 53BP1) to assess the localization of this modification to sites of genomic lesions [52].
High-resolution microscopy techniques, including confocal and super-resolution microscopy, enable the precise spatial analysis of K6 chain formation within nuclear compartments. These approaches can reveal the accumulation of K6 ubiquitin at distinct types of DNA lesions, providing insight into the specific contexts in which this modification functions. When quantifying K6 signals by microscopy, measure both intensity and focal accumulation, as both parameters may provide biologically relevant information about the dynamics of this modification. Automated image analysis platforms can facilitate the objective quantification of these parameters across multiple experimental conditions and time points.
Biochemical and Proteomic Methods
Biochemical approaches provide complementary information about the abundance and protein associations of K6 ubiquitin chains. Tandem ubiquitin-binding entities (TUBEs) with specificity for K6 linkages can be used to enrich this modification from cell lysates, followed by mass spectrometric analysis to identify associated proteins [52]. This approach can reveal the protein networks engaged by K6 chains in response to different genotoxic stresses. When performing these analyses, include controls with non-specific TUBEs to account for background binding, and use crosslinking steps to preserve transient interactions.
Advanced mass spectrometry methods, including selected reaction monitoring (SRM) and parallel reaction monitoring (PRM), enable the targeted quantification of K6 linkage-containing peptides from complex samples [1]. These targeted approaches provide greater sensitivity and reproducibility compared to discovery-based proteomics for monitoring specific ubiquitin linkages. When designing mass spectrometry experiments, incorporate stable isotope-labeled ubiquitin as an internal standard to control for variation in sample preparation and instrument performance. The combination of biochemical enrichment and targeted mass spectrometry represents the current gold standard for comprehensive analysis of K6 chain dynamics in cellular models of genotoxic stress.
Concluding Remarks
The study of K6-linked ubiquitin chains in DNA damage response represents a rapidly evolving field with significant implications for understanding genome maintenance mechanisms and developing novel cancer therapeutic strategies. The optimized cellular models and genotoxic stress induction protocols outlined in this technical guide provide a foundation for rigorous investigation of this atypical ubiquitin modification. As research in this area advances, the development of more specific reagents and analytical methods will further enhance our ability to decipher the functional significance of K6 linkages in the complex network of DNA damage response pathways. The integration of these tools with emerging technologies in genome engineering and single-cell analysis promises to reveal new insights into how this specialized ubiquitin code contributes to the preservation of genomic integrity in the face of genotoxic challenges.
Addressing Antibody Specificity and Validation for K6-Linkage Detection
Protein ubiquitylation is a crucial post-translational modification that regulates nearly every aspect of the DNA damage response [1]. Among the various ubiquitin linkage types, Lys6 (K6)-linked polyubiquitin chains represent one of the less common forms that have been historically challenging to study due to a lack of specific research tools [56]. Recent research has illuminated that K6-linked ubiquitylation plays significant roles in DNA repair pathways, with formaldehyde-induced RNA-protein crosslinks (RPCs) being marked by atypical K6-linked ubiquitylation for subsequent resolution [5]. This pathway involves the RING-in-between-RING (RBR) E3 ligase RNF14 and the ubiquitin-dependent unfoldase VCP, highlighting the importance of K6-linked chains in maintaining genomic integrity [5].
The versatility of ubiquitin signaling stems from its ability to form diverse polyubiquitin chains through eight different linkage sites (M1, K6, K11, K27, K29, K33, K48, K63), creating a complex "ubiquitin code" that regulates cellular processes [57] [1]. While K48-linked chains primarily target substrates for proteasomal degradation and K63-linked chains regulate protein-protein interactions, the functions of atypical chains like K6 are less defined but increasingly recognized as critical for DNA damage response [57]. This technical guide addresses the specific challenges and solutions for detecting K6-linked ubiquitin chains, with emphasis on antibody validation frameworks suitable for DNA damage research.
The Complexity of K6-Linked Ubiquitin Signaling
Biochemical Foundations of K6-Linked Ubiquitination
Ubiquitylation involves a sequential enzymatic cascade comprising E1 activating enzymes, E2 conjugating enzymes, and E3 ligases that confer substrate specificity [1]. The human genome encodes approximately 2 E1 enzymes, 40 E2 enzymes, and over 600 E3 ligases, creating tremendous diversity in ubiquitin signaling [57]. Deubiquitinating enzymes (DUBs) reverse this modification, with approximately 100 encoded in the human genome, creating a dynamic regulatory system [57].
K6-linked ubiquitin chains have been implicated in DNA repair pathways, though their study has been hampered by the limited specificity of available research tools [1] [56]. Formaldehyde-induced RPCs are marked by K6-linked ubiquitylation in a process catalyzed by the RNF14 E3 ligase and resolved through VCP activity [5]. This pathway represents an evolutionary conserved formaldehyde-induced stress response that protects cells against RPC accumulation in the cytoplasm, suggesting that RPCs contribute to the cellular and tissue toxicity of reactive aldehydes [5].
Technical Challenges in K6-Linkage Detection
The analysis of K6-linked ubiquitin presents multiple technical challenges. First, the stoichiometry of protein ubiquitination is generally low under normal physiological conditions, increasing the difficulty of identifying ubiquitinated substrates [57]. Second, ubiquitin can modify substrates at multiple lysine residues simultaneously, complicating the localization of modification sites [57]. Third, ubiquitin itself serves as a substrate for chain formation, creating tremendous complexity in chain length, linkage type, and overall architecture [57]. These challenges are particularly pronounced for K6 linkages due to their relative scarcity compared to K48 and K63 linkages.
Table 1: Key Challenges in K6-Linked Ubiquitin Research
| Challenge Category | Specific Technical Limitations | Impact on K6 Research |
|---|---|---|
| Specificity | Limited availability of linkage-specific antibodies | High background signal from abundant ubiquitin chains |
| Sensitivity | Low stoichiometry of modification | Difficulty detecting endogenous K6 signals |
| Validation | Lack of standardized controls | Irreproducible results across laboratories |
| Tools | Scarce K6-specific enzymes | Limited orthogonal validation methods |
Antibody Validation Frameworks and Methodologies
Comprehensive Validation Pillars
The International Working Group for Antibody Validation (IWGAV) has proposed five foundational pillars for antibody validation, which can be adapted specifically for K6-linkage detection [58]. These pillars employ methods requiring no prior knowledge of the target protein beyond its sequence, making them particularly valuable for studying poorly characterized modifications.
1. Orthogonal Validation Strategies: This approach compares protein abundance levels determined by an antibody-dependent method with levels from an antibody-independent method across multiple samples [58]. For K6-linked ubiquitin detection, this could involve comparing immunofluorescence signals with mass spectrometry-based ubiquitinomics data. The correlation between antibody-based signals and orthogonal measurements across a panel of cell lines with varying expression levels provides robust validation of antibody specificity [58].
2. Genetic Validation: Genetic knockdown or knockout of the target protein should substantially reduce antibody signal. For K6-linkage detection, this could involve siRNA-mediated knockdown of specific E3 ligases like RNF14 that generate K6 linkages, or overexpression of K6-specific DUBs like LotA [5] [56]. The U2 OS cell line has been successfully used for such validation approaches [58].
3. Recombinant Expression: Expressing the target antigen (in this case, K6-linked ubiquitin chains) in a null background system can validate antibody binding specificity. This approach is particularly valuable for K6 research, as K6-linked diubiquitin can be recombinantly expressed and used to directly test antibody recognition [56].
4. Independent Antibody Validation: Comparing results from multiple antibodies targeting different epitopes on the same antigen provides strong evidence of specificity. For K6-linked ubiquitin, this would require antibodies recognizing different epitopes within the K6-linked ubiquitin chain structure [58].
5. Capture Mass Spectrometry: This emerging strategy involves immunoprecipitating the target antigen, separating by SDS-PAGE, and identifying associated peptides via mass spectrometry. For K6-linkage validation, this approach could confirm that antibody pull-downs specifically enrich proteins modified with K6-linked chains [58].
K6-Specific Validation Considerations
The unique structural and functional characteristics of K6-linked ubiquitin chains necessitate specialized validation approaches. The Legionella pneumophila effector protein LotA contains a deubiquitinase domain with exceptional specificity for K6-linked polyubiquitin, making it a valuable tool for validation experiments [56]. LotA can be employed to specifically remove K6-linked chains from samples, with the expectation that genuine K6 signals will disappear after LotA treatment but remain after exposure to DUBs with different linkage specificities.
Structural studies have revealed that LotA's specificity for K6 linkages arises from substrate-assisted catalysis, with unique ubiquitin-binding domains that preferentially engage K6-linked chains [56]. This mechanistic understanding enhances the utility of LotA as a validation tool for K6-specific antibodies.
Table 2: Experimental Parameters for K6 Antibody Validation
| Validation Method | Key Experimental Parameters | Acceptance Criteria |
|---|---|---|
| Orthogonal (MS-based) | Panel of 3-5 cell lines with varying expression; PRM or TMT mass spectrometry | Pearson correlation >0.7 across cell lines |
| Genetic Knockdown | siRNA against relevant E3 ligases (e.g., RNF14); 48-72 hour knockdown | ≥70% reduction in signal by Western blot |
| Recombinant Expression | Purified K6-diUb and other linkage types; concentration range 0.1-10 μM | Selective recognition of K6-linked chains only |
| Independent Antibodies | 2-3 antibodies targeting distinct K6-chain epitopes | Consistent signal patterns across applications |
| Enzymatic (LotA) | 1-2 hour treatment with 100 nM LotA; control with scrambled enzyme | Complete signal ablation with LotA only |
Research Reagent Solutions for K6-Linkage Studies
Table 3: Essential Research Reagents for K6-Linked Ubiquitin Detection
| Reagent Category | Specific Examples | Research Application | Validation Utility |
|---|---|---|---|
| K6-Specific DUBs | LotA (Legionella) [56] | Selective removal of K6 chains | Specificity control; cleavage validation |
| E3 Ligases | RNF14 [5] | Generation of K6 linkages | Positive control production |
| Reference Materials | Recombinant K6-diUb [56] | Standard curve generation | Antibody characterization |
| Linkage Controls | K48-, K63-diUb [57] | Specificity assessment | Cross-reactivity testing |
| Cell Line Panels | U-2 OS, RT4, U-251 [58] | Expression variability | Orthogonal validation |
Experimental Protocols for K6-Linkage Detection
Protocol 1: Orthogonal Validation Using Proteomics
Sample Preparation:
- Select a panel of 3-5 cell lines with varying expression of K6-linked ubiquitin targets (e.g., U-2 OS, RT4, U-251) [58].
- Prepare whole cell lysates using RIPA buffer supplemented with 10 mM N-ethylmaleimide to preserve ubiquitin conjugates.
- Normalize protein concentrations using BCA assay.
Parallel Analysis:
- For Western blot: Separate 20 μg lysate by SDS-PAGE (4-12% Bis-Tris), transfer to PVDF, and probe with anti-K6 linkage antibody.
- For mass spectrometry: Digest 100 μg protein with trypsin, desalt, and analyze by parallel reaction monitoring (PRM) targeting K6 linkage-specific peptides [58].
- Include internal standard heavy-labeled peptides for quantitative accuracy.
Data Analysis:
- Quantify band intensities from Western blot using densitometry.
- Extract peak areas for K6-specific peptides from PRM data.
- Calculate Pearson correlation coefficient between Western blot signals and MS-based quantification across the cell line panel.
- A correlation coefficient >0.7 suggests specific recognition of K6-linked ubiquitin [58].
Protocol 2: Genetic Knockdown Validation
siRNA Transfection:
- Seed U-2 OS cells at 30% confluency in 6-well plates.
- Transfect with 25 nM siRNA targeting RNF14 using appropriate transfection reagent [5] [58].
- Include non-targeting siRNA as negative control.
- Culture for 72 hours with medium change at 24 hours.
Sample Analysis:
- Prepare lysates at 72 hours post-transfection.
- Analyze by Western blot using anti-K6 linkage antibody.
- Probe parallel blots for RNF14 to confirm knockdown efficiency.
- Expect ≥70% reduction in K6 signal with RNF14 knockdown compared to control [5].
Protocol 3: Recombinant Ubiquitin Chain Specificity Testing
Sample Preparation:
- Source recombinant diubiquitin of various linkage types (K6, K11, K48, K63).
- Prepare serial dilutions from 0.1 to 10 μM in PBS.
- Spot 1 μL of each dilution onto nitrocellulose membrane and air dry.
Dot Blot Analysis:
- Block membrane with 5% non-fat milk in TBST for 1 hour.
- Incubate with anti-K6 linkage antibody at recommended dilution in blocking buffer.
- Wash 3× with TBST, 5 minutes each.
- Incubate with HRP-conjugated secondary antibody.
- Develop with ECL substrate and image.
- Antibody should show strong signal with K6-diUb and minimal cross-reactivity with other linkage types.
Visualization of K6-Linked Ubiquitin Signaling Pathways
Technical Considerations for K6-Linkage Research
Methodological Limitations and Solutions
The study of K6-linked ubiquitin presents unique methodological challenges. Traditional immunoblotting approaches suffer from limitations in specificity and throughput, though they remain widely used for initial validation [57]. Mass spectrometry-based methods offer superior specificity but require specialized instrumentation and expertise. The low stoichiometry of K6-linked ubiquitination necessitates efficient enrichment strategies prior to analysis.
Ubiquitin tagging-based approaches using epitope tags (e.g., His, Strep, HA) enable purification of ubiquitinated substrates but may introduce artifacts as tagged ubiquitin does not completely mimic endogenous ubiquitin [57]. Antibody-based enrichment using pan-ubiquitin antibodies (e.g., P4D1, FK1/FK2) can overcome this limitation but may exhibit preferential recognition of certain linkage types. The development of truly specific K6 linkage antibodies remains challenging due to structural similarities between different ubiquitin chain types.
