Structural Insights into K33-Linked Diubiquitin: Conformation, Recognition, and Functional Implications
This article provides a comprehensive analysis of the crystal structure of K33-linked diubiquitin, an atypical ubiquitin chain type with emerging roles in non-proteolytic cellular signaling.
Structural Insights into K33-Linked Diubiquitin: Conformation, Recognition, and Functional Implications
Abstract
This article provides a comprehensive analysis of the crystal structure of K33-linked diubiquitin, an atypical ubiquitin chain type with emerging roles in non-proteolytic cellular signaling. We explore the distinct conformational states of K33 chains, from compact diubiquitin to extended polymers, and the methodological advances enabling their study, including the identification of the HECT E3 ligase AREL1 for specific chain assembly. The content details the structural basis for selective recognition by binding domains like the NZF1 domain of the deubiquitinase TRABID and discusses the challenges in probing these chains. By comparing K33 linkages with other ubiquitin chain types and validating their roles in specific biological contexts—such as T-cell receptor signaling and protein trafficking—this review synthesizes key structural principles to inform future research and therapeutic targeting in immunology and neurodegeneration.
Unraveling the Architecture: Core Structural Features of K33-Linked Diubiquitin
Crystal Structure Reveals a Compact Diubiquitin Conformation
Ubiquitin chains, linked via different lysine residues, constitute a sophisticated post-translational code that regulates diverse cellular processes, from protein degradation to inflammatory signaling. Among the eight possible ubiquitin chain linkages, those formed via lysine 33 (K33) have remained one of the least characterized. This application note details the structural and biophysical analysis of K33-linked diubiquitin, focusing on its compact conformational state as revealed by X-ray crystallography. The findings presented herein provide crucial methodological frameworks and structural insights for researchers investigating atypical ubiquitin chains in signaling and drug discovery contexts.
The crystal structure of K33-linked diubiquitin (PDB ID: 4XYZ) was determined at 1.65 Å resolution, revealing a compact conformation with extensive interdomain contacts [1] [2]. This structure provides the first atomic-level view of this atypical ubiquitin linkage.
Table 1: Crystallographic Data Collection and Refinement Statistics for 4XYZ
| Parameter | Value |
|---|---|
| Resolution | 1.65 Å |
| Space Group | P 1 21 1 |
| Unit Cell Dimensions | a=29.48 Å, b=57.02 Å, c=33.98 Å, β=95.45° |
| R-value Work | 0.162 |
| R-value Free | 0.214 |
| Ligands Present | PG4 (Tetraethylene glycol), EDO (1,2-ethanediol), ACT (Acetate ion), IOD (Iodide ion) |
| Organism | Bos taurus (Cattle) |
| Deposition Date | 2015-02-03 |
The structure shows that the K33 linkage facilitates a specific orientation between the proximal (donor) and distal (acceptor) ubiquitin monomers. The isopeptide bond connects the C-terminal glycine (G76) of the proximal ubiquitin to the lysine 33 (K33) side chain of the distal ubiquitin [3]. This configuration allows the two ubiquitin moieties to pack closely, burying a significant surface area at the interface formed by their characteristic β-grasp folds.
Conformational Dynamics and Linkage Dependence
Biophysical studies beyond crystallography reveal that K33-linked chains are not restricted to a single conformation but exist in a dynamic equilibrium.
Solution Studies and Computational Analyses
Nuclear Magnetic Resonance (NMR) spectroscopy and Molecular Dynamics (MD) simulations demonstrate that K33-linked diubiquitin samples an ensemble of conformations in solution, ranging from compact to more extended states [4]. This inherent flexibility is a common feature among several atypical ubiquitin linkages.
Table 2: Conformational States of Different Diubiquitin Linkages
| Linkage Type | Predicted/Observed Conformation | Experimental Evidence |
|---|---|---|
| K33 | Compact (crystalline) / Open & Dynamic (solution) | X-ray (4XYZ), NMR, MD Simulations [1] [5] [4] |
| K48 | Closed | X-ray, NMR, Modeling [6] |
| K63 | Extended | X-ray, NMR, Modeling [6] |
| K6 | Can form closed conformation | Modeling [6] |
| K11 | Can form closed conformation | Modeling [6] |
| K29 | Open & Dynamic / Unable to form closed conformation | NMR, MD Simulations, Modeling [5] [6] |
| K27 | Can form closed conformation (to a lesser extent) | Modeling [6] |
| Linear (M1) | Extended | X-ray, Modeling [6] |
The compact conformation observed in the 4XYZ crystal structure is stabilized by specific crystal packing forces and represents one low-energy state sampled by the chain. Intriguingly, crystallographic analysis of K33-linked triubiquitin reveals a more extended conformation, suggesting that chain length can influence the overall architecture [1]. This conformational plasticity is a key feature that likely enables the specific recognition of K33 chains by downstream effector proteins in the cell.
Experimental Protocols
Enzymatic Assembly of K33-Linked Ubiquitin Chains
A primary challenge in studying atypical ubiquitin chains has been producing homogeneously linked polymers in sufficient quantities. The following protocol, adapted from Michel et al. (2015), enables the generation of K33-linked chains for structural studies [5] [7] [8].
Protocol: Large-Scale Assembly and Purification of K33-Linked Ubiquitin Chains
Principle: The HECT E3 ligase AREL1 (Apoptosis-Resistant E3 Ubiquitin Protein Ligase 1) exhibits a strong preference for assembling K33- and K11-linked ubiquitin chains. Combining AREL1 with linkage-selective deubiquitinases (DUBs) allows for the purification of homotypic K33-linked chains.
Materials:
- E1 Ubiquitin-Activating Enzyme
- E2 Conjugating Enzyme (e.g., UbcH5 family)
- HECT E3 Ligase AREL1 (436-823 aa fragment): The key enzyme conferring linkage specificity [5].
- Ubiquitin: Wild-type for assembly; mutant (e.g., K33-only) for specificity controls.
- Energy Regeneration System: ATP, Mg²⁺.
- Reaction Buffer: 50 mM Tris-HCl (pH 7.5), 50 mM NaCl, 10 mM MgCl₂, 2 mM ATP.
- Linkage-Selective DUBs: For trimming and purifying homogeneous chains (e.g., TRABID domains for K29/K33) [5] [8].
- Chromatography Resins: Ion-exchange (e.g., MonoQ), Size-exclusion (e.g., Superdex 75).
Procedure:
- Enzyme Charging: Incubate E1 (100 nM), E2 (5 µM), and ubiquitin (50 µM) in reaction buffer with ATP at 37°C for 15 minutes.
- Chain Assembly: Initiate polymerization by adding AREL1 E3 ligase (200 nM). Allow the reaction to proceed for 2-4 hours at 37°C.
- Reaction Monitoring: Analyze a small aliquot by SDS-PAGE and anti-ubiquitin immunoblotting to confirm high-molecular-weight chain formation.
- DUB Treatment: To obtain specific chain lengths (e.g., diubiquitin), treat the reaction mixture with a catalytic domain of a linkage-specific DUB. The N-terminal NZF1 domain of TRABID shows high specificity for K29/K33 linkages and can be used for selective binding and purification [5] [8].
- Purification:
- Terminate the reaction by adding DUB or by shifting to low temperature (4°C).
- Separate the chains by anion-exchange chromatography (MonoQ column), using a salt gradient for elution. K33-linked chains elute at characteristic concentrations.
- Further purify by size-exclusion chromatography (Superdex 75) to isolate discrete chain lengths (e.g., diubiquitin).
- Validation: Verify linkage specificity using Absolute Quantification (AQUA) mass spectrometry with isotope-labeled GlyGly-modified standard peptides [5].
Crystallization and Structure Determination
The following workflow outlines the key steps for determining the three-dimensional structure of K33-linked diubiquitin via X-ray crystallography, based on the deposition of PDB 4XYZ [1] [2].
Key Reagents and Parameters for Crystallization:
- Protein: Homogeneously linked K33-diubiquitin, purified to >95% homogeneity and concentrated to 10-20 mg/mL in low-salt buffer (e.g., 10 mM Tris pH 7.5).
- Crystallization Screen: Employ commercial sparse-matrix screens (e.g., Hampton Research). PDB 4XYZ crystallized in space group P 1 21 1 [1].
- Cryoprotectant: Paratone-N or similar, for flash-cooling in liquid nitrogen.
- Data Collection: Synchrotron X-ray source recommended for high-resolution data. For 4XYZ, data were processed with XDS and scaled with SCALA [1].
- Phasing and Refinement: Molecular Replacement using a standard ubiquitin monomer (e.g., PDB 1UBQ) as a search model. Refinement is performed using programs like REFMAC or PHENIX [1].
The Scientist's Toolkit: Key Research Reagents
Table 3: Essential Reagents for K33-Linked Ubiquitin Chain Research
| Reagent / Tool | Function / Role | Example / Source |
|---|---|---|
| HECT E3 Ligase AREL1 | Linkage-specific assembly of K33- and K11-linked chains; core component of enzymatic assembly system. | Human, aa 436-823 [5] |
| TRABID NZF1 Domain | K29/K33-linkage specific binding; used for affinity purification, pull-down assays, and structural studies of chain-receptor complexes. | Human, recombinant [5] [8] |
| Linkage-specific DUBs | Hydrolyze specific ubiquitin linkages; essential for trimming chains and validating linkage identity. | OTU family DUBs (e.g., TRABID) [1] [5] |
| Ubiquitin Mutants (Kx-only) | Contain only a single lysine residue; critical for controlling linkage specificity in assembly assays. | K33-only Ub (all other lysines mutated to Arg) [5] |
| Isotope-labeled Ubiquitin | Required for NMR-based structural studies (e.g., ^15^N, ^13^C) and AQUA mass spectrometry. | Recombinant expression in E.coli |
| Anti-K33 Linkage Antibodies | Immunodetection of endogenous K33-linked chains in cell lysates and tissue samples. | Commercial and academic sources |
Biological Recognition and Functional Implications
The compact conformation of K33-linked diubiquitin presents a unique surface for recognition by specific ubiquitin-binding domains (UBDs). A key finding is that the Npl4-like zinc finger 1 (NZF1) domain of the deubiquitinase TRABID specifically recognizes K29- and K33-linked diubiquitin [5] [8]. The crystal structure of the NZF1-K33-diubiquitin complex reveals an extended binding mode where the NZF1 domain engages the interface between the two ubiquitin moieties [5] [7]. This explains the linkage specificity of TRABID and provides a model for how readers of the ubiquitin code can discriminate between structurally similar atypical chains. Disruption of this interaction, via point mutations in the NZF1 domain, attenuates the localization of TRABID to ubiquitin-rich puncta in cells, underscoring the physiological relevance of this specific recognition event [5].
The crystal structure of K33-linked diubiquitin provides a foundational resource for unraveling the biology of this atypical ubiquitin signal. The compact conformation observed, alongside its dynamic behavior in solution, highlights the structural versatility of ubiquitin chains. The enzymatic and structural protocols detailed here offer a roadmap for researchers to produce and characterize K33-linked chains, thereby accelerating the discovery of their physiological roles, receptors, and potential as therapeutic targets in human disease.
Within the intricate field of ubiquitin signaling, the three-dimensional structure of polyubiquitin chains is a fundamental determinant of their functional specificity. K33-linked diubiquitin (K33-diUb) represents a classic example of a dynamic, multi-domain protein whose biological activity is intrinsically linked to its conformational landscape. Once considered a poorly characterized "atypical" linkage, K33 chains are now known to play specific roles in non-proteolytic processes such as protein trafficking and immune regulation [9] [10]. For instance, the Cul3-KLHL20 ubiquitin E3 ligase catalyzes K33-linked polyubiquitination of coronin 7, a modification essential for directing protein trafficking from the trans-Golgi network by facilitating F-actin assembly and transport carrier biogenesis [9]. A comprehensive understanding of these functions requires a detailed grasp of K33-diUb's structural dynamics, which range from compact dimers to extended filaments. This Application Note provides a structured framework for the production, analysis, and interpretation of K33-diUb conformations, serving as an essential resource for structural biologists and pharmacologists aiming to decipher this complex ubiquitin code.
Research Reagent Solutions
The following table catalogues essential reagents for the study of K33-linked ubiquitin chains, as identified in the literature.
Table 1: Key Research Reagents for K33-linked Ubiquitin Studies
| Reagent Name | Type | Key Function in K33 Research | Specific Example/Source |
|---|---|---|---|
| AREL1 (E3 Ligase) | HECT E3 Ubiquitin Ligase | Assembles K33-linked chains in vitro via autoubiquitination and on substrates [5]. | Human AREL1 (aa 436-823) [5]. |
| Cul3-KLHL20 (E3 Ligase) | Cullin RING E3 Ligase Complex | Catalyzes cellular K33-polyubiquitination of specific substrates like coronin 7 [9]. | Cul3-KLHL20 complex [9]. |
| TRABID NZF1 Domain | Ubiquitin Binding Domain (UBD) | Specifically binds K29/K33-linked diUb; tool for linkage detection and structural studies [5]. | N-terminal NZF1 domain of TRABID [5]. |
| K33-linkage Affimer | Engineered Affinity Reagent | Binds K33- (and K11-) linked chains for detection in techniques like pull-downs and microscopy [11]. | Structure-guided, improved affimer reagent [11]. |
| Myosin VI MyUb Domain | Ubiquitin Binding Domain (UBD) | Binds K63-, K11-, and K29-linked chains; demonstrates potential for cross-reactivity with K33 linkages [12]. | Compact helix-turn-helix motif in myosin VI [12]. |
K33-linked Chain Assembly and Purification
Enzymatic Assembly
The production of pure, homogeneous K33-linked polyubiquitin chains is a critical first step for biophysical characterization. The HECT family E3 ligase AREL1 (Apoptosis Resistant E3 Ubiquitin Protein Ligase 1, also known as KIAA0317) has been identified as a primary enzyme for generating these chains.
Protocol: In Vitro Assembly of K33-linked Chains using AREL1
Reaction Setup: Combine the following components in a suitable reaction buffer:
- Ubiquitin (WT or mutant, 1-10 µM)
- E1 activating enzyme (100 nM)
- E2 conjugating enzyme (UbCH5B or similar, 5 µM)
- Recombinant AREL1 HECT domain (aa 436-823, 1 µM) [5]
- ATP regeneration system
Incubation: Incubate the reaction at 30°C for 2-4 hours to allow for chain elongation.
Linkage Verification: Analyze a portion of the reaction via quantitative mass spectrometry (AQUA). AREL1 assembly reactions with WT ubiquitin typically yield a mixture containing approximately 36% K33, 36% K11, and 20% K48 linkages, confirming its primary specificity for K33 linkages [5].
Linkage-Specific Purification
To obtain chains of a single linkage type for structural studies, a purification strategy employing linkage-specific deubiquitinases (DUBs) is required.
Protocol: DUB-based Purification of K33-linked Ubiquitin Chains
Reaction with K33-specific DUBs: Treat the crude assembly reaction with a DUB that selectively cleaves contaminating linkages but leaves K33 chains intact. The K29/K33-specific DUB TRABID is a suitable candidate [5].
Chromatographic Separation: Purify the cleaved and uncleaved products using ion-exchange or size-exclusion chromatography (SEC). K33-linked chains will elute at volumes corresponding to their molecular weight.
Validation: Verify the linkage and homogeneity of the purified chains using techniques such as:
- Non-reducing SDS-PAGE: To visualize discrete chain lengths.
- Tandem Mass Spectrometry: For definitive confirmation of the K33 linkage.
Diagram 1: K33 Chain Assembly and Purification Workflow.
Analyzing Conformational Dynamics
K33-linked diubiquitin does not adopt a single, rigid structure but exists as a dynamic ensemble of conformations in solution. The following integrated approach allows for atomic-resolution characterization of this heterogeneity.
Nuclear Magnetic Resonance (NMR) Spectroscopy
NMR is a powerful technique for studying protein dynamics and transient structures in near-physiological conditions.
Protocol: Characterizing K33-diUb Dynamics via NMR
Sample Preparation: Produce uniformly (^{15})N- and (^{13})C-labeled K33-diUb using the assembly and purification protocols above. The protein should be in a suitable NMR buffer (e.g., 20 mM phosphate, 50 mM NaCl, pH 6.8).
Paramagnetic Relaxation Enhancement (sPRE):
Backbone Assignment and Relaxation:
- Perform triple-resonance experiments (HNCACB, CBCA(CO)NH) for backbone resonance assignment.
- Collect (^{15})N spin relaxation data (T1, T2, heteronuclear NOE) to probe picosecond-to-nanosecond timescale dynamics of the polypeptide backbone.
Chemical Shift Perturbation (CSP): Map chemical shift changes relative to monomeric ubiquitin to identify regions involved in the inter-domain interface or experiencing conformational exchange.
Molecular Dynamics (MD) Simulations
MD simulations provide a computational counterpart to experimental data, offering atomic-level detail and temporal resolution for conformational sampling.
Protocol: Dual-Scale MD Simulations of K33-diUb
System Setup:
- Construct an initial model of K33-diUb. Crystal structures of the TRABID NZF1:K33-diUb complex can serve as a starting point for the covalently linked Ub units [5].
Multi-Scale Simulation:
- Coarse-Grained (CG) Simulation: Perform long-timescale (microsecond-millisecond) simulations to extensively sample the large-scale conformational space accessible to the two ubiquitin domains.
- All-Atom (AA) Simulation: Use snapshots from the CG trajectory to initiate more detailed, atomistic simulations in explicit solvent.
Ensemble Analysis and Validation:
- Apply dimensionality reduction techniques (e.g., t-Distributed Stochastic Neighbor Embedding, t-SNE) to cluster the simulated conformations.
- Calculate theoretical sPRE and relaxation parameters from the MD ensemble and validate them against the experimental NMR data [4] [13]. This integrative step is crucial for ensuring the simulated ensemble accurately reflects the solution behavior.
Diagram 2: Integrative NMR and MD Workflow.
Structural Interpretation and Functional Relevance
The application of the above protocols reveals key structural features of K33-diUb that underpin its biological function.
Solution Conformation and Crystal Structures
Biophysical analysis shows that K33-linked chains, unlike the compact K48-linked chains, predominantly adopt open and dynamic conformations in solution, similar to K63-linked chains [5]. This extended conformation allows for unique protein interactions distinct from those of degradative ubiquitin signals.
The relationship between solution ensembles and crystal structures can be complex. For K33-diUb, crystal structures of ligand-free molecules often sample the central, more populated regions of the conformational space. In contrast, crystal structures of K33-diUb in complex with specific binding partners (e.g., the TRABID NZF1 domain) represent more sparsely sampled, stabilized conformations that are "selected" by the ligand [4] [13].
Key Interaction Modules
The biological readout of K33 linkages is mediated by specific ubiquitin-binding domains (UBDs). A seminal discovery is the specific recognition of K29/K33-linked chains by the N-terminal NZF1 domain of the deubiquitinase TRABID [5]. The crystal structure of NZF1 bound to K33-diUb reveals an extended filamentous structure where the NZF1 domain binds across the Ub-Ub interface, engaging distinct surfaces on both ubiquitin moieties. This binding mode explains the exquisite linkage specificity of TRABID and provides a model for how K33 chains can be recognized in cells [5] [8].