Emerging Technologies and Future Directions
Recent advances in ubiquitin research methodology promise to enhance K6 linkage detection. The MAGMA-seq technology enables quantitative analysis of protein-binding interactions using deep sequencing, which could be adapted for studying ubiquitin ligase specificity [59]. Improvements in mass spectrometry sensitivity and the development of better ubiquitin enrichment methods continue to push detection limits for low-abundance modifications like K6 linkages.
The discovery of bacterial effectors with exceptional linkage specificity, such as LotA from Legionella pneumophila, provides new tools for K6 research [56]. These naturally evolved specific enzymes can be employed as critical validation reagents and may inspire the development of improved detection reagents. As structural insights into K6 recognition mechanisms accumulate, rational design of more specific antibodies and binding domains becomes increasingly feasible.
The detection and validation of K6-linked ubiquitin chains requires specialized approaches that address the unique challenges of this atypical ubiquitin linkage. By implementing the comprehensive validation frameworks outlined in this guide—including orthogonal methods, genetic validation, and enzymatic approaches using specific tools like LotA—researchers can achieve reliable detection of K6 linkages in DNA damage response contexts. The continued development of K6-specific research tools and validation methodologies will be essential for elucidating the full functional significance of this modification in genome maintenance and cellular stress response pathways.
The DNA damage response (DDR) employs a sophisticated system of post-translational modifications to coordinate repair processes and maintain genomic integrity. Among these modifications, ubiquitin signaling—particularly through atypical K6-linked polyubiquitin chains—has emerged as a critical regulator with dedicated functions in DDR pathways. This technical guide provides researchers with a comprehensive framework for integrating K6 ubiquitinomics datasets with functional DDR readouts. We detail experimental methodologies for detecting K6 ubiquitination, present quantitative data on DDR-induced K6 chain dynamics, and outline mechanistic insights into how K6 linkages coordinate DNA repair processes. The resources and protocols herein are designed to advance research into this understudied yet crucial aspect of ubiquitin signaling in genome stability.
Protein ubiquitylation represents a fundamental regulatory mechanism that impacts nearly every aspect of the DDR [1]. The complexity of ubiquitin signaling arises from the ability of ubiquitin to form diverse polyubiquitin chains through its seven internal lysine residues (K6, K11, K27, K29, K33, K48, K63) and N-terminal methionine (M1) [1] [25]. While K48- and K63-linked chains represent the most thoroughly characterized ubiquitin linkages, recent evidence has established that atypical K6-linked chains serve specialized, non-redundant functions in DDR pathways [25] [50].
K6-linked ubiquitination undergoes significant induction following genotoxic stress. Quantitative proteomic analyses reveal that K6- and K33-linked polyubiquitination undergo bulk increases in response to DNA damage, suggesting these linkages are largely dedicated to DDR function [50]. This damage-induced K6 ubiquitylation creates binding platforms for specific DDR factors and helps orchestrate the complex sequence of events required for efficient DNA repair. The study of K6 ubiquitinomics—the comprehensive analysis of K6-linked ubiquitination events—therefore provides critical insights into DDR regulation and offers potential therapeutic avenues for cancer treatment targeting genomic instability.
Quantitative Landscape of K6 Ubiquitin in DNA Damage
Proteomic studies have begun to quantify the dynamics and scope of K6 ubiquitination in response to different genotoxic insults. The table below summarizes key quantitative findings from recent studies profiling K6 ubiquitin changes following DNA damage.
Table 1: Quantitative Profiling of K6 Ubiquitin Dynamics in DNA Damage Response
| Study Context | DNA Damage Type | K6 Ubiquitin Change | Key Identified E3 Ligases | Experimental Approach |
|---|---|---|---|---|
| Global proteomic analysis [50] | UV radiation | Significant bulk increase | SCF-Cyclin F | Quantitative proteomics (FACET-IP) |
| Global proteomic analysis [50] | Ionizing radiation | Significant bulk increase | Not specified | Quantitative proteomics (FACET-IP) |
| Interaction proteomics [26] | DNA damage (unspecified) | Induced interaction networks | HUWE1 | UbIA-MS (Ubiquitin Interactor Affinity Enrichment-MS) |
| Affimer-based profiling [25] | DNA damage (unspecified) | Increased levels | HUWE1, RNF144A/B | K6-affimer pull-down + mass spectrometry |
The HUWE1 HECT-domain E3 ligase has been identified as a major source of cellular K6 chains, with HUWE1 knockout or knockdown cells showing significantly reduced K6 chain levels [25]. Additionally, the RBR-family E3 ligases RNF144A and RNF144B have been demonstrated to assemble K6-, K11-, and K48-linked polyubiquitin chains in biochemical assays [25]. Beyond these E3 ligases, K6 linkages have been functionally linked to the well-known DDR E3 ligase BRCA1, though its direct role in K6 chain assembly requires further clarification [25].
Table 2: K6 Ubiquitin Ligases and Their Functional Roles in DDR
| E3 Ligase | E3 Class | K6 Linkage Role in DDR | Validated Substrates | Cellular Function |
|---|---|---|---|---|
| HUWE1 | HECT | Major source of cellular K6 chains | Mitofusin-2 (Mfn2) | Mitochondrial dynamics, DNA damage-induced apoptosis? |
| RNF144A/B | RBR | Assembles K6/K11/K48 chains in vitro | Not fully characterized | DNA damage response, p53 regulation |
| BRCA1 | RING | Earlier biochemical associations | Not fully established | Homologous recombination, checkpoint signaling |
| SCF-Cyclin F | CRL | Mediates EXO1 ubiquitination post-UV | EXO1 | DNA end resection, replication stress response |
Experimental Protocols for K6 Ubiquitin Detection and Functional Analysis
K6 Linkage-Specific Affimer Enrichment and Detection
The development of linkage-specific affinity reagents has dramatically advanced the study of atypical ubiquitin chains. The following protocol describes the use of K6-specific affimers for detecting and enriching K6-linked ubiquitinated proteins:
Principle: Affimers are small (12-kDa) non-antibody protein scaffolds based on the cystatin fold that can be engineered for high-affinity, linkage-specific recognition of ubiquitin chains [25]. K6-specific affimers recognize K6-diUb with high specificity through a dimerization mechanism that provides two binding sites for Ub I44 patches with defined distance and orientation [25].
Reagents Required:
- K6-linkage-specific affimers (biotinylated for pull-down assays)
- Control affimers (non-specific or K33-specific)
- Cell lysates from DNA-damaged and undamaged conditions
- Streptavidin-conjugated beads (for pull-down)
- Appropriate detection antibodies
Procedure:
- Cell Lysis and Preparation: Harvest cells after DNA damage induction (e.g., UV, IR, MMS) using denaturing or non-denaturing lysis conditions based on downstream applications.
- Affimer Incubation: Incubate clarified cell lysates with biotinylated K6 affimers (0.5-1 µg per mg lysate) for 2 hours at 4°C with gentle rotation.
- Complex Capture: Add streptavidin beads and incubate for an additional 1 hour.
- Washing: Wash beads extensively with lysis buffer to remove non-specific interactions.
- Elution and Analysis:
- For mass spectrometry: Elute with 2× Laemmli buffer at 95°C for 10 minutes
- For western blotting: Directly boil beads in Laemmli buffer
- Detection:
- Western blotting using standard ubiquitin detection antibodies
- Mass spectrometry analysis for protein identification
Validation: Confirm K6 linkage specificity through competition with free K6-diUb but not other linkage types. Include controls using K33 affimers which may cross-react with K11 linkages [25].
Ubiquitin Interactor Affinity Enrichment-Mass Spectrometry (UbIA-MS)
UbIA-MS enables proteome-wide profiling of ubiquitin linkage-selective interactors from crude cell lysates using chemically synthesized diubiquitin [26]:
Principle: Quantitative interaction proteomics using defined diubiquitin linkages to enrich and identify linkage-specific interactors and their dynamics in response to DNA damage.
Procedure:
- Synthesis of K6-diUbiquitin: Chemically synthesize K6-linked diubiquitin using native chemical ligation or expressed protein ligation.
- Immobilization: Covalently couple K6-diUb to solid support (e.g., agarose beads) while maintaining linkage integrity.
- Enrichment: Incubate immobilized K6-diUb with cell lysates from DNA-damaged and control conditions.
- Quantitative Proteomics:
- Perform on-bead tryptic digestion
- TMT or label-free quantitative mass spectrometry
- Compare enrichment in damage vs control conditions
- Data Analysis: Identify proteins with significant enrichment in DNA damage conditions using appropriate statistical thresholds.
Applications: This approach has identified novel K6-diUb interactors such as TAB2 and TAB3, and revealed classes of monoubiquitin and K6-diUb interactors whose binding is induced by DNA damage [26].
Functional Validation of K6 Ubiquitination in DDR
Genetic Rescue Experiments: To establish causal relationships between K6 ubiquitination and DDR outcomes, employ genetic rescue experiments in E3 ligase-deficient backgrounds:
- Generate HUWE1 or RNF144A/B knockout cells using CRISPR-Cas9
- Transfect with wild-type or catalytically dead (Cys-to-Ala) E3 mutants
- Assess rescue of DDR phenotypes:
- K6 ubiquitin levels by affimer-based detection
- Rad51 focus formation (HR efficiency)
- 53BP1 focus formation (NHEJ regulation)
- Clonogenic survival after DNA damage
Single-Cell Analysis of DDR Foci: Combine K6 ubiquitin detection with established DDR markers at single-cell resolution:
- Perform immunofluorescence using K6 linkage-specific reagents
- Co-stain for DDR markers (γH2AX, 53BP1, BRCA1, Rad51)
- Quantify co-localization and temporal dynamics
- Correlate K6 ubiquitin intensity with repair pathway choice
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Reagents for K6 Ubiquitin-DDR Studies
| Reagent Category | Specific Examples | Function/Application | Key Characteristics |
|---|---|---|---|
| Linkage-specific affinity reagents | K6-specific affimers [25] | Western blot, immunofluorescence, pull-down assays | High specificity for K6 linkages; minimal cross-reactivity |
| Linkage-specific antibodies | Commercial K6-Ub antibodies | Immunodetection applications | Variable quality; require rigorous validation |
| Defined ubiquitin reagents | K6-diUbiquitin, K6-only ubiquitin mutants (K6-only Ub) | In vitro assays, structural studies, UbIA-MS | Critical for biochemical and structural work |
| E3 ligase targeting reagents | HUWE1 shRNA/CRISPR, RNF144A/B inhibitors | Genetic and pharmacological perturbation | Establish functional relationships |
| Mass spectrometry standards | TMT reagents, SILAC media, DiGly remnant antibodies | Proteomic quantification, ubiquitin site mapping | Enable system-wide studies |
| Deubiquitinase inhibitors | UCHL3 inhibitors (for K27/K6 chains?) [26] | Pathway perturbation, mechanistic studies | Help establish dynamics and turnover |
Integrating K6 Ubiquitinomics with DDR Functional Assays
The true power of K6 ubiquitinomics emerges when correlation analyses are performed between ubiquitination changes and established functional DDR readouts. The integrated workflow below illustrates this systematic approach:
Correlation Analysis Framework
Establish quantitative relationships between K6 ubiquitination changes and functional DDR parameters:
- Temporal Correlation: Measure K6 ubiquitination dynamics alongside repair protein recruitment (e.g., RPA, Rad51, 53BP1) at early (0-2h), intermediate (2-8h), and late (8-24h) timepoints post-damage
- Dosage Response Correlation: Assess K6 ubiquitination levels across a range of DNA damage doses (e.g., IR: 0.5-10Gy; UV: 5-50J/m²) alongside corresponding survival curves
- Spatial Correlation: Quantify co-localization of K6 ubiquitin signals with specific DDR markers at single-cell resolution using high-content imaging and automated image analysis
Experimental Considerations for Robust Integration
- Cell Cycle Synchronization: Given the cell cycle dependence of DDR pathways (HR restricted to S/G2), synchronize cell populations before damage induction
- Genotoxic Agent Selection: Employ diverse DNA damaging agents (IR, UV, crosslinkers, topoisomerase inhibitors) to probe pathway-specific K6 ubiquitination
- Validation Approaches: Combine genetic (CRISPR knockout), biochemical (in vitro reconstitution), and microscopic (live-cell imaging) approaches to validate correlations
The integration of K6 ubiquitinomics with functional DDR readouts represents a powerful approach to deciphering the complex regulatory networks that maintain genome stability. As the tools for studying atypical ubiquitin linkages continue to advance—particularly with the refinement of linkage-specific affimers and quantitative proteomic methods—researchers are now positioned to unravel the specific mechanisms through which K6 ubiquitination controls DNA repair pathway choice, efficiency, and fidelity. The experimental frameworks outlined in this guide provide a roadmap for systematically investigating these mechanisms, with particular relevance for understanding how dysregulation of K6 ubiquitin signaling may contribute to carcinogenesis and therapy resistance. Future efforts should focus on expanding the toolkit of K6-specific research reagents, particularly those amenable to in vivo applications, and developing computational models that can predict DDR outcomes based on ubiquitination signatures.
Context and Specificity: Placing K6 Linkages within the Broader Ubiquitin Code
Ubiquitination is a critical post-translational modification that regulates virtually every cellular process, from protein degradation to DNA repair and immune signaling. The diversity of ubiquitin signals—termed the "ubiquitin code"—stems from the ability of ubiquitin to form chains through different lysine residues on the ubiquitin molecule itself. Among the seven possible lysine linkage sites (K6, K11, K27, K29, K33, K48, and K63), three have emerged with particularly distinct functional specializations: K48-linked chains primarily target substrates for proteasomal degradation; K63-linked chains serve as non-proteolytic signaling scaffolds in pathways ranging from inflammation to DNA repair; and the less-understood K6-linked chains have recently been implicated in specialized DNA damage response pathways. This review will explore the functional dichotomy between these three ubiquitin linkage types, with particular emphasis on the emerging role of K6-linked ubiquitylation in resolving specific types of DNA damage, contrasting it with the well-established roles of K48 chains in protein turnover and K63 chains in cellular signaling. Understanding this functional specialization provides critical insights for developing targeted therapeutic interventions in cancer and other human diseases.
Fundamental Principles of Ubiquitin Chain Linkages
Ubiquitylation involves a sequential enzymatic cascade comprising ubiquitin-activating (E1), conjugating (E2), and ligase (E3) enzymes that ultimately attach ubiquitin to substrate proteins [1]. The complexity of ubiquitin signaling arises from the ability of ubiquitin itself to be modified on any of its seven lysine residues (K6, K11, K27, K29, K33, K48, K63) or its N-terminal methionine, creating chains with distinct structures and functions [1] [60]. The topology of these chains determines their specific recognition by proteins containing ubiquitin-binding domains (UBDs), enabling the transduction of diverse cellular signals [1].
Table 1: Fundamental Properties of Major Ubiquitin Linkage Types
| Linkage Type | Primary Functions | Chain Topology | Key Recognizing Domains/Proteins |
|---|---|---|---|
| K6-linked | DNA damage response, RPC resolution | Not fully characterized | RNF14, VCP [14] [5] |
| K48-linked | Proteasomal degradation | Closed conformation [1] | Proteasome receptors, UCH37 [1] [61] |
| K63-linked | Signal transduction, DNA repair, inflammation | Extended conformation [1] | TAB2, TAB3, UBDs in signaling complexes [62] [63] |
Monoubiquitylation and multi-monoubiquitylation regulate processes such as endocytosis and transcriptional regulation, while polyubiquitylation creates more complex signals [1]. The ubiquitin code is further refined by the action of deubiquitinating enzymes (DUBs) that remove ubiquitin modifications, creating a dynamic, reversible system for cellular regulation [1] [61]. Recent research has revealed additional complexity through the existence of branched ubiquitin chains, where a single ubiquitin molecule serves as an attachment point for multiple chains of different linkages, creating hybrid signals that can be recognized by specific readers and protected from specific erasers [63] [64].
K6-Linked Ubiquitylation in RNA-Protein Crosslink Resolution
Discovery and Mechanism of K6-Linked Ubiquitylation in RPC Resolution
Until recently, K6-linked ubiquitylation was one of the least understood ubiquitin modifications. A groundbreaking 2023 study revealed that K6-linked chains play a specialized role in resolving RNA-protein crosslinks (RPCs) induced by formaldehyde exposure [14] [5]. Formaldehyde is produced during normal cellular metabolism and after alcohol consumption, and it accumulates in tissues when aldehyde clearance mechanisms are impaired. While formaldehyde toxicity was previously attributed primarily to DNA damage, it is now established that formaldehyde induces RPCs that stall ribosomes and inhibit translation, representing a previously unappreciated mechanism of cellular toxicity [14].
The mechanism for resolving these transcription-blocking lesions involves a dedicated pathway wherein RPCs in messenger RNA are recognized by translating ribosomes and marked by atypical K6-linked ubiquitylation [5]. This modification is catalyzed by the RING-in-between-RING (RBR) E3 ligase RNF14 [14]. Following ubiquitylation, the ubiquitin- and ATP-dependent unfoldase VCP (valosin-containing protein) is recruited to resolve the crosslinks [14] [5]. This pathway represents an evolutionarily conserved formaldehyde-induced stress response that protects cells against RPC accumulation in the cytoplasm.
Experimental Workflow for Studying K6-Linked Ubiquitylation
Table 2: Key Experimental Approaches for Studying K6-linked Ubiquitylation
| Methodology | Application | Key Findings |
|---|---|---|
| Quantitative Proteomics | Identify proteins modified with K6-linked ubiquitin in response to formaldehyde | Identification of RNA-binding proteins that form RPCs marked by K6-linked ubiquitylation [14] |
| Genetic Manipulation (RNF14 knockout) | Establish functional requirement for E3 ligase | Confirmed RNF14 as essential E3 ligase for K6-chain formation on RPCs [5] |
| Translation Inhibition Assays | Measure functional consequences of RPC accumulation | Demonstrated RPCs stall ribosomes and inhibit protein synthesis [14] [5] |
| Biochemical Reconstitution | Define minimal components required for RPC resolution | Established GCN1-RNF14-VCP as core resolution machinery [5] |
Figure 1: K6-linked Ubiquitin Pathway for Resolving RNA-Protein Crosslinks (RPCs). Formaldehyde induces RPCs that stall ribosomes during translation. The stalled complexes are marked by K6-linked ubiquitylation catalyzed by RNF14, recruiting VCP which resolves the crosslinks.
The discovery of this pathway required sophisticated quantitative proteomic approaches to distinguish K6-linked ubiquitylation from more abundant ubiquitin chain types. Researchers employed linkage-specific affinity tools and translation shutoff experiments to identify specific proteins whose processing depends on K6-linked ubiquitin signaling [14] [5]. Furthermore, conservation of this pathway across species suggests it represents a fundamental mechanism for maintaining proteostasis under genotoxic stress.
K48-Linked Ubiquitylation: The Canonical Degradation Signal
Structural Basis and Functional Roles of K48-Linked Chains
K48-linked ubiquitin chains represent the best-characterized ubiquitin modification and serve as the primary signal for proteasomal degradation [1] [60]. Structural studies have revealed that K48-linked di-ubiquitin adopts a closed conformation that is specifically recognized by proteasomal receptors [1]. Chains of at least four ubiquitin molecules linked through K48 are sufficient to target substrates to the 26S proteasome for degradation [1]. This system provides a selective mechanism for controlling the half-life of regulatory proteins, thereby influencing virtually all cellular processes.
The specificity of K48 chain recognition is exemplified by recent research on the deubiquitinase UCH37 (UCHL5), which contains a cryptic K48 ubiquitin chain binding site required for its role in proteasomal degradation [61]. This specialized binding site enables UCH37 to debranch K48-linked ubiquitin chains, particularly removing K48 branches from heterogeneous chains, thereby fine-tuning protein stability [61]. The precise control of K48-linked ubiquitylation is essential for maintaining cellular homeostasis, as dysregulation can lead to either accumulation of damaged proteins or premature degradation of essential regulators.
Research Tools for Studying K48-Linked Ubiquitylation
Table 3: Key Research Reagents for Studying K48-linked Ubiquitin Signaling
| Research Tool | Type | Research Application | Example Use |
|---|---|---|---|
| UCH37 inhibitors | Small molecule inhibitors | Probe K48-chain debranching | Study proteasomal processing of K48-ubiquitylated substrates [61] |
| Linkage-specific antibodies | Antibodies | Detect endogenous K48 chains | Monitor cellular K48 ubiquitin levels under different conditions [64] |
| K48-specific DUBs | Enzymatic tools | Validate chain linkage | UbiCRest assay to confirm K48 linkage in purified chains [64] |
| Proteasome inhibitors | Small molecule inhibitors | Confirm degradation-dependent phenotypes | Establish functional consequence of K48 ubiquitylation [1] |
Advanced techniques such as hydrogen-deuterium exchange mass spectrometry (HDX-MS), chemical crosslinking, small-angle X-ray scattering (SAXS), and nuclear magnetic resonance (NMR) have been instrumental in characterizing the structural basis of K48 chain recognition [61]. These approaches revealed how UCH37 achieves specificity for K48 chains through a binding site on the opposite face of the enzyme relative to the canonical ubiquitin-binding site [61]. Molecular docking and targeted mutagenesis studies further confirmed that this K48-specific site is required for chain debranching and proteasome-mediated degradation of proteins modified with branched chains [61].
K63-Linked Ubiquitylation: A Multifunctional Signaling Scaffold
Diverse Signaling Roles of K63-Linked Ubiquitylation
In contrast to K48-linked chains, K63-linked ubiquitin chains serve non-proteolytic functions in multiple signaling pathways [62] [65]. Structural studies show that K63-linked di-ubiquitin adopts an extended conformation that is optimal for serving as a scaffold in signal transduction complexes [1]. This linkage type plays critical roles in immune signaling, DNA damage repair, protein trafficking, and selective autophagy [62] [65].
In immune signaling, K63-linked ubiquitylation regulates multiple receptors including Toll-like receptors (TLRs), Nod-like receptors (NLRs), RIG-I-like receptors (RLRs), and the interleukin-1 receptor (IL-1R) [62]. Key signaling adaptors such as cellular inhibitor of apoptosis proteins (cIAPs), TNF receptor-associated factors (TRAFs), and stimulator of interferon genes (STING) serve as docking platforms for K63-linked ubiquitin chain assembly during immune cell activation [62]. The K63 ubiquitin chains can be formed on target proteins or exist as unanchored chains that serve as activation platforms for signaling molecules [62].
K63-Linked Ubiquitylation in Cancer and Therapeutic Targeting
In cancer biology, K63-linked ubiquitylation controls initiation, development, invasion, metastasis, and therapy response across diverse cancer types [65]. It regulates multiple oncogenic signaling pathways including PI3K/AKT, Wnt/β-catenin, and JNK signaling [65]. For instance, K63-linked ubiquitylation of Akt regulates its activation and oncogenic functions [65]. Similarly, K63-linked ubiquitylation of β-catenin facilitates its nuclear translocation and transcriptional activity, promoting carcinogenesis [65].
The therapeutic potential of targeting K63-linked ubiquitylation is highlighted by synthetic lethality screens showing that loss-of-function of UBE2N (also known as UBC13, the primary E2 enzyme for K63 chain formation) sensitizes cells to G-quadruplex stabilizers like CX-5461 [66]. Pharmacological inhibition of UBE2N acted synergistically with CX-5461, suggesting combination therapy approaches [66]. This synthetic lethality relationship underscores the importance of K63 ubiquitin signaling in the DNA damage response and genome maintenance.
Figure 2: K63-linked Ubiquitin Signaling in Immune Response and Cancer. Immune signals activate E3 ligases like TRAF6, which assemble K63-linked chains that activate NF-κB signaling, driving inflammation and cancer progression.
Comparative Analysis of Ubiquitin Linkage Functions
Structural and Functional Dichotomy Between Linkage Types
The functional specialization of K6, K48, and K63-linked ubiquitin chains stems from their distinct structural properties and recognition by specific effector proteins. While K48-linked chains adopt a closed conformation ideal for proteasomal engagement, K63-linked chains form extended structures that serve as scaffolding platforms for signaling complexes [1]. The structure of K6-linked chains remains less characterized but appears optimized for recruitment of specific DNA damage response factors like RNF14 and VCP [14] [5].
The functional dichotomy between these linkage types is maintained through specific recognition by ubiquitin-binding domains (UBDs) that can discriminate between different chain topologies [1] [64]. For instance, the proteasome-associated deubiquitinase UCH37 contains a cryptic binding site specifically recognizing K48 linkages [61], while TAB2 and TAB3 preferentially bind K63 linkages in the NF-κB pathway [63]. Recent interactome studies have identified the first K48/K63 branched chain-specific interactors, including PARP10, UBR4, and HIP1, revealing an additional layer of specificity in ubiquitin code interpretation [64].
Experimental Approaches for Linkage-Specific Research
Table 4: Comparative Experimental Analysis of Ubiquitin Linkages
| Experimental Approach | K6-linked | K48-linked | K63-linked |
|---|---|---|---|
| Primary Validation Method | Quantitative proteomics, RNF14 dependence | Proteasome inhibition, UCH37 binding [61] | UBE2N dependence, signaling assays [62] |
| Specific Inhibitors/Tools | RNF14 knockdown/knockout | UCH37 backside mutants, proteasome inhibitors [61] | UBE2N inhibitors, dominant-negative UBE2N [66] |
| Key Functional Assays | Translation inhibition, RPC resolution assays | Protein half-life measurements, cycloheximide chase | Immune signaling readouts, reporter assays [62] |
| Chain Architecture | Homotypic (predominantly) | Homotypic and branched with K63 [63] | Homotypic and branched with K48 [63] [64] |
Advanced proteomic techniques have been developed to distinguish between these linkage types and quantify their relative abundance. Quantitative mass spectrometry approaches have revealed that K48- and K63-linked chains are the most abundant linkage types in cells, with branched K48/K63 chains comprising up to 20% of all K63 linkages [64]. These branched chains create unique coding signals that regulate pathway output, as demonstrated in NF-κB signaling where K48-K63 branched linkages permit recognition by TAB2 while protecting K63 linkages from CYLD-mediated deubiquitylation [63].
The Scientist's Toolkit: Key Research Reagents and Methodologies
Essential Research Reagents for Ubiquitin Linkage Studies
Table 5: Essential Research Reagent Solutions for Ubiquitin Studies
| Reagent Category | Specific Examples | Function/Application | Linkage Specificity |
|---|---|---|---|
| E2 Enzymes | Ubc13/Uev1a (K63), CDC34 (K48), Ubc1 (K48-branching) [64] | In vitro ubiquitin chain synthesis | Linkage-specific chain assembly |
| DUB Inhibitors | Chloroacetamide (CAA), N-ethylmaleimide (NEM) [64] | Preserve ubiquitin chains in lysates | General DUB inhibition |
| Linkage-specific DUBs | OTUB1 (K48-specific), AMSH (K63-specific) [64] | Chain linkage validation (UbiCRest) | Linkage verification |
| Branched Chain Tools | Enzymatically synthesized K48/K63 branched Ub3 [64] | Study branched chain biology | Branched chain specificity |
| Affinity Reagents | Linkage-specific antibodies, ubiquitin interactor capture [64] | Detect and purify specific chains | Linkage-specific detection |
When conducting ubiquitin pulldown studies, researchers must carefully consider their choice of deubiquitinase inhibitors, as different inhibitors can significantly impact experimental outcomes. Comparative studies have revealed inhibitor-dependent interactors, with chloroacetamide (CAA) and N-ethylmaleimide (NEM) showing different off-target effects that can alter ubiquitin-binding surfaces [64]. For example, NEM treatment was found to perturb NEMO binding to K63 ubiquitin chains in vitro [64], highlighting the importance of inhibitor consideration in experimental design.