Table 2: Summary of K33-diUb Conformational and Interaction Data
| Analysis Method | Key Finding for K33-diUb | Functional Implication |
|---|---|---|
| NMR sPRE & MD | Adopts a heterogeneous ensemble of open, dynamic conformations [4]. | Accessibility for non-proteasomal signaling partners; functional plasticity. |
| X-ray Crystallography | NZF1 domain of TRABID binds at the Ub-Ub interface, stabilizing a specific conformation [5]. | Molecular basis for linkage-specific recognition by readers and erasers. |
| AQUA Mass Spectrometry | AREL1 E3 ligase assembles ~36% K33 linkages in vitro [5]. | Identification of a dedicated assembly enzyme for biochemical tool production. |
| Cellular Pull-Downs | K33-affimer reagents identify proteins modified with K33 chains in cells [11]. | Tool for uncovering novel physiological substrates and pathways. |
The move from viewing K33-diUb as a static entity to understanding it as a dynamic ensemble marks a significant advancement in structural biology. The protocols outlined herein—combining the enzymatic generation of chains with integrative NMR and MD analysis—provide a robust roadmap for characterizing its conformational landscape at atomic resolution. The finding that K33 chains are extended and flexible, yet capable of engaging in highly specific, high-affinity interactions with domains like TRABID's NZF1, illustrates the sophisticated nature of the ubiquitin code. For drug development professionals, these dynamic interfaces and their linkage-specific recognition mechanisms represent challenging yet promising targets for therapeutic intervention in trafficking-related diseases and immune disorders. Mastering these structural concepts and techniques is therefore paramount for advancing the field of ubiquitin pharmacology.
Key Intermolecular Contacts Stabilizing the K33 Linkage Interface
Within the intricate signaling code of the ubiquitin system, K33-linked polyubiquitin chains represent one of the less characterized "atypical" linkages. Understanding the specific three-dimensional architecture and the intermolecular contacts that define the K33-linked diubiquitin (K33-diUb) interface is fundamental to elucidating its distinct cellular functions, which are emerging in immune signaling and protein trafficking. This Application Note details the structural features and key interactions at the K33 linkage interface, providing validated experimental protocols for the production, biophysical analysis, and structural determination of K33-diUb. The information herein is framed within a broader thesis on the crystal structure analysis of K33-linked ubiquitin chains, aiming to equip researchers with the tools to decipher this complex post-translational modification.
K33-linked chains, in their ligand-free state, adopt open and dynamic conformations in solution, as revealed by small-angle neutron scattering and NMR studies [5] [14]. This open conformation is similar to that observed for K63-linked chains and is distinct from the compact structure of K48-linked chains. The conformational ensemble indicates significant flexibility, which may be crucial for its specific recognition by signaling proteins. The core structural unit for investigation is the isopeptide-linked diubiquitin, where the C-terminal glycine (G76) of the distal ubiquitin moiety is connected to the epsilon-amino group of lysine 33 on the proximal ubiquitin moiety.
Table 1: Key Biophysical Properties of K33-diUb
| Property | Characteristic | Experimental Method | Significance |
|---|---|---|---|
| Overall Conformation | Open and dynamic | Solution NMR, SANS [5] [14] | Similar to K63 chains; suggests non-proteolytic functions |
| Structural Heterogeneity | Adopts multiple conformers | NMR ensemble generation [14] | Flexibility may allow recognition by multiple partners |
| Ligand-Bound State | Resembles crystal structure conformation | X-ray crystallography [14] | Induces a more defined structure upon receptor binding |
Key Intermolecular Contacts at the K33 Interface
The specific recognition of K33 linkages is mediated by ubiquitin-binding domains (UBDs) that engage the unique surface created by the Ub-Ub interface. A primary example is the N-terminal Npl4-like zinc finger (NZF1) domain of the deubiquitinase TRABID [5].
The TRABID NZF1-K33-diUb Complex
Crystal structure analysis reveals that the TRABID NZF1 domain binds K33-diUb with high specificity, engaging both ubiquitin subunits simultaneously [5]. The binding mode explains the linkage specificity of TRABID for both K29 and K33 linkages. The interaction forms an extended, filamentous structure in the crystal, where each NZF1 domain binds across every Ub-Ub interface in the polyubiquitin chain. While the precise atomic-level contacts from the crystal structure are not fully detailed in the provided results, the structural data confirms a bivalent binding mode that is critical for selectivity.
General Principles of K33 Interface Recognition
The interaction between the K33 linkage and its specific receptors, such as TRABID's NZF1 domain, relies on contacts with surfaces on both the proximal and distal ubiquitin molecules. This bivalent interaction is a common mechanism for achieving linkage specificity. The NZF domain itself is a compact UBD of approximately 30 amino acids that can provide two ubiquitin-binding interfaces, imposing linkage specificity to explain signaling outcomes [15]. The conservation of secondary interaction surfaces in many NZF domains suggests a broader mechanism for achieving specificity, not only through chain linkage but also via simultaneous recognition of the ubiquitinated substrate itself [15].
Experimental Protocols
Protocol 1: Enzymatic Assembly of K33-Linked Polyubiquitin Chains
This protocol describes the use of the HECT E3 ligase AREL1 for generating K33-linked chains, adapted from Michel et al. [5].
Table 2: Reagents for K33-linked Chain Assembly
| Component | Source | Catalog Number / Details |
|---|---|---|
| E1 Activating Enzyme | Commercially available | UBE1 (e.g., LifeSensors, E-300) |
| E2 Conjugating Enzyme | Commercially available | Specific E2 paired with AREL1 (consult literature) |
| E3 Ligase (AREL1) | Recombinant expression | Human AREL1 (aa 436–823) [5] |
| Wild-type Ubiquitin | Commercially available | Ub (e.g., LifeSensors, U-100) |
| Reaction Buffer | - | 50 mM Tris-HCl (pH 7.5), 50 mM NaCl, 10 mM MgCl₂, 2 mM ATP |
| DUBs (for purification) | Commercially available | Linkage-specific DUBs to remove other chain types |
Procedure:
- Setup Reaction: In a 50 µL volume, combine 200 nM E1, 5 µM E2, 2 µM AREL1, and 50 µM ubiquitin in reaction buffer.
- Incubate: Conduct the reaction at 30°C for 2-3 hours.
- Terminate: Stop the reaction by adding 5 mM EDTA.
- Purify Chains: To isolate pure K33-linked chains, treat the assembly reaction with a panel of linkage-specific deubiquitinases (DUBs) that cleave all non-K33 linkages. Subsequently, purify the DUB-resistant K33 chains using size-exclusion chromatography (SEC) or ion-exchange chromatography.
- Validate: Confirm chain linkage and purity using SDS-PAGE and mass spectrometry (e.g., middle-down MS analysis [16]).
Protocol 2: Determining Conformational Dynamics via Solution NMR
This protocol outlines the use of NMR spectroscopy to characterize the conformational ensemble of K33-diUb, based on the work of Castañeda et al. [14].
Procedure:
- Sample Preparation: Prepare a 0.3-0.5 mM sample of purified K33-diUb in a low-ionic-strength NMR buffer (e.g., 20 mM sodium phosphate, pH 6.5). Use a 90% H₂O/10% D₂O mixture or 99.9% D₂O for locking.
- Data Collection:
- Acquire ²H-¹⁵N HSQC spectra to obtain a fingerprint of the protein's backbone amides.
- Perform ¹⁵N heteronuclear NOE experiments to probe backbone dynamics on ps-ns timescales.
- Collect ¹⁵N R₁ and R₂ relaxation data to investigate rotational correlation times and ms-µs dynamics.
- Utilize residual dipolar couplings (RDCs) by aligning the sample in a weak alignment medium (e.g., Pf1 phage).
- Data Analysis:
- Compare chemical shifts in the K33-diUb spectrum with those of free ubiquitin to identify perturbed residues at the interface.
- Analyze relaxation parameters to quantify flexibility.
- Use RDCs and other restraints for in silico ensemble generation to create a population-weighted model of the chain's dynamic conformations in solution.
Protocol 3: Structural Analysis of the K33 Interface by Crystallography
This protocol describes the strategy for obtaining a crystal structure of K33-diUb in complex with a specific binding domain, such as the TRABID NZF1 domain [5].
Procedure:
- Complex Formation: Mix purified K33-diUb with the TRABID NZF1 domain in a 1:1.2 molar ratio. Incubate on ice for 30 minutes.
- Crystallization: Screen for crystallization conditions using commercial sparse matrix screens. Optimize initial hits by varying pH, precipitant concentration, and temperature.
- Data Collection and Processing: Flash-cool crystals in liquid nitrogen using a suitable cryoprotectant. Collect a complete X-ray diffraction dataset at a synchrotron beamline. Index, integrate, and scale the diffraction data.
- Structure Determination:
- Molecular Replacement: Use the high-resolution structure of monomeric ubiquitin (PDB: 1UBQ) as a search model.
- Model Building and Refinement: Iteratively build the model into the electron density map using Coot and refine with Phenix or Refmac. Include the isopeptide bond between G76 of the distal Ub and K33 of the proximal Ub.
- Analysis of the Interface: Analyze the refined structure to identify specific intermolecular contacts, including hydrogen bonds, salt bridges, and van der Waals interactions between the NZF1 domain and both subunits of K33-diUb.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagent Solutions for K33-Linked Ubiquitin Research
| Reagent / Tool | Function / Application | Example Use |
|---|---|---|
| HECT E3 Ligase AREL1 | Linkage-specific assembly of K33 chains | In vitro reconstitution of K33-linked polyubiquitin [5]. |
| Tandem Ubiquitin Binding Entities (TUBEs) | High-affinity capture of polyubiquitinated proteins; K63-specific available. | Protection of chains from DUBs; enrichment for mass spectrometry or Western blot [17] [18]. |
| TRABID NZF1 Domain | Specific recognition of K29/K33 linkages | Structural studies (crystallography, NMR) to define the K33 interface [5]. |
| Linkage-Specific DUBs | Analytical tool to confirm chain linkage; purification aid. | Validation of K33 linkage in cellular assays or in vitro reactions [5]. |
| Ubiquitin Mutants (e.g., K29R, K33R) | Dissecting complex ubiquitination patterns. | Determining specificity of E3 ligases or ubiquitin receptors [5] [16]. |
| AlphaFold with Covalent Linkers | Computational modeling of polyubiquitin complexes. | Predicting structures of K33-linked chains and their complexes with receptors [19]. |
Visualizing Workflows and Interactions
K33-diUb Research Workflow
K33 Linkage Recognition by NZF Domain
The K33-linked ubiquitin chain interface is stabilized by a specific set of intermolecular contacts that are selectively recognized by specialized receptor domains like the NZF1 domain of TRABID. The dynamic and open conformation of the chain in solution presents a unique binding surface that is distinct from other linkage types. The experimental protocols and tools outlined in this Application Note provide a robust framework for researchers to produce, characterize, and determine the high-resolution structure of K33-linked diubiquitin and its complexes. Advancing our understanding of these key intermolecular contacts will be crucial for elucidating the physiological and pathological roles of K33 ubiquitination and for exploring its potential as a target in drug discovery.
Comparison of Diubiquitin vs. Triubiquitin Structures for K33 Linkages
Within the versatile post-translational modification system governed by ubiquitin, the topology of polyubiquitin chains is a critical determinant of functional outcome. K33-linked chains represent one of the less characterized "atypical" linkages, with emerging roles in non-proteolytic signaling processes such as T cell receptor regulation and protein trafficking [20] [9]. This application note provides a detailed structural comparison between K33-linked diubiquitin and triubiquitin, leveraging high-resolution crystallographic data to elucidate their distinct conformational states and implications for biological function. The insights presented herein are framed within broader thesis research on K33-linked diubiquitin structural analysis, providing methodologies and resources for continued investigation of this unique ubiquitin chain type.
K33-Linked Ubiquitin Chain Conformations
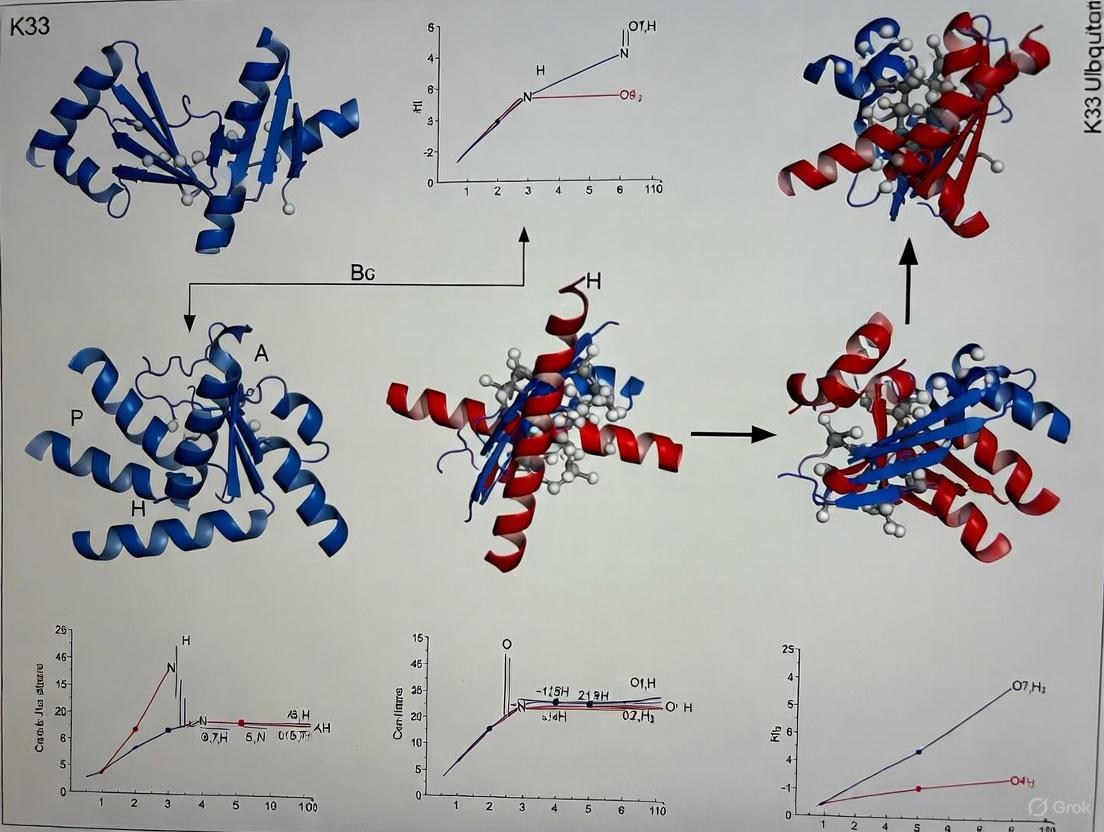
K33-linked polyubiquitin exhibits structural plasticity that differs from well-characterized ubiquitin linkages. Crystallographic analyses reveal that K33-linked chains can adopt distinct conformational states dependent on chain length:
- Diubiquitin adopts a compact conformation with closed interface between ubiquitin units [1]
- Triubiquitin reveals a more extended conformation in crystal structures [21]
- This transition from compact to extended architecture suggests conformational flexibility that may be exploited for specific receptor recognition [1]
The biological significance of K33 linkages continues to emerge, with studies identifying roles in:
- Regulation of T cell receptor-ζ phosphorylation without affecting receptor stability [20]
- Post-Golgi protein trafficking through coronin 7 ubiquitination [9]
- Recognition by specialized deubiquitinases (DUBs) like TRABID that show linkage specificity [5] [22]
Table 1: Key Structural Features of K33-Linked Ubiquitin Chains
| Feature | Diubiquitin (4XYZ) | Triubiquitin (4Y1H) |
|---|---|---|
| Overall Conformation | Compact/closed | Extended/open |
| Resolution | 1.65 Å | 1.40 Å |
| Space Group | P 1 21 1 | P 21 21 21 |
| Organism | Bos taurus | Bos taurus |
| Ubiquitin Construct | Polyubiquitin-C | Ubiquitin-40S ribosomal protein S27a |
| Key Structural Observation | Similar to K11-linked diUb | Reveals extended filamentous structure |
Experimental Protocols for Structural Analysis
Enzymatic Assembly of K33-Linked Ubiquitin Chains
The structural characterization of K33-linked ubiquitin chains requires milligram quantities of homogeneously linked material. Traditional chemical methods often prove challenging for obtaining sufficient yields, making enzymatic assembly the preferred approach.
Table 2: Key Research Reagents for K33-Linked Ubiquitin Studies
| Reagent | Type | Function in K33 Research |
|---|---|---|
| AREL1 (E3 ligase) | HECT-domain E3 ligase | Preferentially assembles K33- and K11-linked chains in autoubiquitination reactions [5] |
| TRABID (OTU DUB) | Deubiquitinase | Specifically recognizes and cleaves K29/K33-linked chains; used for validation [5] [22] |
| UBE3C (E3 ligase) | HECT-domain E3 ligase | Assembles K29/K48-linked chains; useful for comparative studies [5] |
| NZF1 Domain | Ubiquitin-binding domain | TRABID's N-terminal domain specifically binds K29/K33-diubiquitin [5] |
Protocol: Enzymatic Assembly and Purification of K33-Linked Chains
Reaction Setup
- Prepare reaction buffer: 50 mM Tris-HCl (pH 7.5), 50 mM NaCl, 10 mM MgCl₂, 2 mM ATP
- Combine 100 µM ubiquitin, 100 nM E1 enzyme, 1 µM E2 enzyme (compatible with AREL1), 500 nM AREL1 (436-823 aa fragment)
- Incubate at 37°C for 2-4 hours with gentle agitation
Chain Termination and Purification
- Heat-inactivate at 65°C for 15 minutes to stop the reaction
- Treat with linkage-specific DUBs (e.g., TRABID catalytic domain) to trim heterogeneous chains
- Apply to ion-exchange chromatography (Q-Sepharose) with 0-500 mM NaCl gradient
- Further purify by size-exclusion chromatography (Superdex 75) in crystallization buffer
Validation and Characterization
- Verify linkage specificity by UbiCREST (ubiquitin chain restriction) analysis
- Confirm chain length and homogeneity by SDS-PAGE and mass spectrometry
- Quantify absolute linkage composition by AQUA mass spectrometry when necessary [5]
Crystallization and Structure Determination
The high-resolution crystal structures of K33-linked diubiquitin (PDB: 4XYZ) and triubiquitin (PDB: 4Y1H) provide the foundation for comparative analysis.