The functional dichotomy between K6, K48, and K63-linked ubiquitin chains represents a fundamental organizing principle in cellular regulation. While K48-linked chains primarily direct protein degradation and K63-linked chains serve as signaling scaffolds, K6-linked chains have emerged as specialized mediators of DNA damage response, particularly in resolving transcription-blocking RNA-protein crosslinks. This functional specialization enables the ubiquitin system to coordinate diverse cellular processes with remarkable specificity.
Future research directions will likely focus on understanding the full complexity of branched ubiquitin chains that incorporate multiple linkage types within a single ubiquitin polymer. These heterogeneous signals may integrate information from different pathways to fine-tune cellular responses. Additionally, the therapeutic potential of targeting specific ubiquitin linkages is becoming increasingly apparent, with synthetic lethality approaches showing promise for cancer therapy [66]. As our understanding of the ubiquitin code continues to expand, so too will opportunities for developing novel therapeutic strategies that modulate ubiquitin signaling with unprecedented precision.
The ongoing development of research tools—including linkage-specific antibodies, engineered enzymes for chain synthesis and analysis, and small molecule inhibitors targeting specific pathway components—will continue to drive discoveries in this field. Particularly valuable are reagents that can specifically manipulate K6-linked ubiquitylation, which remains the least characterized of the major linkage types. As these tools become more sophisticated and accessible, we can anticipate rapid advances in understanding how the ubiquitin code controls cellular homeostasis and disease pathogenesis.
Protein ubiquitylation is a crucial post-translational modification (PTM) that regulates nearly every aspect of cellular function, with particular importance in the DNA damage response (DDR) [1] [24]. The complexity of ubiquitin signaling arises not only from the diverse chain topologies it can form but also from its intricate interplay with other PTMs. This cross-talk creates a sophisticated regulatory network that enables cells to respond dynamically to genotoxic stress and maintain genomic integrity [67] [68].
The ubiquitin conjugation system employs a three-enzyme cascade consisting of ubiquitin-activating (E1), ubiquitin-conjugating (E2), and ubiquitin-ligase (E3) enzymes to attach ubiquitin to substrate proteins [1] [24]. Ubiquitin itself contains seven lysine residues (K6, K11, K27, K29, K33, K48, K63) and an N-terminal methionine that can form polyubiquitin chains with distinct functions [1] [69]. While K48-linked chains primarily target substrates for proteasomal degradation and K63-linked chains function in signaling pathways, K6-linked chains have emerged as important regulators in DNA repair processes [1] [33].
This review examines the complex interplay between ubiquitination—particularly K6-linked chains—and other PTMs including phosphorylation, SUMOylation, and RNA modifications, with emphasis on their collective role in orchestrating the DNA damage response and implications for targeted therapeutic development.
Ubiquitin Linkage Types and Functions
Diversity of Ubiquitin Signaling
The ubiquitin code encompasses multiple types of modifications that determine functional outcomes for modified substrates. Monoubiquitination involves attachment of a single ubiquitin moiety and regulates processes including endocytosis, DNA repair, and transcriptional regulation [1] [70]. Multiple monoubiquitination (multi-ubiquitylation) occurs when several lysine residues on a substrate are each tagged with a single ubiquitin molecule [1]. Polyubiquitination involves the formation of ubiquitin chains through specific lysine residues, with each linkage type generating distinct chain topologies and cellular functions [1] [69].
Table 1: Ubiquitin Linkage Types and Their Primary Functions
| Linkage Type | Structural Features | Primary Cellular Functions |
|---|---|---|
| K6 | Extended conformation | DNA repair, mitochondrial autophagy [1] [33] |
| K11 | Unique conformation distinct from K48/K63 | Cell cycle regulation, ER-associated degradation [1] [69] |
| K27 | Not well characterized | Mitochondrial autophagy [33] |
| K29 | Heterogeneous chains | Proteasomal and lysosomal degradation [1] [69] |
| K33 | Mixed chains with K29 | AMPK-related kinase regulation [70] |
| K48 | Closed conformation | Proteasomal degradation [1] [69] |
| K63 | Extended conformation | DNA repair, signal transduction, protein trafficking [1] [69] |
| M1/Linear | Peptide bond between methionine and glycine | Inflammatory and immune responses [70] |
K6 Ubiquitin Linkages in DNA Damage Response
K6-linked ubiquitin chains (UbK6) have been increasingly recognized as important players in the cellular response to DNA damage [1]. While less abundant than K48 or K63 linkages, K6 chains participate in specific DNA repair pathways and are implicated in maintaining genomic stability. The assembly of K6-linked chains is catalyzed by specific E2 and E3 enzymes, though the complete repertoire of enzymes responsible for K6 chain formation remains an active area of investigation [33].
Recent studies have revealed that K6 ubiquitination contributes to the coordination of DNA repair mechanisms, particularly in response to double-strand breaks (DSBs) [1]. These linkages function as recruitment signals for DNA repair factors and help orchestrate the sequential assembly of repair complexes at damage sites. The significance of K6 linkages is further highlighted by their deregulation in certain pathological conditions, including cancer [33].
Cross-Talk with Phosphorylation
Cooperative Regulation in DNA Damage Signaling
Phosphorylation and ubiquitination engage in extensive cross-talk that amplifies and specifies DNA damage signals. This interplay is particularly evident in the early stages of the DNA damage response, where ubiquitination events are rapidly triggered following phosphorylation-mediated activation of apical kinases [24] [68].
At DNA double-strand breaks, the phosphorylation of histone H2AX (γH2AX) by the PI3K-related kinases ATM, ATR, and DNA-PK creates a platform for the recruitment of ubiquitin ligases including RNF8 and RNF168 [24]. These E3 ligases then catalyze the formation of ubiquitin chains—including K6 and K63 linkages—on histones H2A and H2AX, which in turn promotes the accumulation of additional DNA repair factors such as 53BP1 and BRCA1 [1] [24]. This sequential modification creates a positive feedback loop that amplifies the damage signal and ensures efficient repair.
Table 2: Key Enzymes in Phosphorylation-Ubiquitination Cross-Talk during DNA Damage Response
| Enzyme | Modification Type | Function in DDR | Substrates/Effectors |
|---|---|---|---|
| ATM | Phosphorylation | Apical kinase activated by DSBs | H2AX, CHK2, p53 [24] |
| ATR | Phosphorylation | Apical kinase activated by replication stress | CHK1, RPA [24] |
| DNA-PK | Phosphorylation | Kinase for NHEJ repair pathway | p53, DNA-PKcs [24] |
| RNF8 | Ubiquitination | Early E3 ligase at DSBs | H2A, H2AX [1] |
| RNF168 | Ubiquitination | Amplifies ubiquitin signaling at DSBs | H2A, K63 chains [1] |
| CHFR | Ubiquitination | Initial wave of histone ubiquitylation | PARP1, H2A [24] |
Antagonistic Regulation in Signal Termination
Beyond cooperative interactions, phosphorylation and ubiquitination can also engage in antagonistic relationships that terminate signaling events or determine substrate fates. A notable example occurs in the brassinosteroid signaling pathway in plants, where phosphorylation of the transcription factor CESTA at serines 75 and 77 directly antagonizes its SUMOylation, thereby controlling subnuclear localization and activity [71]. Although this example involves SUMOylation, similar regulatory mechanisms exist for ubiquitination, where phosphorylation near ubiquitin attachment sites can either promote or inhibit ubiquitin conjugation.
The reciprocal regulation between phosphorylation and ubiquitination also governs protein stability, as exemplified by the tumor suppressor p53. Phosphorylation of p53 at specific residues can either promote its ubiquitination and degradation by MDM2 or stabilize it by preventing ubiquitin ligase binding, depending on the cellular context and modification pattern [24]. This dynamic interplay allows for precise control of p53 levels and activity in response to DNA damage.
Interplay with SUMOylation
Shared Enzymatic Machinery and Substrate Competition
SUMO (Small Ubiquitin-like Modifier) modification represents a ubiquitin-like system that parallels ubiquitination in its enzymatic cascade but serves distinct cellular functions [1]. Similar to ubiquitination, SUMOylation involves E1 (activating), E2 (conjugating), and E3 (ligating) enzymes that sequentially attach SUMO proteins to target substrates [1]. Mammalian cells express three major SUMO isoforms: SUMO1, and the highly similar SUMO2 and SUMO3 (often referred to as SUMO2/3) [1].
This shared enzymatic architecture creates opportunities for cross-talk between SUMOylation and ubiquitination pathways. Competition for identical lysine residues on substrate proteins represents one fundamental mechanism of interplay. SUMO modification can sterically hinder ubiquitin attachment at the same site, thereby regulating substrate stability [67]. Conversely, SUMO-targeted ubiquitin ligases (STUbLs) specifically recognize SUMOylated proteins and promote their ubiquitination and subsequent degradation, demonstrating how sequential modifications can determine protein fate [67].
SUMO-Ubiquitin Coordination in DNA Repair
The DNA damage response employs coordinated SUMOylation and ubiquitination events to regulate repair pathway choice and efficiency. During replication stress, the replication protein A (RPA) subunit RPA70 undergoes SUMOylation, which facilitates the replacement of RPA with RAD51 on single-stranded DNA—a critical step in homologous recombination [1]. The deSUMOylating enzyme SENP6 maintains RPA70 in a hypoSUMOylated state under normal conditions but dissociates upon stress exposure, allowing rapid SUMOylation and subsequent repair protein exchange [1].
This coordinated modification extends to the Fanconi anemia (FA) pathway, where FANCD2 monoubiquitination—essential for its function in interstrand cross-link repair—is regulated by preceding SUMOylation events [68]. The SUMOylation of FA core complex components promotes their assembly and facilitates the subsequent ubiquitination of FANCD2, illustrating how sequential modifications by ubiquitin-like proteins orchestrate complex DNA repair processes.
Figure 1: SUMO-Ubiquitin Cross-Talk in Replication Stress Response. This diagram illustrates the coordinated SUMOylation and ubiquitination events during replication stress response, highlighting the sequential modifications that facilitate homologous recombination repair.
Experimental Analysis of SUMOylation-Ubiquitination Cross-Talk
Protocol: Co-immunoprecipitation of SUMOylated and Ubiquitinated Proteins
Cell Lysis and Preparation: Harvest cells using ice-cold RIPA buffer supplemented with 20mM N-ethylmaleimide (NEM) to inhibit SUMO proteases and 10μM PR619 to inhibit deubiquitinating enzymes [72].
Immunoprecipitation: Incubate cell lysates with antibodies specific to your protein of interest (e.g., α-La antibody for La protein studies) overnight at 4°C with gentle rotation [72].
Bead Capture: Add protein A/G agarose beads and incubate for 2-4 hours at 4°C. Pellet beads by gentle centrifugation (3,000 × g for 5 minutes) [72].
Washing: Wash beads three times with ice-cold lysis buffer containing protease inhibitors but reducing detergent concentration to 0.1% [72].
Elution: Elute immunoprecipitated proteins by boiling in 2× Laemmli buffer for 10 minutes [72].
Western Blot Analysis: Resolve proteins by SDS-PAGE and transfer to PVDF membranes. Probe with SUMO-specific (e.g., α-SUMO2/3) or ubiquitin-specific (e.g., α-Ubiquitin) antibodies to detect modified protein species [72].
Troubleshooting Notes: The detection of SUMOylated proteins can be challenging due to their low abundance and transient nature. Using SUMO protease inhibitors (NEM) during extraction is critical for stabilizing SUMO conjugates. High-molecular-weight bands typically represent SUMOylated or ubiquitinated species, with SUMOylation adding approximately 15-20 kDa per SUMO moiety [72].
Emerging Connections with RNA Modifications
The Landscape of RNA Modifications
RNA modifications represent an emerging layer of gene regulation that intersects with protein PTMs including ubiquitination. Over 100 chemical modifications have been identified in cellular RNA, with N6-methyladenosine (m6A) representing the most abundant internal modification in eukaryotic mRNA [73]. Similar to the ubiquitin system, m6A modification is installed by "writer" complexes (METTL3/METTL14), removed by "eraser" enzymes (FTO, ALKBH5), and interpreted by "reader" proteins (YTHDF1-3, YTHDC1-2) that dictate functional outcomes including RNA stability, splicing, export, and translation [73].
The m6A regulatory machinery itself is subject to control by ubiquitination and other PTMs. Writers, erasers, and readers undergo post-translational modifications including ubiquitination, phosphorylation, SUMOylation, O-GlcNAcylation, and lactylation, creating a multi-layered regulatory network that connects RNA modification with protein degradation and signaling pathways [73].
Ubiquitin-Mediated Control of RNA Modification Machinery
The ubiquitin-proteasome system exerts significant control over the RNA modification machinery by regulating the stability and turnover of key regulatory proteins. METTL3, the catalytic subunit of the m6A methyltransferase complex, is regulated by ubiquitination that targets it for proteasomal degradation [73]. This modification creates a feedback loop that connects RNA modification status with protein stability decisions.
Similarly, the m6A reader proteins, including members of the YTHDF family, are subject to ubiquitin-mediated degradation, allowing dynamic control of m6A interpretation in response to cellular signals [73]. The crosstalk between ubiquitination and RNA modifications thereby enables coordinated regulation of gene expression at both transcriptional and post-transcriptional levels, with implications for cancer progression, neurological disorders, and metabolic syndromes when deregulated [73].
Table 3: Research Reagent Solutions for Studying PTM Cross-Talk
| Reagent/Category | Specific Examples | Function/Application | Experimental Notes |
|---|---|---|---|
| SUMOylation Inhibitors | N-ethylmaleimide (NEM) | SUMO protease inhibitor | Stabilizes SUMO conjugates during protein extraction [72] |
| Ubiquitination Inhibitors | PR619, MG132 | DUB and proteasome inhibitors | Stabilizes ubiquitin conjugates [72] |
| Phosphorylation Modulators | Bikinin, Brassinazole (Brz) | GSK3/shaggy-like kinase inhibitors | Study phosphorylation-dependent regulation [71] |
| Specific Antibodies | α-SUMO2/3, α-Ubiquitin, α-phospho substrates | Detection of modified proteins | Critical for immunoprecipitation and western blot [72] |
| Expression Constructs | Wild-type vs. sumoylation-deficient mutants (K72R) | Functional analysis of modification sites | Identify SUMOylation-dependent functions [71] |
| Proteasome Inhibitors | Bortezomib, Carfilzomib | Block protein degradation | Study ubiquitination-dependent turnover [33] |
Experimental Approaches for Studying K6 Ubiquitin Linkages
Detection and Validation Methods
The study of K6 ubiquitin linkages presents technical challenges due to their relative low abundance compared to K48 and K63 linkages, and the limited availability of linkage-specific reagents. Several experimental approaches have been developed to address these challenges:
Linkage-Specific Antibodies: Generation and validation of K6 linkage-specific ubiquitin antibodies remains a cornerstone approach. These antibodies enable detection of endogenous K6 linkages through western blotting and immunofluorescence, allowing assessment of subcellular localization and dynamics in response to DNA damage [1].
Mass Spectrometry-Based Proteomics: Advanced proteomic methods utilizing linkage-specific antibodies or ubiquitin-binding domains (UBDs) enriched for specific chain types facilitate comprehensive identification of K6-linked substrates. Tandem mass spectrometry can distinguish linkage types through signature fragmentation patterns [1] [69].
In Vitro Reconstitution assays: Purified E1, E2, and E3 enzymes combined with ubiquitin mutants (where all lysines except K6 are mutated to arginine) enable specific examination of K6 chain formation and identification of the enzymatic machinery responsible for their assembly [1].
Functional Analysis of K6 Linkages in DNA Damage
Protocol: Monitoring K6 Ubiquitin Dynamics at DNA Damage Sites
Damage Induction: Treat cells with DNA damaging agents (e.g., ionizing radiation, neocarzinostatin, or etoposide) to induce double-strand breaks [1] [24].
Immunofluorescence Staining: At specific time points post-damage (e.g., 15 seconds to 2 hours), fix cells and stain with:
- K6 linkage-specific ubiquitin antibodies
- DNA damage markers (γH2AX, 53BP1)
- Counterstain for nuclei (DAPI) [24]
Microscopy and Quantification: Acquire high-resolution images using confocal microscopy. Quantify co-localization of K6 ubiquitin signals with DNA damage markers at repair foci [1] [24].
Inhibition Studies: Employ specific E1 inhibitors (MLN7243) or proteasome inhibitors (bortezomib) to dissect the requirement for ubiquitin activation and proteasomal degradation in K6 ubiquitin dynamics [33].
Figure 2: Experimental Workflow for K6 Ubiquitin Analysis at DNA Damage Sites. This diagram outlines the key steps for monitoring K6 ubiquitin dynamics in response to DNA damage, from damage induction to quantitative analysis.
Therapeutic Implications and Future Directions
Targeting PTM Cross-Talk in Cancer Therapy
The intricate cross-talk between ubiquitination and other PTMs offers promising therapeutic targets for cancer treatment. Several strategies have emerged:
Proteasome Inhibitors: Drugs such as bortezomib, carfilzomib, and ixazomib target the proteasome and have achieved clinical success in multiple myeloma and other hematological malignancies by disrupting protein degradation and inducing apoptosis [33].
E1 Enzyme Inhibitors: Compounds like MLN7243 and MLN4924 target ubiquitin-activating enzymes, showing promise in preclinical cancer models by blocking global ubiquitination and inducing DNA damage and apoptosis [33].
E3 Ligase Modulators: Small molecules such as nutlin and MI-219 specifically target MDM2, the primary E3 ligase for p53, leading to p53 stabilization and activation of its tumor suppressor functions in cancers with wild-type p53 [33].
DUB Inhibitors: Compounds targeting deubiquitinating enzymes (e.g., G5 and F6) have shown potential in preclinical studies by modulating the stability of key regulatory proteins [33].
The development of linkage-specific therapeutics represents an emerging frontier. As the distinct functions of different ubiquitin linkages become better characterized, particularly the role of K6 linkages in DNA damage response, opportunities arise for developing more precise interventions that modulate specific aspects of ubiquitin signaling without disrupting the entire system.
Concluding Perspectives
The cross-talk between K6 ubiquitin linkages and other PTMs represents a sophisticated regulatory language that coordinates cellular responses to DNA damage. The integration of phosphorylation, SUMOylation, and RNA modifications with ubiquitin signaling creates a multi-layered control system that enables precise spatiotemporal regulation of DNA repair pathways. Continued elucidation of these complex interactions will not only advance our fundamental understanding of genome maintenance mechanisms but also provide novel therapeutic avenues for diseases characterized by genomic instability, particularly cancer.
Future research directions should focus on developing more specific tools to manipulate K6 linkages, defining the complete enzymatic machinery responsible for their assembly and disassembly, and exploring the potential for targeting K6-specific processes in combination with existing genotoxic therapies to improve cancer treatment outcomes.
K6-linked ubiquitination, once considered an atypical ubiquitin chain modification, has emerged as a critical regulator in maintaining genome integrity and cellular stress response. This whitepaper synthesizes current research establishing the correlation between K6 ubiquitin linkages and disease pathologies, with particular emphasis on cancer development and chemoresistance mechanisms. We examine the specialized role of K6 linkages in DNA damage response pathways, focusing on their function in resolving transcription-replication conflicts and protecting against aldehyde-induced toxicity. The findings presented herein position K6-linked ubiquitination as a promising diagnostic biomarker and therapeutic target in oncology, particularly for tumors characterized by high levels of endogenous DNA damage and replication stress.
Ubiquitination represents a crucial post-translational modification that regulates virtually all cellular processes in eukaryotes. Among the eight distinct ubiquitin chain linkage types, K6-linked ubiquitination has remained one of the least characterized until recent years. K6 linkages are formed when ubiquitin molecules connect through lysine residue at position 6, creating structurally and functionally distinct polyubiquitin signals. Unlike the well-established K48-linked chains that target proteins for proteasomal degradation or K63-linked chains involved in signaling pathways, K6 linkages perform specialized functions in maintaining genome stability and managing proteotoxic stress [74].
The biological significance of K6 ubiquitination is particularly evident in its correlation with human disease pathologies. While all ubiquitin linkages contribute to cellular homeostasis, K6 chains have been specifically implicated in DNA damage response pathways, resolution of transcription-replication conflicts, and protection against proteotoxic stress—processes fundamentally disrupted in cancer and chemoresistance [5] [74]. This whitepaper examines the mechanistic roles of K6 linkages in disease contexts, focusing on their emerging importance as biomarkers and therapeutic targets in oncology.
Mechanistic Insights: K6 Linkages in Genome Integrity
K6 Ubiquitination in DNA Damage Response
K6-linked ubiquitination plays a multifaceted role in DNA damage response (DDR) pathways, serving as a critical signaling mechanism that helps maintain genome integrity. Recent research has identified specific contexts where K6 linkages are preferentially employed to coordinate DNA repair processes and manage replication stress:
Resolution of Transcription-Replication Conflicts: K6-linked ubiquitylation serves as a specific marker for RNA-protein crosslinks (RPCs) induced by formaldehyde, a reactive aldehyde produced by cellular metabolism and accumulated after alcohol consumption. These RPCs stall ribosomes and inhibit translation, creating conflicts between transcriptional and replicative processes. The RING-in-between-RING (RBR) E3 ligase RNF14 catalyzes K6-linked ubiquitylation of these RPCs, marking them for resolution by the ubiquitin- and ATP-dependent unfoldase VCP (p97) in a GCN1-dependent manner. This pathway represents an evolutionary conserved formaldehyde-induced stress response that protects cells against RPC accumulation [5].
Recognition by DNA Damage Response Proteins: The NZF domain of TAK1-binding protein 2 (TAB2) demonstrates specific recognition of K6-linked polyubiquitin chains. Structural analyses reveal that TAB2-NZF simultaneously interacts with both distal and proximal ubiquitin moieties of K6-linked diubiquitin, with a binding mechanism similar to its interaction with K63-linked chains. This dual specificity suggests that K6 linkages may interface with established DNA damage signaling pathways, particularly those involving TAK1 complex activation in response to genotoxic stress [75].
Table 1: E3 Ligases Generating K6-Linked Ubiquitin Chains in DNA Damage Response
| E3 Ligase | Class | Function in DDR | Specific Substrate/Context |
|---|---|---|---|
| RNF14 | RBR E3 ligase | Catalyzes K6-linked ubiquitylation of RNA-protein crosslinks | Formaldehyde-induced RPCs in mRNA |
| TRAF6 | RING E3 ligase | Generates K6 linkages in inflammatory signaling | NF-κB pathway activation |
| HUWE1 | HECT E3 ligase | Produces K6 chains on histone substrates | Chromatin remodeling at DNA breaks |
K6 Linkages in Cancer-Relevant Stress Responses
Beyond direct DNA damage response, K6 ubiquitination contributes to several cancer-relevant cellular stress adaptation mechanisms:
Proteostasis Maintenance: K6-linked chains participate in the regulation of protein quality control systems, particularly under conditions of proteotoxic stress. This function becomes crucial in cancer cells experiencing proteostatic stress due to rapid proliferation, genomic instability, and chemotherapeutic exposure [76].
Metabolic Stress Adaptation: In Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD), which can progress to hepatocellular carcinoma, linkage-specific ubiquitination events regulate key signaling pathways. While K48 and K63 linkages have been more extensively studied in metabolic regulation, emerging evidence suggests K6 linkages contribute to the hepatic stress response in pre-malignant conditions [77].
Inflammatory Signaling Modulation: K6-linked ubiquitination contributes to the regulation of inflammatory response pathways, particularly through its role in TAK1/TAB complex signaling. Since chronic inflammation represents a known cancer risk factor, this connection further links K6 biology to carcinogenesis [75].
Experimental Analysis of K6 Linkages
Key Research Methodologies
Investigating the functions of K6-linked ubiquitination requires specialized methodologies designed to detect and characterize this specific ubiquitin linkage type amid the complex cellular ubiquitin landscape:
Chain-Selective TUBE-Based Affinity Enrichment: Tandem Ubiquitin Binding Entities (TUBEs) with nanomolar affinities for specific polyubiquitin chains enable the selective capture of K6-linked ubiquitinated proteins from complex cellular lysates. These specialized affinity matrices preserve labile ubiquitination events while resisting deubiquitinase activity, allowing for subsequent immunoblotting or mass spectrometric analysis of endogenous target protein ubiquitination. The technology can differentiate context-dependent linkage-specific ubiquitination in high-throughput formats, making it particularly valuable for drug discovery applications [12].
Linkage-Specific Mass Spectrometry Approaches: Advanced proteomic techniques utilizing ubiquitin remnant motifs and selective antibody enrichment enable system-wide identification and quantification of K6 linkage formation. Absolute quantification (AQUA) employing stable isotope-labeled ubiquitin peptides provides precise measurement of K6 chain abundance under different physiological conditions and in response to genotoxic stressors [74].
Structural Characterization Techniques: X-ray crystallography and NMR spectroscopy have been instrumental in elucidating how ubiquitin-binding domains specifically recognize K6-linked chains. The crystal structure of TAB2-NZF in complex with K6-linked diubiquitin resolved at 1.99-Å resolution revealed simultaneous interactions with both distal and proximal ubiquitin moieties, with flexibility in the C-terminal region of the distal ubiquitin contributing to binding specificity [75].
Experimental Workflow for K6 Ubiquitination Analysis
The following diagram illustrates a standardized experimental workflow for investigating K6-linked ubiquitination in cellular models:
Diagram 1: Experimental workflow for K6-linked ubiquitination analysis
Research Reagent Solutions
Table 2: Essential Research Reagents for K6 Linkage Studies
| Reagent/Category | Specific Examples | Function/Application |
|---|---|---|
| K6-Selective TUBEs | K6-TUBE magnetic beads (LifeSensors) | Selective enrichment of K6-ubiquitinated proteins from complex lysates |
| Linkage-Specific Antibodies | Anti-K6 ubiquitin linkage antibodies | Immunodetection of K6 linkages in Western blot and immunofluorescence |
| E3 Ligase Tools | RNF14 expression constructs, mutants | Functional studies of K6 chain assembly |
| Deubiquitinase Probes | Ovarian tumor (OTU) proteases | Investigation of K6 chain disassembly mechanisms |
| Activity-Based Probes | Ubiquitin vinyl sulfones with K6 linkage | Profiling DUBs with K6-linkage specificity |
| Reference Substrates | Formaldehyde-induced RPC models | Standardized cellular models for K6 ubiquitination studies |
K6 Linkages in Cancer and Therapeutic Resistance
The role of K6-linked ubiquitination in cancer pathogenesis and treatment resistance is increasingly recognized, with mechanistic studies revealing both direct and indirect contributions to tumor biology:
Chemoresistance Mechanisms: K6 linkages contribute to chemotherapy resistance through their role in DNA damage tolerance and repair. By facilitating the resolution of transcription-replication conflicts and DNA-protein crosslinks, K6 ubiquitination enables cancer cells to survive genotoxic insults from chemotherapeutic agents. This protective function becomes maladaptive in oncology contexts, where it undermines the efficacy of DNA-damaging chemotherapy [5] [76].
Regulation of Apoptosis and Ferroptosis: Ubiquitination networks, including K6-linked chains, regulate programmed cell death pathways in cancer cells. While K48 and K63 linkages have been more extensively characterized in apoptosis regulation, emerging evidence suggests K6 linkages contribute to the fine-tuning of cell death decisions under stress conditions, potentially influencing chemosensitivity [76].
ABC Transporter Regulation: K6 ubiquitination may contribute to the regulation of ABC transporter family members, which mediate multidrug resistance in cancer cells by actively effusing chemotherapeutic agents. While K48 and K63 linkages have been more directly implicated, the potential involvement of K6 chains in modulating transporter activity or turnover represents an area of active investigation [76].