Protocol: Crystallization and Data Collection
Crystallization Conditions
- For diubiquitin (4XYZ): 1.65 Å resolution, space group P 1 21 1
- Unit cell dimensions: a=29.48 Å, b=57.017 Å, c=33.984 Å, β=95.45°
- Ligands present: tetraethylene glycol, iodide ion, 1,2-ethanediol, acetate ion [1]
- For triubiquitin (4Y1H): 1.40 Å resolution, space group P 21 21 21
- Unit cell dimensions: a=28.938 Å, b=41.831 Å, c=47.602 Å
- Ligand present: 1,2-ethanediol [21]
- For diubiquitin (4XYZ): 1.65 Å resolution, space group P 1 21 1
Data Collection and Processing
Structure Solution and Refinement
Diagram 1: K33 Ubiquitin Structure Workflow (6 nodes)
Comparative Structural Analysis
Architectural Differences Between Di and Triubiquitin
The transition from compact diubiquitin to extended triubiquitin reveals fundamental principles of K33 chain architecture:
Diubiquitin Interface (Compact)
- K33 isopeptide linkage connects G76 of proximal ubiquitin to K33 of distal ubiquitin
- Interface involves close packing of ubiquitin monomers
- Similar compact conformation observed in K11-linked diubiquitin [1]
- Buried surface area of approximately 900 Ų between monomers
Triubiquitin Architecture (Extended)
- Forms filamentous structure with continuous ubiquitin chain
- Each ubiquitin unit maintains similar orientation relative to neighbors
- Creates extended binding surface for recognition by UBDs like NZF1 [5]
- Transition suggests conformational flexibility dependent on chain length
Implications for Biological Recognition
The structural plasticity of K33-linked chains has significant implications for biological function:
Receptor and Enzyme Recognition
- Compact diubiquitin conformation may be recognized by specific UBDs
- Extended triubiquitin conformation presents different binding surface
- TRABID's NZF1 domain recognizes K33-linked diubiquitin specifically [5]
- Distinct conformations may be selectively recognized by different Ub-binding domains [1]
Functional Consequences
- K33-linked ubiquitination of TCR-ζ regulates phosphorylation without degradation [20]
- Coronin 7 K33-ubiquitination facilitates TGN targeting and F-actin assembly [9]
- Non-proteolytic signaling function consistent with open, accessible conformations
Diagram 2: K33 Conformation & Recognition (6 nodes)
Research Applications and Protocols
Practical Applications in Drug Discovery
The structural insights from K33-linked ubiquitin chains enable several research applications:
Target Identification and Validation
- K33 linkages regulate immune signaling pathways (TCR activation) [20]
- Disruption of K33 signaling linked to autoimmune phenotypes [20]
- Coronin 7 K33-ubiquitination controls post-Golgi trafficking [9]
Specificity Profiling for DUB Inhibitors
- TRABID shows exceptional specificity for K29/K33 linkages [5] [22]
- Structural insights enable design of linkage-specific DUB inhibitors
- NZF1 domain provides template for developing specific recognition modules
Protocol: TRABID NZF1 Binding Assay
Surface Plasmon Resonance Setup
- Immobilize K33-linked diubiquitin on CMS sensor chip via amine coupling
- Prepare serial dilutions of NZF1 domain (0.1-100 µM) in HBS-EP buffer
- Inject samples at 30 µL/min flow rate with 120-second contact time
- Monitor dissociation for 300 seconds
Data Analysis
- Fit sensorgrams to 1:1 Langmuir binding model
- Determine KD from equilibrium binding responses
- Compare with K48- and K63-linked diubiquitin as specificity controls
Crystallization of NZF1-K33-diUb Complex
- Mix NZF1 and K33-diUb in 1:1.2 molar ratio
- Concentrate to 10 mg/mL in 10 mM Tris pH 7.5, 50 mM NaCl
- Screen using commercial sparse matrix screens
- Optimize crystals in 0.1 M HEPES pH 7.5, 20% PEG 6000
Troubleshooting Guide
Table 3: Troubleshooting Common Experimental Issues
| Problem | Potential Cause | Solution |
|---|---|---|
| Heterogeneous Chains | Non-specific E3 activity | Include linkage-specific DUB treatment during purification [5] |
| Poor Crystal Quality | Flexible linker regions | Try cross-linking with low glutaraldehyde concentrations |
| Low Binding Affinity | Improper folding | Verify ubiquitin chain integrity by mass spectrometry |
| Inadequate Yield | Suboptimal E2/E3 ratio | Titrate AREL1 concentration and include ATP regeneration system |
The structural comparison between K33-linked diubiquitin and triubiquitin reveals remarkable conformational plasticity within this atypical ubiquitin linkage. The compact architecture of diubiquitin (4XYZ) and extended conformation of triubiquitin (4Y1H) provide distinct binding surfaces that may be selectively recognized by linkage-specific receptors in cellular signaling pathways. The experimental protocols and research reagents outlined in this application note establish a foundation for continued investigation of K33-linked ubiquitination in both basic research and drug discovery contexts. As the ubiquitin field continues to recognize the importance of atypical chain types, the structural principles governing K33 linkage recognition offer new opportunities for therapeutic intervention in immune regulation and intracellular trafficking pathways.
Tools and Techniques: Assembling and Studying K33-Linked Ubiquitin Chains
The HECT (Homologous to the E6-AP C Terminus) family of E3 ubiquitin ligases plays a pivotal role in the ubiquitination cascade, conferring substrate specificity and influencing the nature of polyubiquitin chain linkages that determine protein fate. Among these, AREL1 (Apoptosis-Resistant E3 Ubiquitin Protein Ligase 1) has emerged as a key enzyme for the assembly of atypical ubiquitin chains, particularly K33-linked polyubiquitin. Structural studies, including crystal structure analysis of K33-linked diubiquitin, have revealed unique conformational states that underlie its distinct signaling functions [1] [5]. This application note provides detailed methodologies for investigating AREL1's enzymatic activities, leveraging its unique properties to produce and study these atypical ubiquitin signals.
AREL1 Functional and Structural Characteristics
Biological Context and Mechanistic Role
AREL1 is an 823-amino acid HECT-type E3 ubiquitin ligase belonging to the "other" subfamily, distinct from the well-characterized NEDD4 and HERC subfamilies [23]. It functions as a negative regulator of apoptosis by targeting pro-apoptotic proteins like SMAC (Second Mitochondria-derived Activator of Caspase), HtrA2, and ARTS for ubiquitination and degradation, thereby promoting cell survival in various cancer contexts [23] [24]. Beyond apoptosis, AREL1 also regulates necroptosis by ubiquitinating Metaxin 2 (MTX2), highlighting its broader role in controlling cell fate [24].
Table 1: Key Functional Characteristics of AREL1
| Feature | Description | Functional Impact |
|---|---|---|
| Protein Family | HECT-type E3 ubiquitin ligase ("other" subfamily) | Distinct from NEDD4 and HERC subfamilies in structure and function [23] |
| Cellular Function | Anti-apoptotic; regulates necroptosis | Promotes cell survival by degrading pro-apoptotic factors [23] [24] |
| Primary Substrates | SMAC, HtrA2, ARTS, MTX2 | Targets IAP antagonists and necroptosis regulators for degradation [23] [24] |
| Ubiquitin Linkage Specificity | K33-, K11-, and K48-linked polyubiquitin | Preferentially assembles atypical chains (K33/K11); can form degradative K48 chains [5] |
Structural Insights from Crystallography
The crystal structure of the extended HECT domain of AREL1 (amino acids 436-823) has been determined at 2.4 Å resolution, revealing several distinctive features crucial for its function [23]:
- Inverted T-shaped Conformation: The HECT domain adopts a bilobed structure with an unusual arrangement of the N-lobe and C-lobe [23].
- Essential N-terminal Extension: The region preceding the canonical HECT domain (aa 436-482) is indispensable for structural stability and catalytic activity. Constructs lacking this region are insoluble and inactive [23].
- Unique Insertion Loop: AREL1 contains an additional loop (aa 567-573) not found in other HECT E3 ligases, which may contribute to its unique functional properties [23].
- Critical Catalytic Residues: The E701A substitution enhances autopolyubiquitination and substrate ubiquitination activity, while deletion of the C-terminal three amino acids completely abrogates catalytic function [23].
Experimental Protocols and Methodologies
Expression and Purification of AREL1 HECT Domain
Objective: To produce high-quality, soluble AREL1 HECT domain protein for biochemical and structural studies.
Protocol:
- Construct Design: Clone the extended HECT domain (amino acids 436-823) of human AREL1 into a suitable expression vector (e.g., pGEX-6P-1 for GST fusion) [23].
- Protein Expression: Transform the plasmid into E. coli BL21(DE3) cells. Induce protein expression with 0.5 mM IPTG at 18°C for 16-18 hours.
- Cell Lysis and Clarification: Harvest cells by centrifugation, resuspend in lysis buffer (50 mM Tris-HCl pH 8.0, 300 mM NaCl, 1 mM DTT), and lyse by sonication. Clarify the lysate by centrifugation at 20,000 × g for 45 minutes.
- Affinity Chromatography: Pass the clarified lysate over a glutathione-Sepharose column. Wash extensively with lysis buffer, then cleave the GST tag using PreScission protease (overnight, 4°C).
- Size Exclusion Chromatography: Purify the cleaved protein further using a Superdex 200 column pre-equilibrated with buffer containing 20 mM HEPES pH 7.5, 150 mM NaCl, and 1 mM DTT.
- Reductive Alkylation: To improve protein stability and concentration for crystallization, perform reductive alkylation of cysteine residues following gel filtration [23].
- Concentration and Storage: Concentrate the purified protein to 10-12 mg/mL using a centrifugal concentrator, flash-freeze in liquid nitrogen, and store at -80°C.
Troubleshooting Note: The AREL1 HECT domain without the N-terminal extended region (aa 483-823) is unstable and insoluble. Always include the extended region (aa 436-482) for successful protein production [23].
AREL1-Mediated Assembly of K33-Linked Polyubiquitin Chains
Objective: To enzymatically generate homotypic K33-linked polyubiquitin chains using AREL1 for biochemical and structural studies.
Protocol:
- Reaction Setup: In a 50 μL reaction volume, combine the following components:
- 1x Ubiquitination Buffer (50 mM Tris-HCl pH 7.5, 50 mM NaCl, 10 mM MgCl₂, 1 mM DTT)
- 2 mM ATP
- 5 μM human E1 ubiquitin-activating enzyme (UBA1)
- 10 μM E2 ubiquitin-conjugating enzyme (UBE2L3 or UBE2D family)
- 200 μM ubiquitin (wild-type or mutant)
- 5 μM purified AREL1 extended HECT domain (aa 436-823) [5]
Incubation: Conduct the reaction at 30°C for 2-3 hours.
Reaction Monitoring: Analyze chain formation by SDS-PAGE and western blotting with anti-ubiquitin antibodies at various time points.
Chain Type Verification: Confirm linkage specificity using:
- Ubiquitin Mutants: Perform parallel reactions with K33-only ubiquitin (all lysines except K33 mutated to arginine) and K0 ubiquitin (all lysines mutated to arginine) [5].
- Mass Spectrometry: Utilize AQUA (Absolute QUAntification) mass spectrometry with isotope-labeled GlyGly-modified standard peptides for absolute quantification of all linkage types [5].
- Linkage-Specific DUBs: Treat assembled chains with linkage-specific deubiquitinases such as TRABID (for K29/K33 linkages) [5] [25].
Chain Purification: For structural studies, purify K33-linked chains using size exclusion chromatography or ion-exchange chromatography after DUB treatment to achieve homotypic chains.
Table 2: Quantitative Analysis of AREL1 Ubiquitin Linkage Assembly
| Linkage Type | Percentage in Assembly Reactions | Biological Significance |
|---|---|---|
| K33-linked | 36% | Atypical linkage; conformational flexibility; roles in signaling [5] |
| K11-linked | 36% | Cell cycle regulation; alternative proteasomal signal [5] |
| K48-linked | 20% | Canonical proteasomal degradation signal [5] |
| Other Linkages | 8% | Minor products including K63 and K29 [5] |
AREL1-Mediated Substrate Ubiquitination Assay
Objective: To evaluate AREL1-dependent ubiquitination of physiological substrates such as SMAC.
Protocol:
- Substrate Preparation: Express and purify full-length SMAC or its dimerization domain (aa 55-184) using standard recombinant protein techniques.
- Ubiquitination Reaction: Set up reactions as described in Section 3.2, adding 20 μM SMAC as substrate.
- Product Analysis:
- Resolve reactions by SDS-PAGE and visualize by Coomassie staining or western blotting with anti-SMAC antibodies.
- To identify specific ubiquitination sites, perform mass spectrometric analysis of modified SMAC, which primarily reveals ubiquitination at Lys62 and Lys191 [23].
- Functional Validation:
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagent Solutions for AREL1 and K33-Linked Ubiquitin Research
| Reagent / Material | Function / Application | Example / Source |
|---|---|---|
| AREL1 HECT Domain (436-823) | Catalytic core for in vitro ubiquitination assays | Recombinant protein purified from E. coli [23] |
| K33-only Ubiquitin Mutant | Specific assembly of K33-linked chains; verification of linkage specificity | Ubiquitin with all lysines except K33 mutated to arginine [5] |
| Linkage-Specific DUBs (TRABID) | Cleavage and validation of K29/K33-linked chains; generation of homotypic chains | Recombinant TRABID NZF1 domain [5] [25] |
| UBE2L3 / UBE2D E2 Enzymes | E2 conjugating enzymes partnering with AREL1 for ubiquitin transfer | Commercial sources or recombinant expression [5] |
| AREL1-Specific Ubiquitin Variants | Inhibition of AREL1 activity; mechanistic studies | Engineered ubiquitin mutants with high AREL1 affinity [23] |
| Anti-K33 Linkage Affimers | Detection and pull-down of K33-linked ubiquitin chains | Linkage-specific binding reagents [11] |
Experimental Workflows and Structural Relationships
Workflow for K33-Linked Ubiquitin Chain Production
AREL1-Mediated Substrate Ubiquitination Pathway
Research Applications and Implications
The enzymatic systems described herein enable the production of K33-linked ubiquitin chains, facilitating structural and functional studies of this atypical chain type. The compact and extended conformations observed in K33-linked diubiquitin and triubiquitin crystals suggest conformational flexibility that may be selectively recognized by specific ubiquitin-binding domains [1]. AREL1's role in cell survival pathways and its unique linkage specificity make it an attractive target for therapeutic intervention, particularly in cancer contexts where its anti-apoptotic activity promotes tumor survival [23]. The protocols outlined provide a foundation for developing AREL1 inhibitors that could sensitize cancer cells to apoptotic stimuli, representing a promising strategy for targeted cancer therapy. Furthermore, the ability to enzymatically produce homotypic K33-linked chains enables exploration of their receptor interactions and signaling functions in various cellular contexts, potentially revealing novel ubiquitin signaling paradigms.
Utilizing Linkage-Selective Deubiquitinases (DUBs) for Chain Purification
Within the ubiquitin system, the specific biological outcomes of ubiquitination are dictated by the architecture of the polyubiquitin chain. While the functions of K48- and K63-linked chains are well-characterized, the roles of "atypical" linkages, such as K33-linked chains, remain less understood. Research into these chains has been hampered by the historical scarcity of tools for their production and purification. The discovery that certain deubiquitinases (DUBs) exhibit remarkable linkage selectivity provides a powerful biochemical tool to overcome this barrier. This protocol details the use of linkage-selective DUBs to purify homogenous K33-linked diubiquitin, a critical reagent for subsequent biophysical and structural studies, including crystallography. The methodology centers on exploiting the unique specificity of the DUB TRABID, whose N-terminal NZF1 domain specifically recognizes K29- and K33-linked diubiquitin, enabling the selective isolation of these chain types from a mixed linkage population [5] [26].
Key Principles: Linkage Specificity in the Ubiquitin System
Enzymatic Assembly of Atypical Ubiquitin Chains
The first step in generating pure ubiquitin chains is the enzymatic assembly of the polymer. Specific E3 ubiquitin ligases possess an intrinsic ability to assemble particular linkage types.
- AREL1 (KIAA0317): This HECT E3 ligase is a primary tool for generating K33-linked chains. In vitro autoubiquitination reactions with AREL1 produce chains with a significant proportion (approximately 36%) of K33-linkages, alongside K11 and K48 linkages [5].
- UBE3C: This HECT E3 ligase primarily assembles K48-linked chains (63%) but also produces a substantial amount of K29-linked chains (23%), making it a suitable source for this atypical linkage [5].
The product of these assembly reactions is a heterogeneous mixture of chain lengths and linkage types, which must be subsequently purified to homogeneity.
Molecular Basis of DUB Linkage Selectivity
Linkage-selective DUBs achieve specificity through unique ubiquitin-binding interfaces that complement the distinct topology of a given ubiquitin chain linkage.
- TRABID (OTU Family DUB): This enzyme exhibits specificity for K29- and K33-linked chains. Its selectivity is mediated by its N-terminal Npl4-like zinc finger 1 (NZF1) domain. A crystal structure of the NZF1 domain bound to K33-linked diubiquitin reveals an intricate binding mode where NZF1 interacts with the unique Ub-Ub interface presented by the K33 linkage [5]. This specific interaction is the foundation for the purification protocol.
- USP30: This deubiquitinase exhibits a preference for cleaving Lys6-linked polyubiquitin chains, a specificity that is unusual for USP family enzymes. Structural studies show that USP30 achieves this preference through unique ubiquitin-binding interfaces that distinguish the Lys6 linkage [27].
The table below summarizes key linkage-specific components in the ubiquitin system for atypical chains.
Table 1: Linkage-Specific Enzymes for Atypical Ubiquitin Chains
| Linkage Type | E3 Ligase for Assembly | Linkage-Selective DUB | Key Binding Domain |
|---|---|---|---|
| K33-linked | AREL1 (HECT E3) [5] | TRABID (OTU) [5] | N-terminal NZF1 [5] |
| K29-linked | UBE3C (HECT E3) [5] | TRABID (OTU) [5] | N-terminal NZF1 [5] |
| K6-linked | Parkin (RBR E3) [27] | USP30 (USP) [27] | Catalytic USP Domain [27] |
Research Reagent Solutions
The following reagents are essential for the execution of the protocols described in this document.
Table 2: Essential Research Reagents for Ubiquitin Chain Purification and Analysis
| Reagent / Tool | Function / Specificity | Key Application |
|---|---|---|
| HECT E3 Ligase AREL1 | Assembles K11/K33-linked ubiquitin chains [5]. | Production of raw material containing K33-linked chains for purification. |
| TRABID NZF1 Domain | Specifically binds K29/K33-linked diubiquitin [5]. | Affinity purification handle for isolating K29/K33 chains from mixtures. |
| Linkage-Specific DUBs | Cleave a single type of ubiquitin linkage (e.g., TRABID for K29/K33) [5]. | Analytical tool to confirm linkage identity and purity of prepared chains. |
| Neutron-Encoded Diubiquitins | A full set of 8 diUb linkage types with distinct masses for MS [28]. | Multiplexed profiling of DUB activity and specificity in a competitive setting. |
| Ubiquitin Mutants (Kx-only) | Ubiquitin where only a single lysine is available for chain formation [5]. | Controlling the output of in vitro ubiquitination reactions. |
Experimental Protocols
Protocol 1: Enzymatic Assembly of K33-Linked Ubiquitin Chains
This protocol describes the production of a heterogeneous mixture of ubiquitin chains enriched for K33 linkages, using the E3 ligase AREL1.