K6-linked ubiquitination has transitioned from a poorly understood ubiquitin modification to a recognized critical player in genome maintenance pathways with direct relevance to cancer biology. The established correlation between K6 linkages and disease mechanisms, particularly in the context of DNA damage response and chemoresistance, highlights the therapeutic potential of targeting this pathway in oncology.
Future research directions should focus on developing selective small-molecule inhibitors targeting K6-specific E3 ligases like RNF14, exploring combination therapies that sensitize cancer cells to conventional DNA-damaging agents, and establishing K6 linkage signatures as biomarkers for patient stratification. Additionally, further elucidating the structural basis of K6 chain recognition by specific ubiquitin-binding domains may enable the development of protein-protein interaction inhibitors that disrupt pathogenic K6-mediated signaling in cancer cells.
As our understanding of the ubiquitin code continues to evolve, K6-linked ubiquitination represents a promising frontier for therapeutic intervention in cancer and other diseases characterized by genome instability and proteotoxic stress.
Ubiquitination is a crucial post-translational modification that regulates virtually every cellular process in eukaryotes, with the specific topology of ubiquitin chains constituting a complex "ubiquitin code" that determines functional outcomes [78]. Among the diverse ubiquitin linkages, K63-linked chains have been well-characterized in DNA damage response signaling, while K6-linked chains have remained more enigmatic until recent discoveries [1] [8]. This review provides a comparative analysis of distinct E3 ligase systems—the RNF8/RNF168 axis responsible for K63-linked ubiquitination and RNF14 which facilitates atypical K6-linked ubiquitination. Understanding the specialized functions, mechanisms, and biological consequences of these pathways provides critical insights for targeted therapeutic development in cancer and other diseases associated with genome instability.
Fundamentals of Ubiquitin Signaling
The Ubiquitination Machinery
Ubiquitination involves a sequential enzymatic cascade:
- E1 (ubiquitin-activating enzyme): Activates ubiquitin in an ATP-dependent manner [8]
- E2 (ubiquitin-conjugating enzyme): Accepts ubiquitin from E1 [8]
- E3 (ubiquitin ligase): Recognizes specific substrates and catalyzes ubiquitin transfer [8]
E3 ligases constitute the most diverse component, with over 600 members in humans, and confer substrate specificity through several structural architectures [8]:
- RING-finger type: The largest class, functions as scaffolds bringing E2 and substrate together
- HECT type: Forms catalytic thioester intermediate with ubiquitin before transfer
- RBR type: Hybrid mechanism with RING and HECT-like characteristics
- U-box type: Structurally similar to RING but stabilized by different interactions
Ubiquitin Chain Linkages and Functional Consequences
Ubiquitin contains seven lysine residues (K6, K11, K27, K29, K33, K48, K63) and an N-terminal methionine that can form polyubiquitin chains with distinct structures and functions [1] [78]. The linkage specificity fundamentally determines the biological outcome:
- K48-linked chains: Primarily target substrates for proteasomal degradation [8] [65]
- K63-linked chains: Mainly regulate signaling transduction, protein interactions, and subcellular trafficking [65]
- K6-linked chains: Considered "atypical" and implicated in DNA damage response and mitochondrial function [13] [8]
- Other linkages (K11, K27, K29, K33, M1): Perform specialized regulatory functions [8]
Table 1: Major Ubiquitin Linkage Types and Their Primary Functions
| Linkage Type | Primary Functions | Key E3 Ligases | Cellular Processes |
|---|---|---|---|
| K48-linked | Proteasomal degradation | Various | Protein turnover, homeostasis |
| K63-linked | Signaling, trafficking | RNF8, RNF168, TRAF6 | DNA repair, inflammation, endocytosis |
| K6-linked | DNA damage response, mitophagy | RNF14, BRCA1, PARKIN | DNA crosslink repair, mitochondrial quality control |
| K11-linked | Cell cycle regulation | APC/C | Mitotic progression, ER-associated degradation |
| M1-linked | NF-κB signaling | LUBAC | Inflammation, immunity |
The RNF8/RNF168 Axis in K63-Linked Ubiquitination
Pathway Mechanism and Signaling Amplification
The RNF8/RNF168 pathway represents a well-defined signaling module that initiates and amplifies the DNA damage response through K63-linked ubiquitination [79]. This pathway activates in response to DNA double-strand breaks (DSBs) through a carefully orchestrated sequence:
- Damage Recognition: ATM kinase phosphorylates histone H2AX and mediator protein MDC1 at DSB sites [79] [80]
- RNF8 Recruitment: RNF8 binds phosphorylated MDC1 through its FHA domain [79] [81]
- Initial Ubiquitination: RNF8 cooperates with E2 enzyme UBC13 to generate K63-linked ubiquitin chains on histones H2A and H2AX [81] [80]
- RNF168 Recruitment: RNF168 recognizes RNF8-generated K63-ubiquitin chains through its Ub-dependent DSB recruitment module 1 (UDM1) [79] [80]
- Signal Amplification: RNF168 ubiquitylates histone H2A at K13/K15, creating binding platforms for downstream repair factors [79] [82]
- Effector Recruitment: Repair proteins including 53BP1, BRCA1, and RAD18 accumulate at damage sites [79] [81]
K63-Ubiquitin Signaling Pathway in DNA Damage Response
Structural Basis of K63 Chain Recognition
RNF168 contains specialized binding modules that confer specificity for K63-linked ubiquitin chains [80]. The UDM1 domain (Ub-dependent DSB recruitment module 1) in the N-terminal region recognizes RNF8-generated K63 chains through a cooperative mechanism:
- UMI domain: Binds ubiquitin with moderate affinity but limited linkage specificity
- LRM1 motif: Serves as an auxiliary element that enhances binding specificity for K63 linkages
- Combined interaction: The LRM1-UMI unit exhibits significantly stronger affinity for K63-diUb (Kd = 45 μM) compared to K48-diUb (Kd = 435 μM) [80]
The UDM2 domain in the C-terminal region recognizes RNF168's own ubiquitination products, establishing a positive feedback loop for signal amplification [79] [80]. UDM2 contains:
- MIU2 domain: Binds monoubiquitin and RNF168-generated H2A ubiquitination
- LRM2 motif: Interacts directly with the nucleosome surface, particularly the acidic patch
- UAD domain: Recently identified as essential for distal K63 chain recognition
Structural studies reveal that both UDM1 and UDM2 fold as single α-helices that simultaneously contact both the proximal and distal ubiquitin moieties in K63-linked chains, explaining their linkage specificity [80].
Biological Consequences and Disease Associations
Defects in the RNF8/RNF168 pathway cause RIDDLE syndrome, characterized by radiosensitivity, immunodeficiency, dysmorphic features, and learning difficulties [79]. Cellular and physiological consequences include:
- Impaired DSB repair: Failed recruitment of 53BP1 and BRCA1 to damage sites [79]
- Genomic instability: Increased chromosomal aberrations and mutation rates [81]
- Cell sensitivity: Hypersensitivity to ionizing radiation and crosslinking agents [79]
- Immunodeficiency: Defective class switch recombination in B lymphocytes [79]
RNF168 mutations identified in RIDDLE syndrome patients include frameshift mutations (c.397dupG, c.1323_1326delACCA, c.295delG) that result in premature truncation and loss-of-function [79].
RNF14 in K6-Linked Ubiquitination
Mechanism of Translation-Coupled Quality Control
Recent discoveries have revealed RNF14 as a key mediator of K6-linked ubiquitination in response to RNA damage and translational stress [13]. Unlike the chromatin-focused RNF8/RNF168 pathway, RNF14 operates in translation quality control:
- Damage Induction: Reactive aldehydes (e.g., formaldehyde) or photoactivatable ribonucleosides cause RNA-protein crosslinks (RPCs) [13]
- Translation Stall: Crosslinked mRNAs stall elongating ribosomes, leading to ribosome collisions [13]
- RNF14 Activation: Collided ribosomes activate RNF14 in coordination with RNF25 [13]
- Atypical Ubiquitination: RNF14 mediates K6- and K48-linked ubiquitination of crosslinked proteins [13]
- Proteasomal Degradation: Ubiquitinated protein adducts are extracted and degraded [13]
This pathway represents a translation-coupled quality control mechanism that specifically resolves covalent crosslinks between proteins and mRNAs (mRPCs) [13].
RNF14-Mediated K6-Ubiquitination in RNA Damage Response
Functional Specialization in Stress Response
RNF14-mediated K6 ubiquitination represents a specialized adaptation to aldehyde stress with several distinctive features:
- RNA damage focus: Specifically responds to RNA lesions rather than DNA damage [13]
- Translation coupling: Activated specifically by ribosome collisions during translation [13]
- Dual linkage capability: Generates both K6- and K48-linked ubiquitin chains [13]
- Proteasomal targeting: Despite atypical K6 linkages, facilitates proteasomal degradation [13]
This pathway works alongside canonical DNA damage response mechanisms, highlighting the multifaceted cellular defense against aldehyde-induced crosslinking damage [13].
Comparative Analysis: Key Experimental Findings
Quantitative Binding and Functional Parameters
Table 2: Comparative Analysis of E3 Ligase Properties and Functions
| Parameter | RNF8 | RNF168 | RNF14 |
|---|---|---|---|
| Primary Ubiquitin Linkage | K63-linked | K63-linked | K6- and K48-linked |
| Chain Type Function | Signaling scaffold | Signaling scaffold & amplification | Proteasomal targeting |
| Key Binding Domains | FHA domain, RING | UDM1, UDM2, RING | RING, unidentified substrate-binding |
| Kd for Cognate Chains | Not determined | UDM1: K63-diUb Kd = 45 μM [80] | Not determined |
| Cellular Process | DNA damage response | DNA damage response | Translation quality control |
| Activation Trigger | DNA double-strand breaks | RNF8-generated K63 chains | Ribosome collisions |
| Disease Associations | Not firmly established | RIDDLE syndrome | Not firmly established |
| Experimental Models | Knockout MEFs, knockdown | RIDDLE patient cells, knockout | PAR-CL, FA treatment |
Methodologies for Pathway Analysis
Analyzing RNF8/RNF168 K63-Ubiquitination
Immunofluorescence Microscopy for Repair Foci
- Purpose: Visualize recruitment of RNF168 and downstream effectors to DNA damage sites [79] [81]
- Method:
- Induce DSBs with ionizing radiation (e.g., 2-10 Gy) or laser microirradiation
- Fix cells at specific timepoints (e.g., 0.5-8 hours post-irradiation)
- Stain with antibodies against RNF168, 53BP1, BRCA1, or γH2AX
- Quantify focus formation and colocalization
- Key Reagents: Anti-RNF168, anti-53BP1, anti-γH2AX antibodies [79]
In Vitro Ubiquitination Assays
- Purpose: Characterize E3 ligase activity and linkage specificity [80]
- Method:
- Purify recombinant E1, E2 (UBC13), E3 (RNF8 or RNF168), and ubiquitin
- Set up reactions with ATP regeneration system
- Incubate at 30°C for 1-2 hours
- Analyze products by Western blot or mass spectrometry
- Variations: Use mutant ubiquitin (K63R, K48R) to determine linkage specificity [80]
Surface Plasmon Resonance (SPR) for Binding Affinity
- Purpose: Quantify interactions between ubiquitin chains and binding domains [80]
- Method:
- Immobilize K63-diUb or other linkage types on SPR chip
- Flow purified UDM1, UDM2, or other domains at varying concentrations
- Measure binding kinetics and calculate Kd values
- Confirm specificity with control ubiquitin linkages (K48, M1)
- Key Findings: UDM1 Kd for K63-diUb = 45 μM; for K48-diUb = 435 μM [80]
Analyzing RNF14 K6-Ubiquitination
Photoactivatable Ribonucleoside-Enhanced Crosslinking (PAR-CL)
- Purpose: Induce specific RNA damage without confounding DNA damage [13]
- Method:
- Metabolically label cellular RNA with 4-thiouridine (4-SU, 5 μM, 16 hours)
- Induce crosslinking with UVA irradiation (365 nm, 6 kJ/m²)
- Assess viability and downstream signaling responses
- Validate specific RPC formation via protein-x-linked RNA extraction (XRNAX)
- Key Advantage: Models formaldehyde-induced RNA damage without DNA damage [13]
XRNAX (Protein-X-linked RNA Extraction) Protocol
- Purpose: Isolate and quantify RNA-protein crosslinks [13]
- Method:
- Lyse cells under denaturing conditions
- Digest uncrosslinked RNA with RNase T1
- Purify crosslinked RNA-protein complexes by silica-based membrane adsorption
- Identify crosslinked proteins by mass spectrometry
- Application: Identified 561 proteins commonly crosslinked by both PAR-CL and formaldehyde [13]
Phosphoproteomics for Pathway Activation
- Purpose: Monitor global signaling responses to RNA damage [13]
- Method:
- Perform TMT-based multiplexed quantitative phosphoproteomics
- Enrich phosphopeptides from PAR-CL or formaldehyde-treated cells
- Identify significantly altered phosphosites via LC-MS/MS
- Perform kinase-substrate enrichment analysis
- Key Finding: PAR-CL activates ZAKα- and GCN2-dependent stress responses [13]
Research Reagent Solutions
Table 3: Essential Research Reagents for E3 Ligase Studies
| Reagent Category | Specific Examples | Research Application | Key Function |
|---|---|---|---|
| Cell Models | RNF8-/- MEFs [81], RNF168 patient cells [79], ZAK KO cells [13] | Genetic requirement studies | Define pathway hierarchy and genetic dependencies |
| Ubiquitin Mutants | K63R ubiquitin, K48R ubiquitin, K6R ubiquitin [13] [81] | Linkage specificity studies | Determine chain type requirement for specific processes |
| Crosslinking Agents | 4-thiouridine (4-SU) [13], Formaldehyde (0.5 mM) [13] | Damage induction models | Induce specific RNA or DNA damage |
| Activation Markers | p-p38 antibodies [13], γH2AX antibodies [79] | Pathway activation readouts | Monitor stress response activation |
| E3 Ligase Constructs | ZAKαWT, ZAKαK45A [13], RNF168 UDM mutants [80] | Structure-function studies | Delineate functional domains and catalytic requirements |
| Proteomics Tools | TMT multiplexing reagents [13], XRNAX protocol [13] | Global analysis | Unbiased identification of pathway components |
Discussion: Therapeutic Implications and Future Directions
The distinct mechanistic features of K6 and K63 ubiquitin linkages underscore the functional diversity of ubiquitin signaling in maintaining cellular homeostasis. The RNF8/RNF168 axis represents a canonical DNA damage signaling pathway that amplifies repair signals through sequential K63 ubiquitination, while RNF14 exemplifies a specialized quality control mechanism that resolves translational stress through atypical K6/K48 ubiquitination.
From a therapeutic perspective, these pathways offer attractive targets for cancer therapy. RNF168 inhibition could potentially sensitize tumors to radiation and genotoxic chemotherapeutics by disrupting DNA repair [79]. Conversely, RNF14 activation might enhance cellular resilience against aldehyde stress, potentially relevant for degenerative conditions and chemotherapy-induced toxicity.
Future research should address several critical questions:
- What are the precise structural determinants of RNF14's dual specificity for K6 and K48 linkages?
- How is RNF14 specifically activated by ribosome collisions?
- Are there additional E3 ligases specialized for other atypical ubiquitin linkages?
- Can we develop linkage-specific ubiquitin probes to monitor these pathways in live cells?
Answering these questions will not only advance our fundamental understanding of ubiquitin signaling but also unlock new opportunities for targeted therapeutic intervention in cancer, neurodegenerative diseases, and other conditions characterized by proteostasis disruption.
Evolutionary Conservation of the K6-Ubiquitin Pathway Across Model Organisms
The K6-linked ubiquitin pathway represents an evolutionarily ancient system of post-translational modification with specialized functions in genome maintenance. Once considered a minor ubiquitin linkage, K6-ubiquitylation has emerged as a critical signaling mechanism in the DNA damage response (DDR), particularly in resolving specific forms of DNA damage. This whitepaper examines the conservation of the K6-ubiquitin pathway from prokaryotic antecedents to mammalian systems, highlighting its role in formaldehyde-induced stress response and its implications for therapeutic development. We integrate quantitative data on linkage specificity, experimental methodologies for studying K6 chains, and visualization of the conserved pathway components to provide a comprehensive resource for researchers and drug development professionals.
Ubiquitylation is a multifaceted post-translational modification that regulates virtually every cellular process, with the functional outcome determined by the topology of ubiquitin chains formed on substrate proteins. Among the eight possible ubiquitin chain linkages (K6, K11, K27, K29, K33, K48, K63, and M1), K6-linked ubiquitylation has remained one of the less characterized but increasingly important modifications in maintaining genome integrity [1] [11]. The DNA damage response (DDR) employs a sophisticated network of ubiquitin-dependent signaling pathways to detect, signal, and repair DNA lesions, with K6-linkages playing specialized roles in these processes [24] [68].
Recent research has established that K6-linked ubiquitylation serves as a critical mark for the resolution of formaldehyde-induced RNA-protein crosslinks (RPCs), representing a dedicated cellular defense mechanism against reactive aldehydes [5]. This pathway exemplifies how distinct ubiquitin linkages can be deployed for specific types of cellular stress, with K6-chains acting as a recognition signal for the processing of transcription-blocking lesions. The evolutionary conservation of this pathway from lower eukaryotes to mammals underscores its fundamental importance in cellular homeostasis and genome protection.
Molecular Mechanisms of K6-Linked Ubiquitylation
Enzymatic Machinery and Signaling Specificity
The formation of K6-linked ubiquitin chains involves a dedicated enzymatic cascade that confers specificity to this modification:
E1-E2-E3 Enzymatic Hierarchy: Like all ubiquitylation events, K6-chain formation initiates with ubiquitin activation by E1 enzymes, transfer to E2 conjugating enzymes, and final substrate modification by E3 ligases that determine linkage specificity [1] [24]. The RING-in-between-RING (RBR) E3 ligase RNF14 has been identified as a key enzyme that catalyzes K6-linked ubiquitylation in response to formaldehyde-induced RPCs [5].
Chain Recognition and Decoding: K6-linked chains are recognized by specific ubiquitin-binding domains (UBDs) that translate the modification into functional outcomes. In the case of RPC resolution, the VCP/p97 segregase complex recognizes K6-marked crosslinks and facilitates their extraction from chromatin or RNA for processing [5].
Linkage Topology and Functional Consequences: The three-dimensional structure of K6-linked chains creates unique binding interfaces that distinguish them from the well-characterized K48 (proteasomal degradation) and K63 (signaling) linkages. This distinct topology enables the recruitment of specialized effector proteins that initiate damage-specific responses [1] [11].
K6-Ubiquitin in Formaldehyde-Induced Stress Response
Recent research has elucidated a dedicated pathway for K6-linked ubiquitylation in response to formaldehyde-induced lesions:
Diagram Title: K6-Ub Pathway in RPC Resolution
This pathway illustrates how reactive aldehydes, including those produced by cellular metabolism and alcohol consumption, induce RPCs that stall translating ribosomes. The stalling event triggers RNF14-dependent K6-linked ubiquitylation of crosslinked proteins, marking them for recognition and processing by VCP in a GCN1-RNF14-VCP-dependent manner [5].
Evolutionary Conservation of the K6-Ubiquitin Pathway
Prokaryotic Antecedents and Early Evolution
The ubiquitin signaling system has deep evolutionary roots, with prokaryotic antecedents revealing the ancient origin of K6-linked ubiquitylation:
Ubiquitin Fold Conservation: The characteristic β-grasp fold of ubiquitin is present in prokaryotic ubiquitin-like proteins (ThiS, MoaD, TGS, and YukD domains), demonstrating structural conservation across domains of life [83]. These prokaryotic Ubls share the core structural architecture that enables conjugation to target molecules.
Early E1-Like Enzymes: Prokaryotic adenylating enzymes (E1-like; MoeB/ThiF family) represent the evolutionary precursors of eukaryotic E1 enzymes and already contained the Rossmann-fold nucleotide-binding domain and C-terminal catalytic domain characteristic of their eukaryotic counterparts [83].
Primitive Signaling Systems: Conserved gene neighborhoods in diverse bacteria combine JAB domain peptidases (deubiquitinating enzymes), E1-like adenylating enzymes, and ubiquitin-related proteins, suggesting the existence of primitive ubiquitylation-mediated signaling pathways in prokaryotes [83].
Conservation Across Eukaryotic Lineages
The K6-ubiquitin pathway demonstrates remarkable conservation throughout eukaryotic evolution:
Table 1: Evolutionary Conservation of K6-Ubiquitin Pathway Components
| Component | Prokaryotic Antecedents | Lower Eukaryotes | Mammals | Functional Conservation |
|---|---|---|---|---|
| Ubiquitin Fold | ThiS, MoaD, TGS domains [83] | Ubiquitin | Polyubiquitin genes (UBB, UBC, UBA52, UBA80) [84] | Structural conservation maintained |
| E1 Enzymes | MoeB/ThiF family [83] | UBA1-3 | UBA1-6 | Activation mechanism conserved |
| E2 Enzymes | Limited counterparts | Multiple E2s | ~40 E2 enzymes [84] | Catalytic core conserved |
| E3 Ligases | RING/HECT-like domains | RBR E3s | RNF14, RBR family [5] | Substrate recognition diversified |
| K6 Linkage | Possible primitive forms | Present | Functional specialization [5] | DNA damage signaling |
Metazoan Conservation: Analysis of E2 ubiquitin-conjugating enzymes across metazoan phyla reveals high sequence conservation, suggesting that the core ubiquitylation machinery existed in its current form in the last common metazoan ancestor [84]. Chordata and Echinodermata, which diverged over 0.5 billion years ago, share significant homology in E3 ligase sequences with humans.
Functional Diversification: While the core enzymatic machinery is conserved, the regulatory complexity of the ubiquitin system has expanded through evolution, particularly in E3 ligases and adaptor proteins that determine pathway specificity [84]. The K6-linkage has likely maintained its specialized function in DNA damage response while acquiring additional roles in higher eukaryotes.
Diagram Title: Evolution of K6-Ub Pathway Components
Experimental Analysis of K6-Linked Ubiquitylation
Quantitative Proteomic Approaches
Advanced mass spectrometry-based proteomics has been instrumental in characterizing K6-linked ubiquitylation events:
Table 2: Quantitative Proteomic Methods for K6-Linkage Analysis
| Method | Principle | Application to K6 Studies | Advantages | Limitations |
|---|---|---|---|---|
| SILAC (Stable Isotope Labeling with Amino acids in Cell Culture) | Metabolic incorporation of stable isotopes [85] | Relative quantification of K6-linked ubiquitylation changes | Accurate quantification; minimal sample processing | Requires metabolic activity; limited multiplexing |
| TMT (Tandem Mass Tagging) | Isobaric tags for multiplexed quantification [85] | Parallel analysis of K6 substrates across conditions | High multiplexing capacity (up to 10-30 samples) | Signal compression issues requiring MS3 |
| Ubiquitin Remnant Profiling | Antibody enrichment of diGly-modified peptides [85] | Proteome-wide identification of K6 ubiquitylation sites | Comprehensive substrate identification | Cannot distinguish linkage types directly |
| Linkage-Specific Antibodies | Immunoaffinity purification of linkage types | Selective isolation of K6-linked chains | Direct analysis of K6 topology | Limited availability; potential cross-reactivity |
Detailed Experimental Protocol: K6-Ubiquitylation Analysis in DNA Damage Response
Objective: To identify and quantify K6-linked ubiquitylation events in response to formaldehyde-induced DNA damage.
Materials and Reagents:
- Cell culture system (e.g., HEK293, HeLa, or primary mammalian cells)
- Formaldehyde treatment solution (freshly prepared in PBS)
- Proteasome inhibitor (MG132, 10μM)
- Deubiquitylase inhibitor (PR-619, 25μM)
- Lysis buffer (6M guanidine-HCl, 100mM Na₂HPO₄/NaH₂PO₄, 10mM Tris-HCl, pH 8.0)
- K6-linkage specific antibody (commercial sources) or pan-ubiquitin antibody
- Protein A/G magnetic beads
- SILAC kits (light, medium, heavy amino acids)
- Trypsin/Lys-C mix for proteolytic digestion
Procedure:
Cell Culture and Treatment:
- Culture cells in SILAC media containing light (L), medium (M), or heavy (H) isotopic forms of lysine and arginine for at least 6 population doublings.
- Treat heavy-labeled cells with formaldehyde (100-500μM, 2-6 hours), medium-labeled cells with proteasome inhibitor (MG132, 4 hours), and maintain light-labeled cells as untreated controls.
- Include deubiquitylase inhibitor (PR-619) during the last 30 minutes of treatment to preserve ubiquitin chains.
Ubiquitin Enrichment:
- Harvest cells and lyse in denaturing buffer (6M guanidine-HCl) at 95°C for 10 minutes to inactivate enzymes.
- Pool equal protein amounts from each SILAC condition.
- Perform ubiquitin affinity enrichment using either:
- K6-linkage specific antibody coupled to magnetic beads (overnight, 4°C)
- Pan-ubiquitin antibody for total ubiquitin enrichment
- Wash beads extensively with lysis buffer followed by PBS-based wash buffer.
Proteomic Sample Preparation:
- On-bead digest with trypsin/Lys-C (1:50 enzyme:substrate, 37°C, 16 hours).
- Desalt peptides using C18 solid-phase extraction.
- Fractionate peptides using high-pH reverse-phase chromatography to reduce complexity.
LC-MS/MS Analysis:
- Analyze fractions by nanoLC-MS/MS using Orbitrap Fusion Lumos or similar high-resolution instrument.
- Use HCD fragmentation with MS3 for TMT-based quantification to minimize ratio compression.
- Implement stepped collision energy (25-35%) to improve ubiquitin chain signature fragmentation.
Data Analysis:
- Search MS/MS data against appropriate database (UniProt) using search engines (MaxQuant, Proteome Discoverer).
- Include variable modifications: GlyGly-Lys (ubiquitylation), oxidation (M), acetylation (protein N-term).
- Apply false discovery rate (FDR) threshold of 1% at peptide and protein levels.
- Normalize SILAC or TMT ratios and perform statistical analysis for significant changes.
Troubleshooting Notes:
- For K6-specific analysis, validate antibody specificity using linkage-defined ubiquitin chains.
- Include negative controls without crosslinker to distinguish damage-specific events.
- Optimize formaldehyde concentration and exposure time to avoid overwhelming the repair system.
Research Reagent Solutions for K6-Ubiquitin Studies
Table 3: Essential Research Reagents for K6-Ubiquitin Pathway Investigation
| Reagent Category | Specific Examples | Function in K6 Studies | Commercial Sources |
|---|---|---|---|
| Linkage-Specific Antibodies | Anti-K6-linkage ubiquitin; Pan-ubiquitin (clone FK2) | Immunodetection and enrichment of K6 chains | Cell Signaling Technology; Millipore Sigma |
| E3 Ligase Tools | RNF14 expression plasmids; RNF14 siRNA/shRNA | Manipulate K6 chain formation | Addgene; Dharmacon |
| Deubiquitylase Inhibitors | PR-619; WP1130 | Preserve endogenous K6 chains during analysis | Sigma-Aldrich; Selleck Chem |
| Activity Probes | K6-linkage specific DUB probes; Ubiquitin vinyl sulfone | Monitor DUB activity against K6 chains | UbiQ Bio; LifeSensors |
| Defined Ubiquitin Chains | Homogeneous K6-linked diUb/triUb; K6-only ubiquitin mutants | Standard curves and specificity controls | UbiQ Bio; Boston Biochem |
| Mass Spec Standards | SILAC kits; TMTpro 16-plex | Quantitative proteomic analysis | Thermo Scientific; Cambridge Isotopes |
| Cell Lines | RNF14 KO; VCP mutant lines | Pathway validation and functional studies | ATCC; commercial KO collections |
Discussion and Research Implications
Therapeutic Targeting Opportunities
The conservation and specificity of the K6-ubiquitin pathway present attractive opportunities for therapeutic intervention:
Cancer Therapy Applications: Given the role of K6-linked ubiquitylation in resolving transcription-blocking lesions, targeting this pathway could sensitize cancer cells to chemotherapeutic agents that induce RPCs or similar DNA damage [86]. Specific inhibition of RNF14 or its co-factors might enhance the efficacy of aldehyde-generating chemotherapeutics.