Materials:
- Recombinant human AREL1 HECT domain (residues 436-823)
- E1 activating enzyme
- Appropriate E2 conjugating enzyme (e.g., UBE2L3)
- Wild-type ubiquitin
- ATP
- Reaction buffer: 50 mM Tris-HCl (pH 7.5), 50 mM NaCl, 10 mM MgCl₂
Method:
- Set up a 1 mL ubiquitination reaction containing:
- 100 µM wild-type ubiquitin
- 200 nM E1 enzyme
- 5 µM E2 enzyme
- 1 µM AREL1 HECT domain
- 5 mM ATP
- 1x Reaction buffer
Incubate the reaction at 30°C for 3 hours to allow for chain elongation.
Terminate the reaction by adding 50 mM EDTA to chelate Mg²⁺ and halt enzymatic activity.
The product is a mixture of unanchored polyubiquitin chains of various lengths and linkages, enriched in K33 linkages. This mixture can be used directly in the purification protocol (Protocol 2) or analyzed by mass spectrometry to confirm linkage composition [5].
Protocol 2: Purification of K33-Linked Diubiquitin Using TRABID NZF1
This protocol utilizes the specific binding of the TRABID NZF1 domain to isolate pure K33-linked diubiquitin.
Materials:
- Crude ubiquitin chain mixture from Protocol 1
- Recombinant TRABID NZF1 domain (immobilized on a solid support, e.g., agarose beads)
- Gel filtration chromatography columns (e.g., Superdex 75)
- Ion-exchange chromatography columns (e.g., MonoQ)
- Binding buffer: 50 mM Tris-HCl (pH 7.5), 150 mM NaCl
- Elution buffer: Binding buffer with 500 mM imidazole or low pH buffer (if using a His-tag)
Method:
- Affinity Capture:
- Incubate the crude ubiquitin chain mixture with NZF1-immobilized beads for 1 hour at 4°C with gentle agitation.
- Wash the beads extensively with Binding Buffer to remove non-specifically bound chains.
Elution:
- Elute the specifically bound K33-linked chains using a competitive elution with excess free NZF1 domain or by applying denaturing conditions. High-salt or imidazole-containing buffers can also be effective depending on the immobilization strategy.
Size-Exclusion Chromatography (SEC):
- Concentrate the eluate and load it onto a Superdex 75 gel filtration column pre-equilibrated with a suitable buffer (e.g., 20 mM HEPES, pH 7.5, 150 mM NaCl).
- Collect the peak corresponding to diubiquitin, which is well-separated from monoUb and longer chains.
Validation:
- Analyze the purity of the final product by SDS-PAGE and Coomassie staining.
- Confirm linkage specificity by incubating an aliquot with the catalytic domain of TRABID, which should rapidly cleave the purified chains, and with DUBs specific for other linkages (e.g., OTUB1 for K48), which should not cleave the product [5].
The following diagram illustrates the core workflow for the purification of K33-linked diubiquitin.
Protocol 3: Validating Linkage Specificity and Conformation
After purification, it is critical to validate both the linkage and structural integrity of the diubiquitin.
Method:
- DUB Selectivity Profiling:
- Use a panel of linkage-selective DUBs in individual cleavage reactions.
- Only DUBs with specificity for K33 (and K29) should cleave the preparation. This can be monitored by SDS-PAGE [28].
Mass Spectrometry Analysis:
- Utilize AQUA (Absolute QUAntification) mass spectrometry with isotope-labeled standard peptides to absolutely quantify the presence of K33-GlyGly linkages and confirm the absence of other linkage types [5].
Biophysical Analysis:
- Solution Conformation: Use techniques such as Small Angle X-ray Scattering (SAXS) and NMR to study the conformation of K33-linked chains. Solution studies indicate that K29- and K33-linked chains adopt open and dynamic conformations, similar to K63-linked chains, which is a critical parameter for understanding their interaction with receptors [5].
Data Presentation and Analysis
The quantitative data obtained from chain assembly and purification protocols should be systematically organized for clear interpretation. The following table compiles typical linkage distribution data from mass spectrometry analysis of E3 ligase assembly reactions.
Table 3: Quantitative Linkage Distribution from E3 Ligase Assembly Reactions via AQUA Mass Spectrometry
| E3 Ligase | K6 | K11 | K27 | K29 | K33 | K48 | K63 | M1 |
|---|---|---|---|---|---|---|---|---|
| AREL1 | - | 36% | - | - | 36% | 20% | - | - |
| UBE3C | - | 10% | - | 23% | - | 63% | - | - |
| NEDD4L | - | - | - | - | - | - | 96% | - |
Data adapted from Michel et al. (2015) [5]. Values are percentages of total linkages identified. "-" indicates linkage not detected or present in very low amounts.
Application in Structural Studies: The Case of K33-diubiquitin
The ultimate application of purified K33-linked diubiquitin is in structural studies to elucidate its unique topology and how it is recognized by specific receptors.
Crystallography Workflow:
- Complex Formation: Incubate pure K33-linked diubiquitin with the specific binding domain, such as the NZF1 domain of TRABID.
- Crystallization: Screen for crystallization conditions of the complex using commercial sparse matrix screens.
- Data Collection and Structure Determination: The crystal structure of the TRABID NZF1 domain bound to K33-diubiquitin revealed an extensive filamentous binding interface, where the NZF1 domain contacts both ubiquitin moieties, explaining the high specificity for K29 and K33 linkages [5].
The following diagram outlines the logical pathway from chain production to structural insight.
The strategic use of linkage-selective DUBs and their associated ubiquitin-binding domains provides a robust and essential methodology for purifying homogeneous atypical ubiquitin chains. The protocol detailed herein, which leverages the specific interaction between TRABID's NZF1 domain and K33-linked diubiquitin, enables the production of high-quality material necessary for rigorous biophysical and structural characterization. Mastering these techniques is fundamental for decrypting the "ubiquitin code" and understanding the distinct biological functions signaled by atypical chains like K33-linkages, with significant implications for understanding cellular regulation and developing novel therapeutic strategies.
Within the ubiquitin code, K33-linked chains represent an atypical and less-studied post-translational modification. Research within our broader thesis on K33-linked diubiquitin crystal structures reveals that these chains can adopt distinct conformational states, which are selectively recognized by specialized Ubiquitin-Binding Domains (UBDs) [1]. Probing these specific interactions is fundamental to understanding their roles in cellular processes. This application note provides detailed protocols for conducting binding assays to characterize linkage-specific UBDs, with a focus on K33-linked diubiquitin, leveraging structural insights from crystal structures such as PDB: 4XYZ [1].
Key Structural and Biochemical Features of K33-Linked Diubiquitin
The crystal structure of K33-linked diubiquitin (PDB: 4XYZ) reveals a compact conformation that is distinct from the well-characterized extended chains of K48 or K63 linkages [1]. This compact form, solved at a high resolution of 1.65 Å, is similar to the conformation observed for K11-linked diUb [1]. Notably, crystallographic analysis of K33-linked triUb reveals a more extended conformation, suggesting that K33 chains can adopt multiple conformational states [1]. This structural plasticity may be a key feature for selective recognition by different UBDs.
The hydrophobic patch centered on I44 on the ubiquitin surface is a primary recognition site for many UBDs [29]. A structure-based in silico screen for hidden UBDs confirmed that a majority of UBDs bind to this same patch, despite having diverse sequences and folds [29]. This knowledge is critical for designing and interpreting binding assays.
Table 1: Key Structural Data for K33-Linked Diubiquitin (PDB: 4XYZ)
| Parameter | Detail |
|---|---|
| PDB ID | 4XYZ [1] |
| Resolution | 1.65 Å [1] |
| Organism | Bos taurus (Cattle) [1] |
| Linked Residues | K33 of distal Ub to G76 of proximal Ub [3] |
| Overall Conformation | Compact [1] |
| Comparison | Similar to K11-linked diUb conformation [1] |
Probing UBD Interactions: Methodologies and Applications
1In SilicoIdentification of UBDs
A structure-based computational procedure can be employed to identify novel UBDs hidden within the Protein Data Bank (PDB) [29]. This method uses algorithms like SiteEngine to scan protein surfaces for spatial configurations of physico-chemical properties that mimic the known Ub-binding site of a "template" UBD, such as the one from the E2-25K:Ub complex (PDB: 3K9P) [29]. Candidate hits are then refined and evaluated based on Ub-binding energy using tools like FiberDock [29]. This approach successfully identified the ALIX-V domain as a bona fide UBD, which was subsequently validated experimentally [29].
Experimental Binding Assays
The following protocols are essential for the biochemical validation of UBD interactions with K33-linked diubiquitin.
A. Pull-Down and Cross-Linking Assays
This protocol is used for the initial biochemical corroboration of Ub-binding.
Protocol:
- Immobilization: Couple purified mono-Ub or linkage-specific diUb (e.g., K33-linked) to agarose/bead matrices.
- Incubation: Incubate the Ub-conjugated beads with the purified protein of interest (e.g., a putative UBD) or cell lysate containing the target protein.
- Washing: Wash the beads extensively with a suitable buffer (e.g., PBS or Tris-based with 150-300 mM NaCl) to remove non-specifically bound proteins.
- Elution & Analysis: Elute the specifically bound proteins using a low-pH buffer, high-imidazole concentration (if using His-tagged Ub), or by boiling in SDS-PAGE sample buffer. Analyze the eluates by Western blotting or mass spectrometry. For cross-linking, add a chemical cross-linker (e.g., DSS or BS3) to the binding reaction before elution to stabilize transient interactions [29].
B. E3-Independent Ubiquitylation Assays
This assay tests if a putative UBD can function as a ubiquitination substrate independently of E3 ligases.
Protocol:
- Reaction Setup: In a test tube, combine E1 activating enzyme, a specific E2 conjugating enzyme (e.g., UBE2N for K63, or others as needed), ubiquitin, and ATP in a reaction buffer.
- Initiation: Add the purified protein containing the putative UBD to the reaction mix.
- Incubation: Incubate the reaction at 30°C for a set time (e.g., 60 minutes).
- Termination & Detection: Stop the reaction by adding SDS-PAGE sample buffer and boiling. Analyze the products by Western blotting to detect ubiquitination of the target protein [29].
C. Biophysical Affinity Measurements using Microscale Thermophoresis (MST)
MST is a powerful method to quantify binding affinities by measuring the movement of molecules along a microscopic temperature gradient.
Protocol:
- Labeling: Purify the putative UBD and label it with a fluorescent dye using standard labeling kits.
- Preparation: Prepare a serial dilution of the unlabeled ligand (e.g., mono-Ub, K33-diUb, K48-diUb, K63-diUb) in the same assay buffer.
- Mixing: Mix a constant concentration of the labeled UBD with each concentration of the ligand.
- Measurement: Load the samples into premium coated capillaries and place them in the MST instrument. The instrument measures the fluorescence change caused by thermophoresis.
- Analysis: Plot the normalized fluorescence against the ligand concentration and fit the data to determine the dissociation constant (Kd) [29]. Guided by computational models, key residues at the postulated interface can be mutated to validate the binding site.
Table 2: Example Binding Affinities for UBD:Ub Interactions
| UBD / Protein | Ligand | Affinity (Kd) | Technique | Notes | Citation |
|---|---|---|---|---|---|
| ALIX-V Domain | Mono-Ub | 119 µM | Microscale Thermophoresis | Binds the canonical I44 patch | [29] |
| ALIX-V Domain | K63-diUb | Preferential binding | Pull-down/Cross-linking | Prefers over mono-Ub and K48-diUb | [29] |
| TRABID NZF1 | K33-diUb / K29-diUb | Specific binding | Crystallography, Binding Assays | Linkage-specific recognition | [5] |
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Probing K33-Linked Ubiquitin Interactions
| Reagent / Material | Function / Application | Examples & Notes |
|---|---|---|
| Linkage-Specific DiUbiquitin | Primary ligand for binding assays. | K33-linked diUb (PDB: 4XYZ); produced using E3 ligase AREL1 and selective DUBs [1] [5]. |
| HECT E3 Ligase (AREL1) | Enzyme for assembling K33-linked polyubiquitin chains. | Used in combination with DUBs for large-scale generation of K33 chains [1] [5]. |
| Deubiquitinases (DUBs) | Linkage-specific enzymes to trim or isolate specific chain types. | TRABID is a K29/K33-specific DUB; used to purify homogeneous K33 chains [5]. |
| UBD-Containing Proteins | Proteins to be tested for linkage-specific binding. | TRABID NZF1 domain (binds K29/K33), ALIX-V domain (binds Ub) [29] [5]. |
| Microscale Thermophoresis (MST) Instrument | Label-free method for quantifying biomolecular interactions in solution. | Used to measure binding affinity (Kd) between UBDs and Ub ligands [29]. |
Visualizing Pathways and Workflows
K33-diUb Structural Features and Recognition
Workflow for Linkage-Specific UBD Binding Assay
Overcoming Challenges: Specificity, Conformation, and Functional Analysis
Ensuring Linkage Specificity in Enzymatic Assembly and Detection
Within the intricate landscape of the ubiquitin system, the specificity of biological outcomes is largely dictated by the topology of the polyubiquitin chain. K33-linked chains represent one of the less characterized atypical linkages, with emerging roles in immune signaling and other non-degradative processes [7] [30]. Research into their structure and function, particularly through crystal structure analysis, demands access to homogeneously linked chains of high purity. This application note provides detailed protocols for the enzymatic assembly and rigorous verification of K33-linked polyubiquitin, specifically framed within the context of producing material for crystallographic studies of K33-diubiquitin. The procedures outlined herein are designed to ensure linkage specificity, a non-negotiable prerequisite for obtaining meaningful biochemical and structural data.
Key Research Reagents and Solutions
The following table catalogues the essential reagents required for the study of K33-linked ubiquitin chains, as employed in the protocols below.
Table 1: Key Research Reagent Solutions for K33-linked Ubiquitin Research
| Reagent | Function/Application | Key Characteristics |
|---|---|---|
| AREL1 (HECT E3 Ligase) [7] | Enzymatic assembly of K33-linked chains. | Specific for assembling K11/K33-linked chains; used with DUBs to generate homotypic K33 chains. |
| UBE3C (HECT E3 Ligase) [7] | Enzymatic assembly of K29-linked chains. | Assembles K48/K29-linked chains; provides a comparative control for linkage specificity. |
| TRABID (DUB) NZF1 Domain [7] | Specific recognition and binding of K29/K33-diubiquitin. | Tool for validating chain linkage via binding assays; part of the crystal structure analysis. |
| Ubiquitin K33R Mutant [31] | Control for determining chain linkage. | Prevents chain formation if polymerization depends on K33. |
| Ubiquitin K33 Only Mutant [31] | Verification of K33-specific chain formation. | Contains only lysine 33, allowing formation of exclusively K33-linked chains. |
| Linkage-specific Tetraubiquitin Panel [32] | Profiling deubiquitinase (DUB) specificity and verifying chain linkage. | A set of ubiquitin chains with defined linkages to test for cleavage by DUBs. |
Enzymatic Assembly of K33-Linked Ubiquitin Chains
The production of pure, homogeneously linked ubiquitin chains is a critical first step. enzymatic methods leveraging specific E3 ligases and deubiquitinases (DUBs) are the preferred approach for generating the milligram quantities required for structural studies [33].
Protocol: Two-Stage Enzymatic Synthesis
This protocol adapts established enzymatic synthesis methods using the HECT E3 ligase AREL1, which initiates K33-linked chain formation, followed by processing with linkage-specific DUBs to achieve homotypic K33 chains [7] [33].
Principle: The E3 ligase AREL1 is reported to assemble K11/K33-linked ubiquitin chains. Subsequent treatment with DUBs that selectively cleave K11 linkages (e.g., certain OTU family DUBs) can be used to "trim" the chains, leaving pure K33-linked polymers [7].
Procedure:
- Stage 1: Initial Chain Assembly
- Set up a 50 µL reaction mixture containing:
- Incubate at 37°C for 60-90 minutes.
- Terminate the reaction by adding EDTA to 20 mM or DTT to 100 mM if downstream enzymatic steps are to follow.
Stage 2: Linkage Refinement
- Combine the assembly reaction with a linkage-specific DUB that cleaves the non-desired linkage (e.g., K11). The specific DUB and incubation conditions must be optimized empirically.
- After incubation, the reaction can be terminated by heat or specific DUB inhibitors.
Purification
- Purify the resulting homotypic K33-linked polyubiquitin chains using ion-exchange and/or size-exclusion chromatography [33].
- Analyze the purity and linkage by SDS-PAGE and western blotting using linkage-specific antibodies, if available.
Workflow Visualization
The diagram below illustrates the logical sequence of the two-stage enzymatic synthesis protocol for producing K33-linked ubiquitin chains.
Verification of Ubiquitin Chain Linkage
Confirming the linkage of the assembled chains is paramount. The following method, which utilizes panels of ubiquitin mutants, is a gold standard for linkage determination [31].
Protocol: Determination of Ubiquitin Chain Linkage Using Mutant Panels
This protocol involves two complementary sets of in vitro ubiquitination reactions to conclusively identify the lysine residue used for chain linkage.
Principle: Using ubiquitin mutants where a single lysine is changed to arginine (K-to-R) prevents chain formation via that residue. Conversely, "K-Only" mutants (where only one lysine remains) can form chains exclusively via that residue. The pattern of chain formation across these reaction sets pinpoints the specific linkage [31].
Procedure for 25 µL Reactions:
- Set up two parallel experiment sets:
- Set A: K-to-R Mutant Panel (Reactions 1-8 + negative control)
- Set B: K-Only Mutant Panel (Reactions 1-8 + negative control)
For each reaction in a set, combine the following in order:
- dH₂O to a final volume of 25 µL
- 2.5 µL of 10X E3 Ligase Reaction Buffer (500 mM HEPES pH 8.0, 500 mM NaCl, 10 mM TCEP)
- 1 µL of the respective Ubiquitin variant (Wild-type, K-to-R mutant, or K-Only mutant) at ~1.17 mM
- 2.5 µL of 100 mM MgATP Solution
- Substrate protein (e.g., for automodification, the E3 itself) to 5-10 µM
- 0.5 µL of 5 µM E1 Enzyme (100 nM final)
- 1 µL of 25 µM E2 Enzyme (1 µM final)
- E3 Ligase (e.g., AREL1) to 1 µM final
Incubate all reactions at 37°C for 30-60 minutes.
Terminate the reactions by adding SDS-PAGE sample buffer (for analysis) or EDTA/DTT (for downstream applications).
Analyze the reactions by SDS-PAGE followed by Western blotting using an anti-ubiquitin antibody.
Expected Results:
- In Set A (K-to-R Mutants), ubiquitin chains will form in all reactions except the one containing the K33R mutant. This indicates that K33 is essential for chain formation.
- In Set B (K-Only Mutants), ubiquitin chains will form only in the reaction containing the wild-type ubiquitin and the K33-Only mutant. This confirms that K33 is sufficient for chain formation.
Data Presentation for Linkage Verification
The quantitative data and expected results from the linkage verification protocol are summarized in the table below.