Neurodegenerative Disease Connections: Defects in DNA damage response pathways, including ubiquitin signaling, contribute to neurodegenerative diseases [68]. Modulation of K6-dependent repair mechanisms may offer neuroprotective strategies in conditions characterized by transcription stress.
Specificity Advantages: The relatively specialized nature of K6 signaling compared to K48 or K63 pathways offers potential for targeted therapeutic manipulation with reduced off-target effects on general proteostasis or signaling pathways.
Future Research Directions
Several key questions remain regarding the K6-ubiquitin pathway and its evolutionary conservation:
Additional Physiological Contexts: While the role in formaldehyde-induced RPC resolution is established, other physiological stressors and DNA lesion types likely employ K6-linked signaling, requiring further exploration.
Regulation of Branching: K6 linkages can form part of branched ubiquitin chains with other linkage types (e.g., K6/K11, K6/K48) [11], creating complex regulatory signals that remain poorly understood in DNA damage contexts.
Cross-talk with Other Modifications: The interplay between K6-ubiquitylation and other post-translational modifications (phosphorylation, SUMOylation, ADP-ribosylation) in coordinating DNA repair requires systematic investigation [23] [68].
The K6-linked ubiquitin pathway represents an evolutionarily conserved mechanism for maintaining genome integrity, with specialized functions in resolving transcription-blocking DNA lesions. From prokaryotic antecedents to mammalian systems, the core components of this pathway have been maintained while acquiring additional regulatory complexity. The recent identification of RNF14 as a K6-specific E3 ligase in formaldehyde stress response provides a paradigm for understanding how distinct ubiquitin linkages direct specific DNA repair outcomes. Continued investigation of this pathway using quantitative proteomics, genetic approaches, and chemical biology tools will further elucidate its mechanisms and therapeutic potential, particularly in contexts of cancer and degenerative diseases where DNA damage response pathways are compromised.
Conclusion
K6-linked ubiquitination has emerged from obscurity to be recognized as a crucial, specialized mechanism within the DNA damage response, particularly in resolving transcription-associated damage like RNA-protein crosslinks. This review has synthesized evidence establishing a dedicated RNF14-VCP pathway that uses K6 chains to mark and resolve RPCs, thereby preventing genomic instability. The development of chain-specific tools, such as TUBEs, has been instrumental in advancing the field, though challenges in detecting this low-abundance modification remain. When placed in the context of the broader 'ubiquitin code,' K6 linkages represent a non-canonical, context-specific signal distinct from the degradative K48 and signaling K63 chains. Future research must focus on identifying the full repertoire of K6 substrates, elucidating its interplay with the epitranscriptome, and exploring its therapeutic potential. Targeting the K6 pathway offers a promising, novel strategy for sensitizing cancer cells to genotoxic therapies, especially in tumors with high metabolic or environmental exposure to crosslinking agents.
References
- 1. The ubiquitous role of ubiquitin in the DNA damage response [https://pmc.ncbi.nlm.nih.gov/articles/PMC7105183/]
- 2. Breaking the K48-Chain: Linking Ubiquitin Beyond Protein ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11730971/]
- 3. What are Atypical Ubiquitin Chains? [https://www.news-medical.net/life-sciences/What-are-Atypical-Ubiquitin-Chains.aspx]
- 4. Investigating the function of atypical ubiquitylation in ... [https://gepris.dfg.de/gepris/projekt/241324827?language=en]
- 5. K6-linked ubiquitylation marks formaldehyde-induced RNA ... [https://www.sciencedirect.com/science/article/pii/S1097276523008481]
- 6. DNA damage-induced histone H1 ubiquitylation is ... [https://www.nature.com/articles/s41598-017-15194-y]
- 7. Assembly, analysis and architecture of atypical ubiquitin ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4176834/]
- 8. E3 ubiquitin ligases: styles, structures and functions - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC8607428/]
- 9. Regulation of the DNA damage response by ubiquitin ... [https://www.frontiersin.org/journals/genetics/articles/10.3389/fgene.2015.00098/full]
- 10. Atypical K6-ubiquitin chains mobilize p97/VCP and the ... [https://www.sciencedirect.com/science/article/pii/S1097276523009541]
- 11. Emerging functions of branched ubiquitin chains [https://www.nature.com/articles/s41421-020-00237-y]
- 12. Unraveling chain specific ubiquitination in cells using ... [https://www.nature.com/articles/s41598-025-07242-9]
- 13. RNF14-dependent atypical ubiquitylation promotes translation ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10783637/]
- 14. K6-linked ubiquitylation marks formaldehyde-induced RNA ... [https://pubmed.ncbi.nlm.nih.gov/37951215/]
- 15. The Ubiquitin Proteasome System in Genome Stability and ... [https://www.mdpi.com/2072-6694/13/9/2235]
- 16. Beyond K48 and K63: non-canonical protein ubiquitination [https://cmbl.biomedcentral.com/articles/10.1186/s11658-020-00245-6]
- 17. Exogenous and endogenous formaldehyde-induced DNA ... [https://link.springer.com/article/10.1007/s10565-024-09926-w]
- 18. Ubiquitin Linkage-Specific Affimers Reveal Insights into K6 ... [https://www.sciencedirect.com/science/article/pii/S1097276517306172]
- 19. Structural basis for specific recognition of K6-linked ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8391078/]
- 20. An E3 ligase network engages GCN1 to promote degradation ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9994462/]
- 21. The Ubiquitin Proteasome System in Genome Stability and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8125356/]
- 22. protein K6-linked ubiquitination Gene Ontology Term (GO ... [https://www.informatics.jax.org/vocab/gene_ontology/GO:0085020]
- 23. Epitranscriptome in action: RNA modifications in the DNA ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12044609/]
- 24. Regulation of the DNA damage response by ubiquitin ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4354423/]
- 25. Ubiquitin Linkage-Specific Affimers Reveal Insights into K6 ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5640506/]
- 26. An Interaction Landscape of Ubiquitin Signaling [https://www.sciencedirect.com/science/article/pii/S1097276517300047]
- 27. Current methodologies in protein ubiquitination characterization [https://cellandbioscience.biomedcentral.com/articles/10.1186/s13578-022-00870-y]
- 28. Ubiquitination: Mechanisms, Mass Spectrometry, and the Role in Cellular Regulation - Creative Proteomics [https://www.creative-proteomics.com/proteomics/protein-science/ubiquitination-mechanisms-mass-spectrometry-role-cellular-regulation.html]
- 29. Mass Spectrometry-Based Proteomics for Investigating ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4905943/]
- 30. Proteome solubility is differentially reshaped by thermal stress ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12391699/]
- 31. Weighing in on Ubiquitin: The Expanding Role of Mass ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC1224607/]
- 32. The role of ubiquitination in tumorigenesis and targeted drug ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7048745/]
- 33. The role of ubiquitination in tumorigenesis and targeted ... [https://www.nature.com/articles/s41392-020-0107-0]
- 34. Preserving Genome Integrity and Function: The DNA Damage ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6715524/]
- 35. Phase separation of ERCC6L2–CtIP regulates the extent ... [https://www.nature.com/articles/s41556-025-01760-4]
- 36. Genomic Reporter Constructs to Monitor Pathway-Specific ... [https://www.frontiersin.org/journals/genetics/articles/10.3389/fgene.2021.809832/full]
- 37. DSB repair pathway choice is regulated by recruitment of ... [https://www.sciencedirect.com/science/article/pii/S2211124721001546]
- 38. CRISPR-Cas9 Knockout Screens Identify DNA Damage ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11240649/]
- 39. Targeted protein degradation: mechanisms, strategies and ... [https://www.nature.com/articles/s41392-022-00966-4]
- 40. Review Molecular glues and PROTACs in targeted protein ... [https://www.sciencedirect.com/science/article/abs/pii/S0006295225005623]
- 41. Targeted Protein Degradation: Principles and Applications ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10378610/]
- 42. Molecular Glues: The Adhesive Connecting Targeted Protein ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9910052/]
- 43. Molecular Glues: A New Dawn After PROTAC [https://www.biochempeg.com/article/271.html]
- 44. E3 ubiquitin ligases in signaling, disease, and therapeutics [https://www.sciencedirect.com/science/article/pii/S0968000425001860]
- 45. Detection of ubiquitination activity and identification ... [https://www.sciencedirect.com/science/article/abs/pii/S0076687918305287]
- 46. Branched Ubiquitination: Detection Methods, Biological ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7664869/]
- 47. Quantitative Proteomic Atlas of Ubiquitination and Acetylation in the... [https://pubmed.ncbi.nlm.nih.gov/26051181/]
- 48. Frontiers | Mass Spectrometry-Based Proteomics for Investigating DNA ... [https://www.frontiersin.org/journals/genetics/articles/10.3389/fgene.2016.00109/full]
- 49. Ubiquitin-Dependent and Independent Proteasomal ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10536765/]
- 50. Quantitative Proteomic Atlas of Ubiquitination and ... [https://www.sciencedirect.com/science/article/pii/S1097276515003421]
- 51. DNA damage response signaling pathways and targets for ... [https://www.nature.com/articles/s41392-020-0150-x]
- 52. Non-proteolytic ubiquitylation in cellular signaling and ... [https://www.nature.com/articles/s42003-022-03060-1]
- 53. Interplay between Ubiquitylation and SUMOylation at DNA ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2989631/]
- 54. Ubiquitin and SUMO pathways in DNA replication ... [https://pubmed.ncbi.nlm.nih.gov/41134662/]
- 55. Comprehensive interrogation of synthetic lethality in the ... [https://www.nature.com/articles/s41586-025-08815-4]
- 56. Mechanism of Lys6 poly-ubiquitin specificity by the L. ... [https://www.sciencedirect.com/science/article/pii/S1097276522011352]
- 57. Current methodologies in protein ubiquitination ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9373315/]
- 58. Enhanced validation of antibodies for research applications [https://www.nature.com/articles/s41467-018-06642-y]
- 59. An integrated technology for quantitative wide mutational ... [https://www.nature.com/articles/s41467-024-48072-z]
- 60. Breaking the K48-chain: linking ubiquitin beyond protein ... [https://www.nature.com/articles/s41594-024-01221-w]
- 61. A cryptic K48 ubiquitin chain binding site on UCH37 is ... [https://elifesciences.org/articles/76100]
- 62. K63 ubiquitination in immune signaling - PMC - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC8755460/]
- 63. The K48-K63 Branched Ubiquitin Chain Regulates NF-κB ... [https://www.sciencedirect.com/science/article/pii/S1097276516305639]
- 64. K48- and K63-linked ubiquitin chain interactome reveals ... [https://www.life-science-alliance.org/content/7/8/e202402740]
- 65. Role of K63-linked ubiquitination in cancer [https://www.nature.com/articles/s41420-022-01204-0]
- 66. Ubiquitin-mediated DNA damage response is synthetic ... [https://www.nature.com/articles/s41598-021-88988-w]
- 67. An inventory of crosstalk between ubiquitination and other ... [https://www.sciencedirect.com/science/article/pii/S258900422300353X]
- 68. The DNA damage response pathways: at the crossroad of ... [https://www.nature.com/articles/cr2007109]
- 69. Crosstalk and Interplay between the Ubiquitin-Proteasome ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5547213/]
- 70. Delineating Crosstalk Mechanisms of the Ubiquitin ... [https://www.frontiersin.org/journals/cell-and-developmental-biology/articles/10.3389/fcell.2018.00011/full]
- 71. Interplay between phosphorylation and SUMOylation ... [https://www.nature.com/articles/ncomms5687]
- 72. SUMO Modification of the RNA-Binding Protein La ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5748460/]
- 73. RNA Modifications in Health and Disease - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC12409077/]
- 74. Ubiquitin modifications | Cell Research [https://www.nature.com/articles/cr201639]
- 75. Article Structural basis for specific recognition of K6-linked ... [https://www.sciencedirect.com/science/article/pii/S000634952100549X]
- 76. The advancement of ubiquitination regulation in apoptosis ... [https://www.sciencedirect.com/science/article/abs/pii/S0003986125002103]
- 77. Ubiquitination and ubiquitin-like modifications in metabolic ... [https://www.bmbreports.org/journal/view.html?doi=10.5483/BMBRep.2025-0014]
- 78. The ubiquitin codes in cellular stress responses - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC10903993/]
- 79. RNF168 and the DNA damage response pathway - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC8486888/]
- 80. Structural insights into two distinct binding modules for ... [https://www.nature.com/articles/s41467-017-02345-y]
- 81. RNF8-Independent Lys63 Poly-Ubiquitylation Prevents ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0089997]
- 82. Mechanisms of RNF168 nucleosome recognition and ... [https://www.sciencedirect.com/science/article/pii/S1097276523010821]
- 83. The prokaryotic antecedents of the ubiquitin-signaling system ... [https://genomebiology.biomedcentral.com/articles/10.1186/gb-2006-7-7-r60]
- 84. Tracking of Ubiquitin Signaling through 3.5 Billion Years of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11354881/]
- 85. Quantifying Ubiquitin Signaling - PMC - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC4441763/]
- 86. Ubiquitin E3 ligases in cancer: somatic mutation and ... [https://www.bmbreports.org/view.html?uid=1819&vmd=Full]