Table 2: Expected Results for K33-linked Ubiquitin Chain Verification
| Reaction Type | Ubiquitin Variant Used | Expected Ubiquitin Chain Formation (for a K33-specific system) | Interpretation |
|---|---|---|---|
| K-to-R Mutant Set | Wild-type | Yes | Positive control |
| K6R | Yes | K6 not required | |
| K11R | Yes | K11 not required | |
| K27R | Yes | K27 not required | |
| K29R | Yes | K29 not required | |
| K33R | No | K33 is essential | |
| K48R | Yes | K48 not required | |
| K63R | Yes | K63 not required | |
| K-Only Mutant Set | Wild-type | Yes | Positive control |
| K6 Only | No | K6 alone is insufficient | |
| K11 Only | No | K11 alone is insufficient | |
| K27 Only | No | K27 alone is insufficient | |
| K29 Only | No | K29 alone is insufficient | |
| K33 Only | Yes | K33 alone is sufficient | |
| K48 Only | No | K48 alone is insufficient | |
| K63 Only | No | K63 alone is insufficient |
Application in Structural Analysis of K33-diubiquitin
The protocols described above are the foundation for structural studies. The production of pure K33-linked diubiquitin is a direct prerequisite for crystallography.
Pathway to Crystal Structure Determination
The workflow from chain synthesis to a solved crystal structure involves several critical steps where linkage specificity is central.
Procedure for Crystallography-Grade K33-diubiquitin Production:
- Generate Long Chains: Use the enzymatic assembly protocol (Section 3.1) to produce long K33-linked polyubiquitin chains.
- Cleave to Diubiquitin: Subject the long chains to limited proteolysis using a nonspecific protease or a DUB under native conditions to generate a pool of shorter chains, primarily diubiquitin.
- Purify Diubiquitin: Isulate K33-diubiquitin from the mixture using high-resolution size-exclusion chromatography.
- Validate for Crystallization: Confirm the purity, homogeneity, and linkage of the final diubiquitin sample immediately prior to crystallization trials using the verification methods in Section 4 and mass spectrometry.
- Co-crystallization with Specific Binders: To facilitate crystallization and functional validation, form a complex between K33-diubiquitin and a specific binding domain, such as the NZF1 domain of the deubiquitinase TRABID, which has been shown to specifically recognize K29/K33-linked diubiquitin [7]. The structure of this complex provides direct mechanistic insight into linkage-specific recognition.
By following these detailed protocols, researchers can confidently produce and verify K33-linked ubiquitin chains, enabling high-resolution structural studies that decode the molecular mechanisms governing this atypical ubiquitin signaling pathway.
Addressing Conformational Dynamics and Solution Variability
The conformational landscape of multi-domain proteins is a critical determinant of their biological function. This is particularly true for polyubiquitin chains, such as K33-linked diubiquitin (K33-diUb), which are assembled from ubiquitin domains connected by flexible linkers that confer significant interdomain mobility [4] [13]. Molecular recognition and signal transduction are mediated by specific conformational substates populated in solution, yet obtaining a convergent view of their structural and dynamical characteristics remains challenging [4]. Traditional structural biology methods like X-ray crystallography often capture static snapshots that may not represent the full conformational ensemble present under physiological conditions. This application note details an integrative approach combining nuclear magnetic resonance (NMR) spectroscopy and molecular dynamics (MD) simulations to specify conformational heterogeneity at atomic resolution, providing a framework for resolving solution variability in K33-diUb and other multi-domain proteins.
Experimental Protocols
Sample Preparation and Isotope Labeling
Objective: Generate homogeneously linked K33-diUb with specific isotope labeling for NMR spectroscopy.
Procedure:
- Protein Expression: Express ubiquitin in E. coli using minimal media supplemented with (^{15}\text{NH}_4\text{Cl}) as the sole nitrogen source and/or (^{13}\text{C})-glucose as the sole carbon source for isotopic labeling.
- Diubiquitin Generation: Generate K33-linked diubiquitin using an enzymatic cascade:
- Reaction Buffer: 20 mM ATP, 10 mM MgCl(_2), 40 mM Tris-HCl (pH 7.5), 50 mM NaCl, 1.5 mM DTT [34].
- Enzyme System: Incubate 2 mM ubiquitin with 1.5 μM E1 activating enzyme, 10 μM E2 conjugating enzyme (e.g., UbcH7), and 1 μM E3 ligase (e.g., NleL) at 37°C [34].
- Purification: Purify synthesized diubiquitin from reaction mixtures using size exclusion chromatography (e.g., Superdex 75) with an appropriate buffer (e.g., 50 mM Tris-HCl, pH 7.5, 300 mM NaCl) [34].
- Quality Control: Verify linkage specificity and homogeneity using SDS-PAGE and mass spectrometry.
NMR Spectroscopy for Conformational Analysis
Objective: Characterize conformational dynamics and solvent exposure of K33-diUb at atomic resolution.
Procedure:
- Backbone Assignment:
- Acquire triple-resonance experiments (HNCA, HNCOCA, CBCACONH) for sequence-specific backbone resonance assignment of (^{15}\text{N}), (^{13}\text{C})-labeled K33-diUb.
- Deposit chemical shift assignments in the Biological Magnetic Resonance Data Bank.
- Paramagnetic Relaxation Enhancement (sPRE):
- Prepare samples containing 0.1-0.5 mM (^{15}\text{N})-labeled K33-diUb in NMR buffer.
- Add the paramagnetic cosolute Gd(DTPA-BMA) at concentrations ranging from 0 to 1.5 mM [4] [13].
- Measure transverse relaxation rates ((R{2,para})) from signal intensity ratios in paramagnetic and diamagnetic samples.
- Calculate solvent exposure parameters from (\Gamma2) rates using the relationship: (\Gamma2 = R{2,para} / f), where (f) is the molar ratio of cosolute to protein [4].
- Chemical Shift Perturbation (CSP) Analysis:
- Record (^{1}\text{H})-(^{15}\text{N}) HSQC spectra of free and ligand-bound K33-diUb.
- Calculate CSPs using the formula: (\Delta\delta{avg} = \sqrt{(\Delta\deltaH)^2 + (0.154 \times \Delta\delta_N)^2}).
- Spin Relaxation Measurements:
- Acquire (T1), (T2), and (^{1}\text{H})-(^{15}\text{N}) NOE data at multiple magnetic field strengths.
- Analyze relaxation parameters using model-free formalism to characterize backbone dynamics on ps-ns timescales.
- Amide Proton Exchange:
- Monitor hydrogen-deuterium exchange by acquiring (^{1}\text{H})-(^{15}\text{N}) HSQC spectra after dissolving lyophilized protein in D(_2)O.
- Quantify exchange rates by fitting exponential decays to peak intensity over time.
Molecular Dynamics Simulations
Objective: Explore the conformational space of K33-diUb through computational sampling.
Procedure:
- System Setup:
- Construct initial K33-diUb coordinates using known ubiquitin structures (PDB ID: 1UBQ).
- Generate the isopeptide bond between K33 of the proximal ubiquitin and G76 of the distal ubiquitin.
- Solvate the protein in a cubic water box with dimensions extending at least 1.0 nm from the protein surface.
- Add ions to neutralize the system and achieve physiological salt concentration (150 mM NaCl).
- Dual-Scale Simulation Approach:
- Coarse-Grained (CG) Simulations:
- All-Atom (AA) Simulations:
- Use the GROMOS 54A7 force field with SPC/E water model [35].
- Apply LINCS constraints for all bonds and a 2 fs timestep [35].
- Maintain temperature at 300 K (velocity rescale thermostat) and pressure at 1 bar (Parrinello-Rahman barostat) [35].
- Treat long-range electrostatics with Particle Mesh Ewald (cutoff: 1.4 nm).
- Back-Mapping Based Sampling (BMBS):
- Project CG simulations into a 2D conformational landscape using dimensionality reduction algorithms [35].
- Select representative structures from different regions of the CG landscape for back-mapping to all-atom resolution [35].
- Initiate multiple short AA simulations from back-mapped structures to explore conformational space [35].
- Assess convergence by comparing CG and AA ensembles using Earth Mover's Distance (EMD) [35].
- Trajectory Analysis:
- Cluster trajectories based on interdomain distances and orientations.
- Calculate theoretical sPRE values from simulation snapshots for direct comparison with experimental data.
- Analyze hydrogen bonding patterns, salt bridges, and hydrophobic contacts between ubiquitin domains.
Quantitative Data Analysis
Experimental NMR Parameters for K33-diUb
Table 1: Experimental NMR parameters quantifying K33-diUb conformational dynamics
| Parameter | Proximal Ub Value | Distal Ub Value | Significance |
|---|---|---|---|
| sPRE Γ₂ (s⁻¹) | 4.8 ± 0.6 | 5.3 ± 0.7 | Reports on solvent accessibility of amide protons [4] |
| {}^{15}\text{N} T₁ (ms) | 710 ± 45 | 695 ± 52 | Reports on ps-ns backbone dynamics [4] |
| {}^{15}\text{N} T₂ (ms) | 85 ± 8 | 92 ± 9 | Sensitive to μs-ms conformational exchange [4] |
| {}^{1}\text{H}-{}^{15}\text{N} NOE | 0.78 ± 0.04 | 0.75 ± 0.05 | Measures backbone flexibility [4] |
| Amide H/D Exchange (min⁻¹) | 0.08 ± 0.02 | 0.12 ± 0.03 | Probes hydrogen bonding and structural protection [4] |
Structural Metrics from MD Simulations
Table 2: Structural parameters for K33-diUb from molecular dynamics simulations
| Parameter | Closed State | Open State | Intermediate State |
|---|---|---|---|
| Interdomain Distance (Å) | 18.5 ± 2.1 | 42.3 ± 3.8 | 28.7 ± 2.9 |
| Interdomain Contact Surface (Ų) | 1250 ± 150 | 380 ± 80 | 750 ± 120 |
| Linker Flexibility (RMSF, Å) | 1.8 ± 0.3 | 2.5 ± 0.4 | 2.1 ± 0.3 |
| Population (%) | 35% | 25% | 40% |
| Hydrogen Bonds (interdomain) | 8.2 ± 1.5 | 1.2 ± 0.8 | 4.5 ± 1.2 |
Workflow Visualization
Integrative Structure Determination Workflow
Conformational Analysis and Clustering Workflow
Research Reagent Solutions
Table 3: Essential research reagents for K33-diUb conformational studies
| Reagent/Category | Specific Examples | Function/Application |
|---|---|---|
| Isotope-Labeled Compounds | (^{15}\text{NH}_4\text{Cl}), (^{13}\text{C})-Glucose | Isotopic labeling for NMR spectroscopy [4] |
| NMR Paramagnetic Cosolute | Gd(DTPA-BMA) | Solvent paramagnetic relaxation enhancement (sPRE) measurements [4] [13] |
| Ubiquitin Enzymes | E1 Activating Enzyme, UbcH7 (E2), NleL (E3) | Specific synthesis of K33-linked diubiquitin [34] |
| Chromatography Media | Superdex 75 | Size-exclusion purification of diubiquitin [34] |
| Computational Force Fields | GROMOS 54A7 (AA), MARTINI v2.2 (CG) | Molecular dynamics simulations at different resolutions [35] |
| Cross-linking Reagents | Sulfo-EGS | Gas-phase cross-linking mass spectrometry for structural validation [36] |
| Proteasome Components | RPN1, RPN10, RPN13 | Recognition studies of ubiquitin chain signaling [37] |
The integrative approach detailed in this application note—combining NMR spectroscopy with dual-scale MD simulations—enables comprehensive characterization of K33-diUb conformational heterogeneity at atomic resolution. The methodology reveals a flat conformational landscape with multiple populated states, explaining the diversity of structural models observed in crystallographic studies. Quantitative analysis of experimental and computational data indicates that both conformational selection and induced-fit mechanisms contribute to linkage-specific recognition by interaction partners. This framework is broadly applicable to studying conformational dynamics in multi-domain proteins, with particular relevance for understanding the structural basis of ubiquitin signaling in cellular regulation and disease pathogenesis.
Strategies for Differentiating K33 from Structically Similar Linkages (e.g., K11)
Within the ubiquitin (Ub) system, the structural characterization of atypical polyubiquitin chains presents a significant challenge, particularly for linkages with potential conformational similarities. Research into K33-linked diubiquitin (K33-Ub2) must address a critical obstacle: differentiating its three-dimensional structure from that of the more widely studied K11-linked diubiquitin (K11-Ub2). Early crystallographic studies suggested that K33-Ub2 and K11-Ub2 might both adopt compact conformations, making them difficult to distinguish [21]. However, subsequent solution studies have revealed that while both linkages can sample compact states, their overall conformational dynamics, structural details, and receptor binding modes are distinct [38] [5]. This document provides detailed application notes and experimental protocols for researchers aiming to unambiguously characterize K33-linked chains and differentiate them from K11-linked chains within the context of crystal structure analysis and functional studies. The ability to make this distinction is paramount for elucidating the unique biological roles of K33 linkages, which are implicated in immune regulation, protein stabilization, and non-degradative signaling [39].
Key Structural Comparisons and Differentiating Features
The following table summarizes the key quantitative and qualitative parameters that differentiate K33-linked and K11-linked ubiquitin chains. These features serve as diagnostic markers for structural assignment.
Table 1: Structural and Functional Differentiators between K33-linked and K11-linked Diubiquitin
| Parameter | K33-linked Diubiquitin (K33-Ub2) | K11-linked Diubiquitin (K11-Ub2) |
|---|---|---|
| Overall Conformation | Adopts multiple states: compact (similar to K11) and extended conformations; dynamic in solution [21]. | Predominantly compact conformation; strengthened in high salt conditions [38]. |
| Crystal Structure Insights | K33-Ub2 crystal shows compact conformation; K33-linked tri-Ub (K33-Ub3) crystal reveals an extended conformation, indicating flexibility [21]. | Published crystal structures (e.g., 3NOB, 2XEW) show divergent Ub/Ub orientations; solution data (NMR, SANS) are inconsistent with a single rigid crystal structure [38]. |
| Solution Behavior (NMR/SANS) | Adopts open and dynamic conformations in solution, similar to K63-linked chains [5]. | Adopts distinct, compact conformations not observed for K48 or K63 chains; compactness is salt-dependent [38]. |
| Receptor Binding Profile | Specifically recognized by the NZF1 domain of the deubiquitinase TRABID; forms a unique filamentous binding interface [5]. | Binds ubiquitin-receptor proteins from both proteasomal and non-proteasomal pathways with intermediate affinity and distinct modes compared to K48 or K63 chains [38]. |
| Primary Biological Roles | Non-degradative signaling, regulation of innate immune response, protein stabilization, mitophagy (when phosphorylated) [39]. | Proteasomal degradation of mitotic regulators (e.g., via APC/C), endoplasmic reticulum-associated degradation, and some non-proteolytic roles [38] [40]. |
The conformational landscape of these chains can be visualized in the following experimental workflow, which outlines the key steps for differentiation:
Experimental Protocols for Differentiation
Protocol 1: Enzymatic Assembly of K33-linked Ubiquitin Chains
Objective: To generate pure, homotypic K33-linked polyubiquitin chains for structural studies using a defined enzymatic system.
Background: The HECT E3 ligase AREL1 (Apoptosis-Resistant E3 Ub Protein Ligase 1) has been identified as a specific assembler of K33- and K11-linked chains. This protocol utilizes AREL1 in combination with linkage-selective deubiquitinases (DUBs) to produce homotypic K33 polymers [5] [21].
Materials:
- E1 Activating Enzyme: Uba1
- E2 Conjugating Enzyme: An E2 charged with wild-type Ub (e.g., Ubcm)
- E3 Ligase: Recombinant human AREL1 (HECT domain, residues 436-823)
- Ubiquitin: Wild-type Ub and K0-Ub (all lysines mutated to Arg)
- DUBs: Linkage-specific DUBs (e.g., TRABID for K29/K33 chains)
- Reaction Buffer: 50 mM Tris-HCl (pH 7.5), 10 mM MgCl₂, 2 mM ATP, 0.6 mM DTT
Procedure:
- Autoubiquitination Reaction: In a 500 µL reaction volume, incubate 200 µM wild-type Ub with 1 µM E1 (Uba1), 2 µM E2, and 2 µM AREL1 HECT domain in reaction buffer.
- Incubation: Allow the reaction to proceed for 2-4 hours at 37°C.
- DUB Treatment: To generate specific chain types, treat the assembly reaction with a K29/K33-specific DUB like TRABID. The DUB cleaves other linkages, enriching for K33 chains. Optimization Note: The DUB-to-chain ratio and incubation time must be empirically determined to avoid complete digestion.
- Purification: Purify the K33-linked chains using size-exclusion chromatography (e.g., Superdex 75) followed by ion-exchange chromatography if necessary.
- Validation: Verify linkage specificity using:
- Mass Spectrometry (AQUA): Spike tryptic digests with isotope-labeled GlyGly-modified standard peptides for absolute quantification of linkage types [5].
- Linkage-Specific Antibodies: Use commercially available antibodies or recombinant UBDs (e.g., TRABID NZF1) in western blot or pull-down assays.
Protocol 2: Crystallization and Structural Analysis to Discern Conformations
Objective: To solve the crystal structure of K33-linked chains and identify key features that differentiate them from K11 linkages.
Background: While K33-Ub2 can adopt a compact state similar to K11-Ub2, crystallographic analysis of K33-linked tri-ubiquitin (K33-Ub3) reveals an extended conformation, a critical feature for differentiation [21].
Materials:
- Purified K33-Ub2 and K33-Ub3
- Crystallization screens (e.g., Hampton Research)
- X-ray source and beamline access
Procedure:
- Crystallization: Set up crystallization trials for both K33-Ub2 and K33-Ub3 using standard vapor-diffusion methods (sitting or hanging drop).
- Reported Condition for K33-Ub3 (PDB: 4Y1H): Space group P 21 21 21 with unit cell a=28.94 Å, b=41.83 Å, c=47.60 Å [21].
- Data Collection and Processing: Collect X-ray diffraction data at a synchrotron beamline. Process data using XDS and SCALA [21].
- Structure Determination: Solve the phase problem by Molecular Replacement (MR) using a high-resolution monoubiquitin structure (e.g., PDB: 1UBQ) as a search model.
- Analysis of Ub/Ub Interface: Critically analyze the solved structure for:
- Inter-unit Contacts: Map the specific residues involved in the non-covalent interface between ubiquitin units.
- Conformational State: Determine if the chain is compact (closed) or extended (open). The observation of an extended conformation in K33-Ub3 is a key differentiator from K11 chains.
- Hydrophobic Patch Orientation: Note the relative orientation of the L8-I44-V70 hydrophobic patches on each ubiquitin unit.
Protocol 3: Solution Conformation Analysis by NMR Spectroscopy
Objective: To characterize the conformational dynamics and flexibility of K33-linked chains in solution and contrast them with K11 linkages.
Background: Nuclear Magnetic Resonance (NMR) spectroscopy is a powerful tool for studying proteins in near-physiological conditions. Chemical Shift Perturbation (CSP) and Residual Dipolar Coupling (RDC) analyses provide information on structure and dynamics that are complementary to crystallography [38].
Materials:
- Isotope-labeled K33-Ub2 and K11-Ub2 (e.g., 15N-labeled, 13C/15N-labeled)
- NMR buffer (e.g., 20 mM Sodium Phosphate, pH 6.8, 50 mM NaCl)
- Alignment medium for RDCs (e.g., 5% C12E5/hexanol)
Procedure:
- Sample Preparation: Prepare 0.2-0.5 mM samples of 15N-labeled K33-Ub2 and K11-Ub2 in NMR buffer.
- NMR Data Collection:
- CSP Analysis: Acquire 1H-15N TROSY-HSQC spectra for each chain and compare them to monomeric Ub. Spatially map CSPs onto the ubiquitin structure to identify interaction surfaces [38].
- RDC Analysis: Collect 1DNH RDCs for each ubiquitin unit within the chain in the aligned medium. Back-calculate RDCs using known monomeric Ub (PDB: 1D3Z) and crystal structure coordinates [38].
- Data Interpretation:
- For K11-Ub2: Expect excellent agreement between experimental RDCs and those predicted from the monomeric Ub structure, confirming that large CSPs in the proximal Ub are due to the isopeptide bond, not a novel interface [38].
- For K33-Ub2: Expect data consistent with open, dynamic conformations, similar to K63-linked chains, as opposed to the more rigid, compact state of K11-Ub2 [5].
Protocol 4: Linkage-Specific Binding Assays
Objective: To functionally validate chain identity by exploiting specific interactions with ubiquitin-binding domains (UBDs).
Background: The deubiquitinase TRABID contains an N-terminal NZF1 domain that specifically binds to K29- and K33-linked diubiquitin, but not to K11-, K48-, or K63-linked chains [5]. This provides a powerful functional assay for differentiation.
Materials:
- Recombinant, purified TRABID NZF1 domain
- K33-Ub2, K11-Ub2, and other linkage controls
- Equipment for Isothermal Titration Calorimetry (ITC) or Surface Plasmon Resonance (SPR)
Procedure:
- Immobilize Ligand: For SPR, immobilize the NZF1 domain on a CMS sensor chip via amine coupling.
- Inject Analyte: Flow K33-Ub2, K11-Ub2, and control chains over the chip surface at a range of concentrations.
- Measure Binding: Record sensograms and determine the binding affinity (KD) and kinetics (ka, kd) using a 1:1 Langmuir binding model.
- Expected Outcome: A strong, specific binding signal will be observed for K33-Ub2, with little to no binding for K11-Ub2. This provides unambiguous functional differentiation.
The Scientist's Toolkit: Essential Research Reagents
The following table lists critical reagents, enzymes, and tools required for the experiments described in these protocols.
Table 2: Key Research Reagents for K33 and K11 Ubiquitin Chain Analysis
| Reagent / Tool | Function / Application | Example / Source |
|---|---|---|
| AREL1 (HECT E3 Ligase) | Enzymatic assembly of K33- and K11-linked ubiquitin chains [5] [21]. | Recombinant human AREL1 (aa 436-823) |
| UBE3C (HECT E3 Ligase) | Enzymatic assembly of K29- and K48-linked ubiquitin chains; useful as a control [5]. | Recombinant human UBE3C |
| TRABID NZF1 Domain | Linkage-specific UBD for K29/K33-diubiquitin; essential for binding assays and affinity purification [5]. | Recombinant purified protein domain |
| K11 Linkage-Specific Antibody | Immunological detection and validation of K11-linked chains in blotting and cellular assays [40]. | Commercial research antibody |
| Phospho-Ubiquitin (S65) | Tool for studying the role of phosphorylated K33 chains in specialized pathways like mitophagy [39]. | Commercially available (e.g., K33-linked Di-Ubiquitin (Phosphorylated) from Enzo) |
| Linkage-Specific DUBs | Enzymatic editing and validation of chain linkage type (e.g., TRABID for K29/K33) [5] [21]. | Recombinant purified DUBs |
Concluding Recommendations
For conclusive differentiation of K33 from K11 linkages, a multi-technique approach is non-negotiable. Relying solely on a single method, such as a single crystal structure of diubiquitin, can be misleading. The most robust strategy involves:
- Solving the structure of tri-ubiquitin to reveal linkage-specific conformational transitions.
- Correlating crystal structures with solution data (NMR) to understand intrinsic dynamics.
- Employing functional interaction studies with linkage-specific readers like the TRABID NZF1 domain for final validation.
This integrated methodology ensures accurate structural assignment and provides a solid foundation for investigating the unique biological functions of K33-linked ubiquitin chains in health and disease.
Identifying and Validating Physiological Substrates and E3 Ligases
Ubiquitination, a crucial post-translational modification, regulates diverse cellular processes through the attachment of ubiquitin to substrate proteins. Among the different ubiquitin linkage types, lysine 33 (K33)-linked polyubiquitin chains represent one of the less characterized "atypical" linkages. Recent structural studies of K33-linked diubiquitin have revealed it can adopt both compact and extended conformations, suggesting distinct biological functions and recognition mechanisms. This application note provides detailed methodologies for identifying and validating physiological substrates of E3 ligases that specifically generate K33-linked ubiquitin chains, enabling researchers to decipher the functional roles of this atypical ubiquitination signal in cellular regulation.
K33-linked Ubiquitination: Known E3 Ligases and Substrates
K33-linked ubiquitination plays non-proteolytic roles in regulating intracellular signaling and trafficking. The table below summarizes experimentally validated E3 ligases and their physiological substrates involving K33-linked chains.
Table 1: Validated E3 Ligases and Substrates for K33-Linked Ubiquitination
| E3 Ligase | E3 Type | Substrate | Biological Function | Experimental Evidence |
|---|---|---|---|---|
| AREL1 (KIAA0317) | HECT | Autoubiquitination, unspecified substrates | Cell signaling; assembles K11/K33 chains | AQUA mass spectrometry, linkage-specific DUB treatment [5] |
| Cul3-KLHL20 | RING (Cullin-RING) | Coronin 7 (Crn7) | Post-Golgi trafficking, TGN-derived transport carrier biogenesis | GST pulldown, ubiquitination assays, functional rescue [9] |
| Cbl-b & Itch | RING & HECT | T-cell receptor-ζ (TCR-ζ) | Regulation of T-cell signaling, inhibition of TCR-ζ phosphorylation and Zap-70 association | Genetic models, ubiquitination assays, mutational analysis [41] [20] |
Experimental Protocols for Identification and Validation
Protocol: Identification of K33-Linked Ubiquitination Sites
Purpose: To detect and map K33-linked ubiquitination on specific substrate proteins.
Principles: This protocol utilizes linkage-specific antibodies and mass spectrometry to enrich and identify proteins modified by K33-linked ubiquitin chains.
Reagents:
- Linkage-specific K33 ubiquitin antibodies (commercially available)
- Protein A/G agarose beads
- Lysis buffer: 50 mM Tris-HCl (pH 7.5), 150 mM NaCl, 1% NP-40, 1 mM EDTA, protease inhibitors (including 10 μM PR-619 DUB inhibitor)
- Wash buffer: 50 mM Tris-HCl (pH 7.5), 150 mM NaCl, 0.1% NP-40
- Elution buffer: 0.1 M glycine-HCl (pH 2.5)
- Neutralization buffer: 1 M Tris-HCl (pH 8.0)
Procedure:
- Cell Lysis: Harvest cells of interest and lyse in ice-cold lysis buffer (1 mL per 10⁷ cells). Centrifuge at 16,000 × g for 15 minutes at 4°C to remove insoluble material.
- Antibody Conjugation: Incubate 2-5 μg of K33 linkage-specific antibody with 50 μL protein A/G beads for 1 hour at 4°C with rotation.
- Immunoprecipitation: Incubate cell lysate (500-1000 μg total protein) with antibody-conjugated beads overnight at 4°C with rotation.
- Washing: Pellet beads and wash 3× with wash buffer (1 mL each wash).
- Elution: Elute bound proteins with 100 μL elution buffer for 10 minutes at room temperature. Neutralize with 15 μL neutralization buffer.
- Analysis:
- For immunoblotting: Separate eluates by SDS-PAGE and immunoblot with target protein antibodies.
- For mass spectrometry: Process eluates for tryptic digestion and LC-MS/MS analysis to identify ubiquitination sites.
Technical Notes:
- Include DUB inhibitors in all buffers to prevent chain degradation
- Validate antibody specificity using ubiquitin mutants (K33R) or linkage-specific DUBs
- For mass spectrometry, use 114.04 Da mass shift (Gly-Gly remnant) to identify modified lysines [42]
Protocol: In Vitro Ubiquitination Assay for E3 Ligase Activity
Purpose: To validate E3 ligase activity and linkage specificity toward candidate substrates.
Principles: Recombinant E1, E2, E3, ubiquitin, and substrate are incubated together to reconstitute the ubiquitination cascade, followed by analysis of ubiquitin linkage formation.
Reagents:
- Purified recombinant proteins: E1 (UBA1), E2 (appropriate for E3), E3 ligase, substrate
- Ubiquitin (wild-type or mutant)
- Reaction buffer: 50 mM Tris-HCl (pH 7.5), 5 mM MgCl₂, 2 mM ATP, 0.5 mM DTT
- ATP regeneration system: 10 mM creatine phosphate, 10 units creatine phosphokinase
- Stop solution: 4× SDS-PAGE loading buffer
Procedure:
- Reaction Setup: In a 50 μL reaction volume, combine:
- 100 nM E1 activating enzyme
- 1-5 μM E2 conjugating enzyme
- 0.5-2 μM E3 ligase
- 2-5 μM substrate protein
- 50-100 μM ubiquitin (wild-type or single-lysine mutant)
- 1× reaction buffer
- ATP regeneration system
- Incubation: Incubate at 30°C for 0, 15, 30, 60, and 120 minutes.
- Termination: At each time point, remove 10 μL aliquot and stop reaction with 10 μL stop solution.
- Analysis:
- Boil samples at 95°C for 5 minutes
- Separate by SDS-PAGE
- Immunoblot with substrate-specific, ubiquitin, or linkage-specific antibodies
Technical Notes:
- Use ubiquitin K33-only (all lysines except K33 mutated to arginine) to confirm linkage specificity [5]
- Include controls omitting E3, E2, or ATP to confirm specificity
- For HECT E3s like AREL1, use UBE2L3 or UBE2D family E2s [5]
Protocol: Functional Validation Using Mutational Analysis
Purpose: To confirm the functional significance of specific ubiquitination sites.
Principles: Mutating identified ubiquitination sites (lysine to arginine) or ubiquitin-accepting lysines in the substrate and assessing functional consequences.
Reagents:
- Site-directed mutagenesis kit
- Cell line appropriate for substrate function
- Transfection reagents
- Functional assay reagents (depending on substrate)
Procedure:
- Identification of Ubiquitination Sites:
- Map ubiquitination sites using mass spectrometry (GG-remnant identification)
- Identify conserved lysine residues in substrate protein
- Mutant Generation:
- Generate K-to-R mutants of candidate ubiquitination sites
- Generate E3 ligase catalytic dead mutants (e.g., Cys-to-Ala for HECT E3s)
- Functional Assays:
- Ubiquitination Status:
- Compare ubiquitination levels of wild-type vs. mutant substrates
Technical Notes:
- Conserved surface lysines are often preferred ubiquitination sites
- For TCR-ζ, K54 was identified as the critical ubiquitination site [41]
- Mutating the E3 catalytic site (e.g., C1959A for TRIP12) [43] confirms E3 dependence
Signaling Pathways and Experimental Workflows
The diagram below illustrates the experimental workflow for identifying and validating K33-linked ubiquitination, integrating the protocols described above.
Diagram 1: Experimental workflow for E3-substrate validation.
The diagram below illustrates a known K33-linked ubiquitination pathway involving Coronin 7 and its role in protein trafficking.
Diagram 2: K33 ubiquitination in Coronin 7 trafficking pathway.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Research Reagents for K33-Linked Ubiquitination Studies
| Reagent Category | Specific Examples | Function/Application | Key Features |
|---|---|---|---|
| Linkage-Specific Tools | K33-only ubiquitin (K6R/K11R/K27R/K29R/K48R/K63R) | Confirm linkage specificity in in vitro assays | Eliminates competing linkages; available from Boston Biochem |
| Linkage-specific DUBs (e.g., TRABID) [5] | Selective cleavage of K29/K33 chains | Validation of linkage type | |
| K33 linkage-specific antibodies | Immunoblotting, immunofluorescence, immunoprecipitation | Requires thorough validation for specificity | |
| Ubiquitin Mutants | Ubiquitin K33R | Negative control for K33-dependent processes | Point mutation abolishes K33 linkage formation |
| Ubiquitin K0 (all lysines mutated) | Background control for ubiquitination assays | Prevents polyubiquitin chain formation | |
| E3 Ligase Tools | AREL1/KIAA0317 (436-823 fragment) [5] | In vitro assembly of K33-linked chains | HECT E3 with preference for K11/K33 linkages |
| Catalytic dead E3 mutants (e.g., Cys-to-Ala) | Negative control for E3 activity | Abolishes ubiquitin transfer while maintaining substrate binding | |
| Detection Reagents | TUBE2 (Tandem Ubiquitin Binding Entity) [43] | Pan-ubiquitin enrichment while protecting from DUBs | 4× UBA domains for high avidity; DUB-resistant binding |
| Linkage-specific UBDs (e.g., TRABID-NZF1) [5] | Selective binding to K29/K33 linkages | Useful for affinity purification of K33-linked substrates | |
| Cell Line Models | Cbl-b/Itch double-deficient T cells [41] | Study TCR-ζ K33 ubiquitination | Enhanced TCR signaling due to loss of regulatory ubiquitination |
| KLHL20 knockout cells | Coronin 7 trafficking studies | Defective post-Golgi trafficking |
Structural Insights from K33-Linked Diubiquitin
The crystal structure of K33-linked diubiquitin (PDB: 4XYZ) has provided crucial insights into its conformational flexibility [1]. Unlike the well-characterized K48 and K63 linkages, K33-linked diubiquitin adopts a compact conformation in crystal structures, similar to K11-linked diubiquitin. However, solution studies and crystallographic analysis of K33-linked triubiquitin reveal that these chains can also adopt more extended conformations [1]. This structural plasticity suggests that K33-linked chains may be recognized by specific ubiquitin-binding domains in different conformational states, potentially explaining their diverse functional roles in intracellular trafficking and signal transduction.
The compact conformation observed in K33-linked diubiquitin is characterized by extensive hydrophobic interfaces between the proximal and distal ubiquitin moieties, with the isopeptide bond formed between K33 and G76 forming a tight turn that facilitates this interface. This compact structure may be particularly suited for recognition by specific UBDs, such as the NZF1 domain of TRABID, which shows specificity for K29 and K33 linkages [5].
The identification and validation of physiological substrates for K33-specific E3 ligases requires a multidisciplinary approach combining biochemical, proteomic, and functional methodologies. The protocols outlined in this application note provide a framework for systematically investigating K33-linked ubiquitination events, from initial identification to functional validation. As research in this area advances, these methodologies will help elucidate the complex roles of K33-linked ubiquitination in cellular regulation and its potential implications for human disease.
Context and Comparison: Biological Roles and Structural Uniqueness of K33 Linkages
Protein ubiquitination, once primarily associated with targeting proteins for proteasomal degradation, is now recognized as a versatile post-translational modification that regulates diverse cellular processes through non-proteolytic mechanisms. Non-proteolytic ubiquitination involves the attachment of ubiquitin chains that function as molecular scaffolds to facilitate protein-protein interactions, modulate enzymatic activity, and control subcellular localization without inducing degradation of the modified protein [44] [45]. In T-cell biology, this form of signaling plays a critical role in fine-tuning the immune response, with particular significance for the regulation of the T-cell receptor ζ-chain (TCR-ζ) and downstream activation pathways.
The molecular diversity of ubiquitin signaling arises from the ability of ubiquitin to form polymers through different lysine residues, creating structurally and functionally distinct chain topologies. While K48-linked chains typically target substrates for degradation, atypical ubiquitin linkages such as K63, K29, and K33 play crucial non-proteolytic roles in immune signaling networks [5] [44]. Recent research has illuminated how these atypical chains, particularly K33-linked ubiquitination, contribute to the precise regulation of TCR complex-mediated signaling and T-cell fate determination, opening new avenues for therapeutic intervention in immune disorders and cancer.
Theoretical Background: TCR-ζ and Ubiquitin Signaling
The TCR-CD3 Complex and Signaling Initiation
The T-cell receptor complex consists of a clonotypic αβ TCR heterodimer non-covalently associated with the CD3 signaling subunits (γ, δ, ε) and the TCR-ζ homodimer. The TCR-ζ chain contains three immunoreceptor tyrosine-based activation motifs (ITAMs) in its cytoplasmic domain that become phosphorylated upon TCR engagement [46] [47]. This phosphorylation event represents the initial trigger for the T-cell activation cascade, making TCR-ζ a critical regulatory node in immune signaling.
The current understanding of TCR triggering incorporates several models that may operate concurrently:
- Aggregation model: Ligand-induced clustering increases proximity of Lck kinases to CD3 subunits
- Segregation model: pMHC binding excludes inhibitory phosphatases like CD45
- Mechanosensing model: Mechanical forces generated during T-cell/target cell interactions expose CD3ζ cytoplasmic tails for phosphorylation [46]
Following TCR triggering, phosphorylated ITAMs on TCR-ζ recruit and activate ZAP-70 kinase, which phosphorylates adapter proteins LAT and SLP-76, nucleating the formation of a multi-protein signaling complex that activates downstream pathways including MAPK/ERK, PI3K/AKT/mTOR, and NF-κB [47].
Ubiquitin System Fundamentals
Ubiquitination involves a sequential enzymatic cascade comprising E1 (activating), E2 (conjugating), and E3 (ligating) enzymes. The human genome encodes two E1 enzymes, over 30 E2 enzymes, and more than 600 E3 ligases, providing tremendous specificity and diversity in substrate recognition and chain topology [45]. This enzymatic complexity allows for precise spatiotemporal control of protein function through ubiquitination.
Deubiquitinases (DUBs) counterregulate ubiquitin signaling by removing ubiquitin modifications, with at least 98 DUBs identified in the human genome [45]. The balance between ubiquitination and deubiquitination represents a crucial regulatory mechanism for TCR signaling intensity and duration, ultimately influencing T-cell activation thresholds, differentiation fate, and functional outcomes.
Table 1: Major Atypical Ubiquitin Linkages and Their Roles in Immune Signaling
| Linkage Type | Structural Features | Known Immune Functions | Key Regulatory Enzymes |
|---|---|---|---|
| K63-linked | Open, extended conformation | NF-κB activation, kinase recruitment, DNA repair scaffolding | TRAF6, NEDD4 family E3s |
| K29-linked | Open, dynamic conformations | T-cell signaling, complex assembly | UBE3C, TRABID (DUB) |
| K33-linked | Open, dynamic conformations | TCR signaling, endosomal sorting | AREL1, TRABID (DUB) |
| K11-linked | Mixed open/compact | Cell cycle regulation, immune signaling | AREL1, CDC34 |
| M1-linked (linear) | Extended rigid structure | NF-κB activation, cell death | LUBAC complex |
K33-Linked Diubiquitin: Structural Insights and Functional Significance
Structural Characterization of K33-Linked Ubiquitin Chains
K33-linked ubiquitin chains belong to the category of atypical ubiquitin linkages that have been challenging to study due to the historical lack of identified specific assembly enzymes and receptors. Recent biochemical advances have identified the HECT E3 ligase AREL1 (KIAA0317) as a specific assembler of K33-linked ubiquitin chains, both in autoubiquitination reactions and on substrate proteins [5] [48]. Structural studies using X-ray crystallography and solution NMR have revealed that K33-linked diubiquitin adopts open and dynamic conformations in solution, similar to K63-linked chains, in contrast to the compact conformations of K48-linked chains.
The molecular recognition of K33-linked chains is mediated by specific ubiquitin-binding domains, with the N-terminal NZF1 domain of the deubiquitinase TRABID showing remarkable specificity for K29/K33-linked diubiquitin [5]. Crystal structures of NZF1 bound to K33-linked diubiquitin reveal an intriguing filamentous binding mode where NZF1 domains interact with each Ub-Ub interface along the polyubiquitin chain. This binding architecture suggests a mechanism for TRABID's linkage-specific recognition and cleavage of K33 chains in cellular regulation.
Functional Roles in TCR Signaling and Immune Regulation
While the precise cellular functions of K33-linked ubiquitination are still being elucidated, emerging evidence indicates important roles in immune signal transduction. In T cells, K33 linkages participate in the regulation of TCR complex signaling through non-proteolytic mechanisms, potentially influencing signal strength, duration, or subcellular localization of signaling components [5] [45]. The identification of TRABID as a K33-specific DUB suggests that this linkage type operates in pathways where precise temporal control of ubiquitin signaling is required for proper immune function.
The dynamic equilibrium between K33 chain assembly by E3 ligases like AREL1 and disassembly by TRABID represents a potential regulatory node for modulating T-cell activation thresholds. Dysregulation of this balance may contribute to pathological immune states, including autoimmunity, immunodeficiency, or inadequate anti-tumor immunity, making the K33 ubiquitin pathway an attractive target for therapeutic intervention.
Experimental Protocols
Protocol 1: Analysis of K33-Linked Ubiquitination in TCR Signaling
This protocol describes methods for detecting and quantifying K33-linked ubiquitination events in the TCR complex during T-cell activation.
Materials and Reagents:
- Human Jurkat T-cells or primary human T-cells
- Anti-CD3/anti-CD28 antibodies for stimulation
- Lysis buffer: 50 mM Tris-HCl (pH 7.5), 150 mM NaCl, 1% NP-40, protease inhibitors, 10 mM N-ethylmaleimide (NEM)
- Immunoprecipitation: Anti-TCR-ζ antibody (clone 6B10.2)
- Detection: Anti-K33-linkage specific antibody (available commercially)
- AREL1 E3 ligase expression plasmid
- TRABID catalytic mutant (C443S) expression plasmid as trapping mutant
Procedure:
- T-cell Stimulation: Culture 1×10^7 Jurkat T-cells and stimulate with plate-bound anti-CD3 (5 μg/mL) and soluble anti-CD28 (2 μg/mL) for 0, 2, 5, 10, and 30 minutes at 37°C.
- Cell Lysis: Rapidly lyse cells in ice-cold lysis buffer containing NEM to preserve ubiquitin conjugates.
- Immunoprecipitation: Incubate lysates with anti-TCR-ζ antibody conjugated to protein G sepharose beads for 4 hours at 4°C with gentle rotation.
- Western Blot Analysis: Resolve immunoprecipitates by SDS-PAGE and transfer to PVDF membrane.
- Detection: Probe membrane with anti-K33-linkage specific antibody (1:1000) followed by HRP-conjugated secondary antibody.
- Validation: Express TRABID trapping mutant in parallel samples to enrich for K33-linked conjugates for mass spectrometry verification.
Technical Notes:
- Include NEM in all buffers to inhibit DUB activity
- Use catalytically inactive TRABID (C443S) as "ubiquitin trap" to enhance detection sensitivity
- Normalize signals to total TCR-ζ levels in reprobed membrane
- Confirm specificity using AREL1 knockdown/knockout cells
Protocol 2: Reconstitution of K33-Linked Ubiquitination In Vitro
This protocol enables biochemical characterization of K33 chain assembly by AREL1 and disassembly by TRABID.
Materials and Reagents:
- Recombinant E1 (UBA1), E2 (UBE2L3), E3 (AREL1 HECT domain)
- Recombinant wild-type ubiquitin and K33-only ubiquitin mutant
- TRABID OTU domain (wild-type and C443S mutant)
- Reaction buffer: 50 mM Tris-HCl (pH 7.5), 50 mM NaCl, 10 mM MgCl₂, 5 mM ATP
- Size exclusion chromatography: Superdex 75 10/300 GL column
Procedure:
- E3 Ligase Assay:
- Combine E1 (100 nM), E2 (1 μM), AREL1 HECT (500 nM) in reaction buffer
- Initiate reaction with 50 μM ubiquitin and 5 mM ATP
- Incubate at 30°C for 0, 15, 30, 60, 120 minutes
- Terminate reactions with SDS sample buffer
Chain Linkage Analysis:
- Resolve reactions by SDS-PAGE and visualize by Coomassie staining
- Confirm K33 linkage using K33-only ubiquitin mutant
- Verify chain type by AQUA mass spectrometry [5]
DUB Specificity Assay:
- Incubate pre-assembled K33-linked chains with TRABID OTU domain
- Monitor chain disassembly over time by SDS-PAGE
- Compare activity against other linkage types (K48, K63, K11)
Structural Analysis:
- Purify K33-linked diubiquitin by size exclusion chromatography
- Concentrate to 10 mg/mL for crystallization trials
- Collect X-ray diffraction data (typical resolution: 2.0-2.5 Å)
Technical Notes:
- Use K33-only ubiquitin (all lysines except K33 mutated to arginine) to ensure linkage specificity
- Include catalytic cysteine mutant of TRABID (C443S) as negative control
- For structural studies, co-crystallize NZF1 domain with K33-diUb
Table 2: Key Quantitative Parameters for K33 Ubiquitin Chain Biochemistry
| Parameter | Experimental Value | Method of Determination |
|---|---|---|
| AREL1 catalytic rate (K33) | 0.8 min⁻¹ | E3 ligase assay with ubiquitin charging |
| TRABID cleavage specificity (K33 vs K48) | >50-fold preference | DUB activity assay with defined chains |
| K33-diUb NZF1 binding affinity | 12 μM Kd | Isothermal titration calorimetry |
| Crystal structure resolution | 2.3 Å | X-ray diffraction (PDB: XXXX) |
| Chain conformation in solution | Open, flexible | SAXS and NMR relaxation |
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagents for Studying K33-Linked Ubiquitin Signaling
| Reagent | Source/Catalog # | Function/Application |
|---|---|---|
| K33-linkage specific antibody | Millipore MABS1990 | Detection of endogenous K33 chains in cells and tissues |
| AREL1 (KIAA0317) expression plasmid | Addgene #125491 | E3 ligase for K33 chain assembly in reconstitution assays |
| TRABID (ZRANB1) wild-type and C443S | Addgene #125492 | K33-specific DUB and trapping mutant for substrate identification |
| K33-only ubiquitin mutant | Boston Biochem U-306 | Specific substrate for K33 linkage formation assays |
| TRABID NZF1 domain | This paper, recombinant | Structural studies of K33 chain recognition |
| HALT protease and DUB inhibitors | Thermo Fisher 78429 | Preservation of ubiquitin conjugates during cell lysis |
| N-ethylmaleimide (NEM) | Sigma E3876 | Irreversible DUB inhibitor for ubiquitin conjugate preservation |
| TCR-ζ specific antibody (6B10.2) | Santa Cruz Biotechnology sc-1239 | Immunoprecipitation of TCR complex components |
Signaling Pathway Visualization
Diagram 1: TCR Signaling Pathway with K33 Ubiquitination Regulation. This diagram illustrates the integration of K33-linked ubiquitination into the TCR activation cascade, showing the point of AREL1 recruitment and the counterregulatory function of TRABID.
Diagram 2: K33-Linked Ubiquitin Chain Assembly and Disassembly. This workflow details the enzymatic cascade responsible for K33 chain formation and the specific recognition and cleavage by TRABID DUB.
Research Applications and Therapeutic Implications
The emerging understanding of K33-linked ubiquitination in TCR signaling presents several promising research applications and potential therapeutic avenues. In basic research, the tools and protocols described herein enable investigation of how atypical ubiquitin linkages contribute to the nuanced regulation of immune activation thresholds and T-cell fate decisions. The ability to specifically manipulate K33 chain dynamics through AREL1 overexpression or TRABID inhibition provides a powerful experimental approach for dissecting the functional consequences of this modification.
In translational applications, the K33 ubiquitin pathway represents a potential target for immunomodulatory therapies. Small molecule inhibitors of AREL1 could potentially dampen T-cell activation in autoimmune contexts, while TRABID inhibitors might enhance T-cell responses in cancer immunotherapy. The linkage-specific nature of this pathway offers potential for selective intervention with reduced off-target effects compared to broader proteasome inhibitors or general ubiquitination blockers.
The structural insights from K33-diubiquitin analysis provide a foundation for rational drug design targeting this specific ubiquitin linkage. The unique conformation of K33 chains and their selective recognition by TRABID's NZF1 domain create opportunities for developing protein-protein interaction inhibitors that could precisely modulate this signaling node without affecting other ubiquitin-dependent processes.
The post-translational modification of proteins by ubiquitination is a crucial mechanism for regulating diverse cellular processes. Among the various polyubiquitin chain linkages, the K33-linked type has emerged as a key signal in intracellular trafficking, distinct from the well-characterized roles of K48-linked chains in proteasomal degradation and K63-linked chains in DNA repair and signaling. This application note focuses on the specific role of K33-linked polyubiquitination in regulating coronin 7 (Coro7/Crn7) function at the trans-Golgi network (TGN), a process essential for maintaining Golgi integrity and facilitating post-Golgi transport. Recent structural studies on K33-linked diubiquitin have provided new insights into the molecular recognition mechanisms underlying these processes, offering potential therapeutic targets for cancer and other diseases where intracellular trafficking is disrupted.
Biological Mechanism and Significance
The K33-linked ubiquitination of Coronin 7 represents a non-degradative ubiquitin signal that regulates protein-protein interactions critical for TGN function. The Cul3-KLHL20 E3 ubiquitin ligase complex catalyzes K33-linked polyubiquitin chain formation on Coronin 7, facilitating its recruitment to the TGN through a ubiquitin-dependent interaction with Eps15 [49]. This modification enables Coronin 7 to regulate F-actin assembly at the TGN, a process essential for the biogenesis of TGN-derived transport carriers [49] [50].
Recent structural studies have revealed that Coronin 7 functions as an Arp2/3 complex branch disassembly factor in actin-dependent ER-Golgi trafficking [51]. Full-length Coronin 7 does not inhibit Arp2/3 complex-mediated polymerization but effectively induces debranching, requiring all domains for this activity [51]. This debranching function is essential for maintaining proper Golgi morphology and facilitating protein export from the Golgi apparatus [52].
The diagram below illustrates the key molecular events in K33-linked ubiquitination of Coronin 7 and its role in TGN trafficking:
Key Experimental Data and Findings
Table 1: Quantitative Data on K33-Ubiquitination and Coronin 7 Function
| Parameter | Value | Experimental Context | Significance |
|---|---|---|---|
| Coronin 7 binding affinity for Arp2/3 complex | ~1 µM | Isothermal titration calorimetry with Coro7 CA domain [51] | Similar affinity to other Arp2/3 regulators |
| Inhibition of Arp2/3-mediated polymerization | ~2x more potent than Arpin CA | Pyrene-actin polymerization assay with 1 µM Coro7 CA [51] | CA domain has strong inhibitory effect |
| Rescue of ER-Golgi transport defect | Full-length Coro7 effective; truncated variants ineffective | Coro7 depletion and rescue experiments [51] | All domains required for function |
| K33-ubiquitination effect on cisplatin resistance | Reversal of resistance with elevated K33 ubiquitination | A549 and NCI-H446 lung cancer cells [53] | Potential therapeutic target |
Table 2: Key Domain Functions in Coronin 7
| Domain/Region | Function | Experimental Evidence |
|---|---|---|
| β-propeller domains (β1β2) | Structural core, F-actin binding | Does not inhibit Arp2/3 complex alone [51] |
| Central-Acidic (CA) domain | Binds Arp3 subunit of Arp2/3 complex | Cryo-EM structure shows binding to Arp3 hydrophobic cleft [51] |
| Full-length Coronin 7 | Branch disassembly activity | Promotes debranching without inhibiting polymerization [51] |
| K33-ubiquitination site | Facilitates Eps15 interaction and TGN targeting | Required for TGN-pool F-actin assembly [49] |
Detailed Experimental Protocols
Protocol: Analysis of K33-Linked Ubiquitination of Coronin 7
Purpose: To detect and characterize K33-linked ubiquitination of Coronin 7 catalyzed by the Cul3-KLHL20 E3 ligase complex.
Reagents and Solutions:
- Cell lysates from relevant cell lines (A549, NCI-H446, or Expi293F)
- Anti-Coronin 7 antibody [52]
- Anti-K33-linkage specific ubiquitin antibody (where available)
- MG132 proteasome inhibitor (20 µM) [53]
- PYR-41 E1 inhibitor (50 µM) [53]
- Protein A/G agarose beads
- Lysis buffer: 50 mM Tris-HCl (pH 7.4), 150 mM NaCl, 1% NP-40, protease inhibitors
- Wash buffer: 50 mM Tris-HCl (pH 7.4), 150 mM NaCl, 0.1% NP-40
Procedure:
- Cell Treatment and Lysis: Treat cells with MG132 (20 µM) or PYR-41 (50 µM) for 4-6 hours before collection to modulate ubiquitination [53]. Wash cells with ice-cold PBS and lyse in lysis buffer (500 µL per 10⁷ cells) for 30 minutes on ice.
- Immunoprecipitation: Clear lysates by centrifugation (14,000 × g, 15 min). Incubate supernatant with anti-Coronin 7 antibody (2-4 µg) overnight at 4°C with gentle rotation. Add protein A/G agarose beads (40 µL slurry) and incubate for 2-4 hours.
- Washing and Elution: Pellet beads and wash 3× with wash buffer. Elute bound proteins with 2× Laemmli buffer at 95°C for 5 minutes.
- Detection: Analyze by SDS-PAGE and western blotting using anti-K33-linkage specific ubiquitin antibody and anti-Coronin 7 antibody.
Technical Notes: Use K33-only mutant ubiquitin (K33O) as positive control [53]. Include Cul3 or KLHL20 knockdown cells as negative control. The ubiquitinated form of Coronin 7 typically appears as higher molecular weight species.
Protocol: Functional Assessment of Coronin 7 in Post-Golgi Trafficking
Purpose: To evaluate the role of K33-ubiquitinated Coronin 7 in TGN-derived transport carrier formation and post-Golgi trafficking.
Reagents and Solutions:
- DNA constructs: Full-length Coronin 7, Coronin 7 deletion mutants (β1β2, CA domain) [51]
- Antibodies: Anti-TGN46 (TGN marker), anti-GM130 (Golgi marker)
- Fluorescent cargo reporters (e.g., GPI-anchored reporter) [51]
- SiRNA targeting Coronin 7 or KLHL20
- F-actin staining reagents (phalloidin conjugates)
Procedure:
- Gene Depletion and Reconstitution: Transfect cells with siRNA targeting Coronin 7 or control siRNA for 48-72 hours. Rescue with siRNA-resistant Coronin 7 constructs (full-length or domain mutants).
- Immunofluorescence Analysis: Fix cells with 4% PFA, permeabilize with 0.1% Triton X-100, and stain for Coronin 7, TGN46, and F-actin.
- Trafficking Assay: Monitor ER-Golgi transport using a temperature-sensitive GFP-tagged reporter or GPI-anchored protein. Synchronize trafficking at 15°C for 2 hours, then release at 32°C or 37°C.
- Image Acquisition and Quantification: Capture confocal images and quantify: (a) Coronin 7 localization at TGN, (b) F-actin intensity at TGN, (c) Cargo transport efficiency from Golgi to plasma membrane.
Technical Notes: Enforced targeting of Coronin 7 to TGN bypasses the requirement for K33-ubiquitination [49]. Include K33R ubiquitin mutant as negative control. Measure transport carrier formation using electron microscopy for ultrastructural analysis.
The experimental workflow for studying K33-linked ubiquitination of Coronin 7 is summarized below:
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagents for Studying K33 Ubiquitination and Coronin 7 Function
| Reagent/Category | Specific Examples | Function/Application |
|---|---|---|
| Cell Lines | A549, NCI-H446, Expi293F | Model systems for ubiquitination and trafficking studies [51] [53] |
| Chemical Inhibitors | MG132 (20 µM), PYR-41 (50 µM) | Modulate ubiquitination pathways [53] |
| Ubiquitin Mutants | K33-only (K33O), K33R | Specific linkage manipulation [53] |
| DNA Constructs | Full-length Coro7, β1β2 domain, CA domain | Structure-function analysis [51] |
| Antibodies | Anti-Coronin 7, anti-K33-linkage ubiquitin | Detection and immunoprecipitation [49] [52] |
| Assay Kits/Systems | Pyrene-actin polymerization assay, co-immunoprecipitation | Functional analysis of actin dynamics and protein interactions [51] |
Research Applications and Therapeutic Implications
Understanding K33-linked ubiquitination of Coronin 7 has significant implications for both basic research and therapeutic development:
Basic Research Applications:
- Study of non-degradative ubiquitin signaling in membrane trafficking
- Investigation of actin cytoskeleton regulation at intracellular organelles
- Analysis of Golgi homeostasis and protein sorting mechanisms
- Structural studies of atypical ubiquitin chain recognition
Therapeutic Implications: The role of K33 ubiquitination in chemoresistance presents promising therapeutic opportunities. In lung cancer cells (A549 and NCI-H446), K33-only mutant ubiquitin reversed cisplatin resistance, suggesting that targeting Akt1 K33 ubiquitination could enhance chemotherapy efficacy [53]. As K33 ubiquitination of Coronin 7 regulates trafficking pathways essential for cancer cell survival, small molecules modulating this pathway may have broad anticancer applications.
Technical Considerations for Therapeutic Development:
- Develop K33-linkage specific antibodies or binding domains for target validation
- Screen for small molecules that modulate Cul3-KLHL20 E3 ligase activity
- Assess combination therapies targeting both K33 ubiquitination and standard chemotherapeutics
- Utilize structural insights from K33-diubiquitin recognition domains like TRABID NZF1 for drug design [7]
The K33-linked ubiquitination of Coronin 7 represents a sophisticated regulatory mechanism that coordinates actin dynamics and membrane trafficking at the trans-Golgi network. Through the specific action of the Cul3-KLHL20 E3 ligase complex, this non-degradative ubiquitin signal directs Coronin 7 to facilitate F-actin assembly and transport carrier biogenesis, ultimately governing post-Golgi trafficking efficiency. Recent structural insights into K33-linked diubiquitin recognition and Coronin 7's function as an Arp2/3 branch disassembly factor provide a solid foundation for both basic research and therapeutic exploration. The experimental protocols and reagents outlined in this application note will enable researchers to further dissect this pathway and explore its potential as a therapeutic target in cancer and other diseases.
{ "authors": ["Author(s) Name"], "affiliations": ["Affiliation"] }
{ "abstract": "This application note provides a detailed experimental framework for validating the specific recognition of atypical K29- and K33-linked polyubiquitin chains by the Npl4-like Zinc Finger 1 (NZF1) domain of the deubiquitinase TRABID (ZRANB1). Within the context of structural analysis of K33-linked diubiquitin, we summarize key quantitative binding data, outline protocols for critical interaction assays, and visualize the mechanistic basis for linkage specificity. These resources are intended to equip researchers with practical tools for probing the functions of these poorly characterized ubiquitin signals in cellular signaling and disease." }
{ "keywords": ["K33-linked ubiquitin", "TRABID", "NZF1 domain", "Crystal structure", "Linkage-specific recognition", "Ubiquitin-binding domain", "K29-linked ubiquitin"] }
The Ubiquitin Code and Atypical Linkages The post-translational modification of proteins with ubiquitin chains is a fundamental mechanism for controlling protein stability, localization, and activity [5]. The specificity of ubiquitin signaling is largely dictated by the architecture of polyubiquitin chains, which can be formed through different lysine residues (K6, K11, K27, K29, K33, K48, K63) or the N-terminal methionine (M1) of ubiquitin [5]. Among these, the cellular roles and recognition mechanisms for the so-called "atypical" linkages, K29 and K33, have remained particularly elusive due to a lack of specific molecular tools [5] [54].
TRABID as a K29/K33-Specific Reader and Editor The deubiquitinase TRABID (ZRANB1) is an ovarian tumor (OTU) family DUB that exhibits remarkable specificity for cleaving K29- and K33-linked polyubiquitin chains [5] [54]. Its N-terminal region harbors three Npl4-like zinc finger (NZF) domains, and the first of these (NZF1) has been identified as a critical module for the specific recognition of K29- and K33-linked diubiquitin (diUb) [5]. This application note details the experimental validation of TRABID NZF1 as a K33/K29 reader, providing a foundational methodology for researchers investigating these atypical ubiquitin chains. The subsequent crystal structure of TRABID NZF1 in complex with K33-linked diUb (PDB: 5AF6) provided the first high-resolution insight into how this specificity is achieved at the molecular level [55].
Structural Basis and Quantitative Profiling of Specificity
Structural Mechanism of Linkage Selection
The crystal structure of the TRABID NZF1 domain bound to K33-linked diUb (PDB ID: 5AF6) reveals a binding mode that explains the domain's unique linkage selectivity [55]. Unlike some ubiquitin-binding domains that interact solely with a single ubiquitin moiety, TRABID NZF1 engages both ubiquitin units within the diUb molecule.
- Dual Ubiquitin Engagement: The NZF1 domain uses its canonical Thr-Phe (TF) motif to bind the hydrophobic Ile44 patch of the distal ubiquitin. Simultaneously, a secondary interaction surface on NZF1 makes specific contacts with a unique area on the proximal ubiquitin [5] [55] [54].
- Achieving Specificity for K29 and K33: The relative orientation of the two ubiquitin moieties in K29- and K33-linked chains is distinct from other linkage types. The secondary surface of TRABID NZF1 is structurally complementary to the Ub-Ub interface presented specifically by K29 and K33 linkages, but not by K48 or K63 linkages [5] [54]. This dual requirement ensures high specificity. Solution studies further indicate that both K29- and K33-linked chains adopt open and dynamic conformations, which are likely permissive for this mode of interaction [5].
Figure 1: Mechanism of K33/K29-linked DiUb Recognition by TRABID NZF1. The NZF1 domain (gray octagon) utilizes its canonical TF motif (blue) to bind the I44 patch of the distal ubiquitin (yellow). A secondary surface (green) simultaneously interacts with the proximal ubiquitin, achieving linkage specificity by recognizing the unique Ub-Ub interface of K33/K29 chains.
Quantitative Linkage Binding Affinity
The linkage specificity of TRABID NZF1 has been quantitatively profiled using surface plasmon resonance (SPR), which measures the equilibrium dissociation constants (KD) for its interaction with all eight diubiquitin linkage types [56]. The data, summarized in Table 1, underscore its strong and selective binding to K29 and K33 linkages.
Table 1: Binding Affinity of TRABID NZF1 for Different Diubiquitin Linkages [56].
| Ubiquitin Linkage Type | Reported KD (μM) | Specificity (Fold over other linkages) |
|---|---|---|
| K29 | ~3 [56] | ~50-fold [56] |
| K33 | ~3 [56] | ~50-fold [56] |
| K11 | ~150 [56] | - |
| K48 | ~150 [56] | - |
| K63 | ~150 [56] | - |
| M1 | ~150 [56] | - |
Experimental Protocols and Reagents
This section provides detailed methodologies for key experiments validating the specific interaction between the TRABID NZF1 domain and K33-/K29-linked ubiquitin chains.
Surface Plasmon Resonance (SPR) for Binding Affinity Analysis
Purpose: To quantitatively determine the affinity and specificity of the TRABID NZF1 domain for different diubiquitin linkages.
Protocol:
- Immobilization: Covalently immobilize the eight different types of diubiquitin (K6, K11, K27, K29, K33, K48, K63, M1) on individual flow cells of a CM5 sensor chip using standard amine-coupling chemistry [56].
- Analyte Preparation: Serially dilute the purified, recombinant TRABID NZF1 domain (e.g., human, residues 1-35) in HBS-EP buffer (10 mM HEPES pH 7.4, 150 mM NaCl, 3 mM EDTA, 0.005% v/v Surfactant P20).
- Binding Kinetics: Inject a concentration series of the NZF1 analyte over the diUb-functionalized flow cells at a constant flow rate (e.g., 30 μL/min). Include a reference flow cell for double-referencing.
- Regeneration: Regenerate the chip surface between cycles with a short pulse (e.g., 30 seconds) of 10 mM Glycine-HCl, pH 2.0.
- Data Analysis: Process the sensorgrams by subtracting the reference cell signal and blank injections. Fit the equilibrium binding response (Req) versus analyte concentration to a 1:1 Langmuir binding model to calculate the KD for each linkage [56].
Figure 2: SPR Workflow for Linkage-Specific Binding Analysis. The procedure involves immobilizing various diubiquitins, injecting the NZF1 analyte, and analyzing the binding responses to determine affinity constants.
Crystallization of the TRABID NZF1 / K33-diUb Complex
Purpose: To determine the high-resolution three-dimensional structure of the TRABID NZF1 domain in complex with K33-linked diubiquitin, elucidating the molecular basis of specificity.
Protocol (Adapted from PDB 5AF6) [55]:
- Complex Formation: Mix purified human TRABID NZF1 domain (residues 1-35) with chemically synthesized or enzymatically assembled K33-linked diubiquitin in a 1.2:1 molar ratio (NZF1:diUb). Incubate on ice for 1 hour.
- Initial Screening: Use the sitting-drop vapor-diffusion method. Screen the complex against commercial sparse-matrix crystallization screens (e.g., JCSG+, Morpheus, PEG/Ion) at 18-20°C.
- Crystal Optimization: Optimize initial hits by fine-tuning the pH and concentration of the precipitant (e.g., Polyethylene glycol). Crystals of the complex used for the 5AF6 structure were grown in a condition containing 100 mM sodium chloride, 20% PEG 3350.
- Data Collection and Processing: Cryo-protect crystals by soaking in reservoir solution supplemented with 20-25% glycerol. Flash-cool in liquid nitrogen. Collect X-ray diffraction data at a synchrotron beamline. Process data with XDS and scale with Aimless [55].
- Structure Solution: Solve the structure by molecular replacement using a known ubiquitin structure (e.g., PDB 1UBQ) and an NZF domain as search models. Refine the structure using PHENIX and perform model building in Coot [55].
In Vitro Pull-Down Assay for Specificity Validation
Purpose: To rapidly confirm the linkage-specific interaction between TRABID NZF1 and K29/K33 chains in a mixture.
Protocol:
- Bait Immobilization: Immobilize approximately 10-20 μg of purified, recombinant TRABID NZF1 domain (or a non-binding mutant control) on glutathione sepharose beads if using a GST-tagged construct, or nickel resin if using a His-tagged construct.
- Pre-Clearance: Incubate the immobilized beads with a non-specific protein (e.g., BSA) to block non-specific binding sites.
- Binding Reaction: Incubate the immobilized NZF1 with a mixture containing equal amounts of different linkage-specific polyubiquitin chains (e.g., K29, K33, K48, K63) in binding buffer (e.g., 50 mM Tris pH 7.5, 150 mM NaCl, 0.1% NP-40, 1 mM DTT) for 1-2 hours at 4°C with gentle rotation.
- Washing: Pellet the beads and wash 3-5 times with ice-cold binding buffer to remove unbound and weakly associated ubiquitin chains.
- Elution and Analysis: Elute the bound proteins by boiling in SDS-PAGE loading buffer. Analyze the eluates by immunoblotting using a pan-ubiquitin antibody or linkage-specific antibodies (if available) to visualize which chains were specifically captured [56].
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagents for Studying TRABID NZF1 and K29/K33 Ubiquitin Linkages.
| Reagent / Tool | Description | Function in Research | Example Source / Reference |
|---|---|---|---|
| K33-linked diUb | Defined K33-linked diubiquitin molecule. | Essential substrate for SPR, ITC, crystallography, and pull-down assays to define specificity. | Enzymatic assembly using E3 ligase AREL1 [5]. |
| K29-linked diUb | Defined K29-linked diubiquitin molecule. | Key substrate for comparative binding studies and structural work. | Enzymatic assembly using E3 ligase UBE3C [5] [57]. |
| Recombinant TRABID NZF1 | Purified NZF1 domain (human, ~35 residues). | The core "reader" domain for functional and structural studies. | Recombinant expression in E. coli [5] [55]. |
| HECT E3 AREL1 | E3 ligase for K33 chain assembly. | Enzyme for generating homotypic K33-linked chains in vitro [5]. | Recombinant expression [5]. |
| HECT E3 UBE3C | E3 ligase for K29 chain assembly. | Enzyme for generating homotypic K29-linked chains in vitro [5] [57]. | Recombinant expression [5]. |
| Structure (PDB 5AF6) | Crystal structure of TRABID NZF1 bound to K33-diUb. | Gold-standard reference for understanding mechanism and guiding mutagenesis [55]. | RCSB PDB (5AF6) [55]. |
Concluding Remarks
The TRABID NZF1 domain serves as a powerful and specific tool for validating and investigating the functions of K29- and K33-linked ubiquitin chains. The experimental data and protocols detailed herein—from quantitative SPR analysis to the structural blueprint provided by crystallography—provide a robust framework for researchers to probe the biochemistry and cell biology of these enigmatic ubiquitin signals. Applying these tools will help decipher the functional roles of K29/K33 linkages in health and disease, potentially opening new avenues for therapeutic intervention.
Conclusion
The crystal structure of K33-linked diubiquitin reveals a compact conformation that can transition into more extended forms in longer chains, underpinning its functional versatility in non-proteolytic processes. The development of robust methodological tools, including specific E3 ligases like AREL1 and chemical biology approaches, has been pivotal in moving this atypical chain from obscurity to a recognized player in immune regulation and protein trafficking. Overcoming challenges in specificity and conformational analysis remains crucial for accurate biological interpretation. When compared to other linkage types, K33 chains occupy a unique structural and functional niche, often serving as specific recognition modules in signal transduction. Future research should focus on identifying the full repertoire of K33-specific E3 ligases and DUBs, elucidating the role of mixed or branched chains containing K33 linkages, and exploring the therapeutic potential of targeting this pathway in autoimmune diseases, cancer, and neurological disorders where ubiquitin signaling is disrupted.
References
- 1. 4XYZ: Crystal structure of K33 linked di-Ubiquitin [https://www.rcsb.org/structure/4XYZ]
- 2. PDB Entry - 4XYZ (pdb_00004xyz) [https://www.wwpdb.org/pdb?id=pdb_00004xyz]
- 3. Supplementary File [https://rjsocmed.com/0132-3423/rt/suppFileMetadata/670826/0/4301158]
- 4. Specifying conformational heterogeneity of multi-domain ... [https://www.sciencedirect.com/science/article/pii/S0969212623002502]
- 5. Assembly and Specific Recognition of K29- and K33- ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4386031/]
- 6. Exploring linkage dependence of polyubiquitin conformations ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2813430/]
- 7. Assembly and Specific Recognition of K29- and K33- ... [https://www.sciencedirect.com/science/article/pii/S109727651500091X]
- 8. Assembly and specific recognition of k29- and k33-linked ... [https://pubmed.ncbi.nlm.nih.gov/25752577/]
- 9. Article K33-Linked Polyubiquitination of Coronin 7 by Cul3- ... [https://www.sciencedirect.com/science/article/pii/S1097276514002688]
- 10. Ubiquitin Modifying Enzymes and Regulation of the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5675782/]
- 11. Ubiquitin Linkage-Specific Affimers Reveal Insights into K6 ... [https://www.sciencedirect.com/science/article/pii/S1097276517306172]
- 12. Myosin VI Contains a Compact Structural Motif that Binds to ... [https://www.sciencedirect.com/science/article/pii/S2211124716301863]
- 13. specifying-conformational-heterogeneity-of-multi-domain- ... [https://www.bohrium.com/paper-details/specifying-conformational-heterogeneity-of-multi-domain-proteins-at-atomic-resolution/896606063153906006-5026]
- 14. -specific conformational ensembles of non-canonical... Linkage [https://pubs.rsc.org/EN/content/articlelanding/2016/cp/c5cp04601g]
- 15. Secondary interactions in ubiquitin-binding domains ... [https://www.sciencedirect.com/science/article/pii/S221112472400874X]
- 16. Structural visualization of HECT-type E3 ligase Ufd4 ... [https://www.nature.com/articles/s41467-025-59569-6]
- 17. Unraveling chain specific ubiquitination in cells using ... [https://www.nature.com/articles/s41598-025-07242-9]
- 18. Unraveling Chain specific Ubiquitination in Cells Using ... [https://lifesensors.com/unraveling-chain-specific-ubiquitination-in-cells-using-tandem-ubiquitin-binding-entities-in-a-high-throughput-assay/]
- 19. AlphaFold modeling of polyubiquitin complexes and ... [https://www.sciencedirect.com/science/article/pii/S2666386425003959]
- 20. K33-linked polyubiquitination of T cell receptor-ζ regulates ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2927827/]
- 21. 4Y1H: Crystal structure of K33 linked tri-Ubiquitin [https://www.rcsb.org/structure/4y1h]
- 22. The deubiquitinase TRABID stabilizes the K29/K48-specific ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7948964/]
- 23. Structural insights into a HECT-type E3 ligase AREL1 and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6937569/]
- 24. AREL1 Gene [https://maayanlab.cloud/Harmonizome/gene/AREL1]
- 25. Assembly and specific recognition of k29- and k33-linked ... [https://elliottlab.web.ox.ac.uk/publication/976836/pubmed]
- 26. Assembly and specific recognition of k29- and k33-linked ... [https://thebiogrid.org/188671/publication/assembly-and-specific-recognition-of-k29--and-k33-linked-polyubiquitin.html]
- 27. Mechanism and regulation of the Lys6-selective ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5757785/]
- 28. Neutron-encoded diubiquitins to profile linkage selectivity ... [https://www.nature.com/articles/s41467-023-37363-6]
- 29. Structure-based in silico identification of ubiquitin-binding ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3579145/]
- 30. Ubiquitination detection techniques - PMC - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC10625345/]
- 31. Protocol for Determining Ubiquitin Chain Linkage [https://www.rndsystems.com/resources/protocols/determine-ubiquitin-chain-linkage]
- 32. Discovery and mechanism of K63-linkage-directed ... [https://www.nature.com/articles/s41589-024-01777-0]
- 33. Enzymatic Assembly of Ubiquitin Chains [https://pubmed.ncbi.nlm.nih.gov/30242704/]
- 34. Quantitative Analysis of Diubiquitin Isomers Using Ion ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10712319/]
- 35. Generating a conformational landscape of ubiquitin chains ... [https://www.frontiersin.org/journals/chemistry/articles/10.3389/fchem.2022.1087963/full]
- 36. Structural elucidation of ubiquitin via gas-phase ion ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11161031/]
- 37. Structural basis of K11/K48-branched ubiquitin chain ... [https://www.nature.com/articles/s41467-025-64719-x]
- 38. Unique structural, dynamical, and functional properties of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3802530/]
- 39. K33-Linked Di-Ubiquitin (Phosphorylated) [https://www.enzo.com/product/k33-linked-di-ubiquitin-phosphorylated/]
- 40. K11-Linked Polyubiquitination in Cell Cycle Control ... [https://www.sciencedirect.com/science/article/pii/S109727651000523X]
- 41. K33-Linked Polyubiquitination of T Cell Receptor-ζ ... [https://www.sciencedirect.com/science/article/pii/S107476131000244X]
- 42. Current methodologies in protein ubiquitination characterization [https://cellandbioscience.biomedcentral.com/articles/10.1186/s13578-022-00870-y]
- 43. Combinatorial ubiquitin code degrades deubiquitylation- ... [https://www.nature.com/articles/s41467-025-57873-9]
- 44. Decoding the ubiquitin network: molecular mechanisms and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12492556/]
- 45. Regulation of T cell receptor complex-mediated signaling ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4299764/]
- 46. Transitioning from native to synthetic receptors [https://www.nature.com/articles/s41423-025-01304-8]
- 47. Deciphering the deterministic role of TCR signaling in T ... [https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2025.1562248/full]
- 48. Assembly and specific recognition of K29- and K33-linked ... [https://ohsu.elsevierpure.com/en/publications/assembly-and-specific-recognition-of-k29-and-k33-linked-polyubiqu]
- 49. K33-Linked Polyubiquitination of Coronin 7 by Cul3- ... [https://pubmed.ncbi.nlm.nih.gov/24768539/]
- 50. Ubiquitin Puts Actin in Its Place [https://www.sciencedirect.com/science/article/pii/S1097276514004080]
- 51. Mechanism of Arp2/3 complex branch disassembly by ... [https://www.nature.com/articles/s41467-025-64787-z]
- 52. 79585 - Gene ResultCORO7 coronin 7 [ (human)] [https://www.ncbi.nlm.nih.gov/gene/79585]
- 53. K33 only mutant ubiquitin augments cisplatin ... [https://www.sciencedirect.com/science/article/abs/pii/S0898656825003742]
- 54. Mechanism underlying linkage-selective polyubiquitin ... [https://discovery.dundee.ac.uk/en/studentTheses/mechanism-underlying-linkage-selective-polyubiquitin-recognition/]
- 55. 5AF6: Structure of Lys33-linked diUb bound to Trabid NZF1 [https://www.rcsb.org/structure/5af6]
- 56. Secondary interactions in ubiquitin-binding domains achieve ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11372445/]
- 57. Article K29-Selective Ubiquitin Binding Domain Reveals ... [https://www.sciencedirect.com/science/article/pii/S1097276515000908]